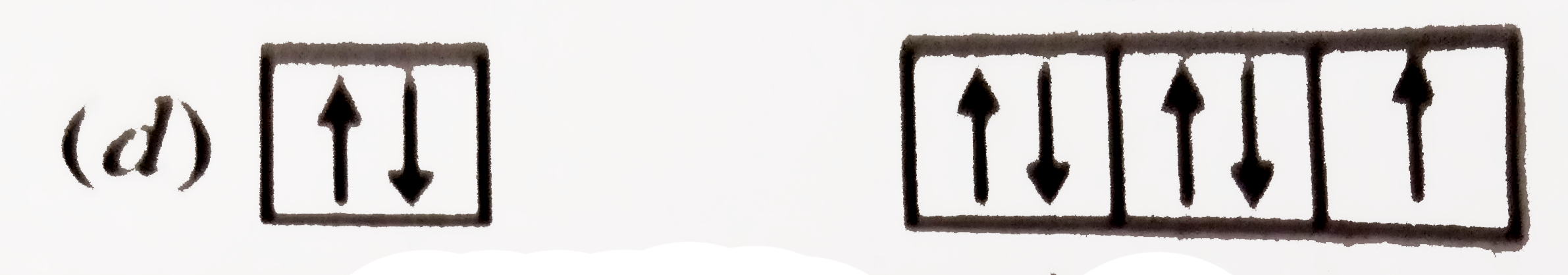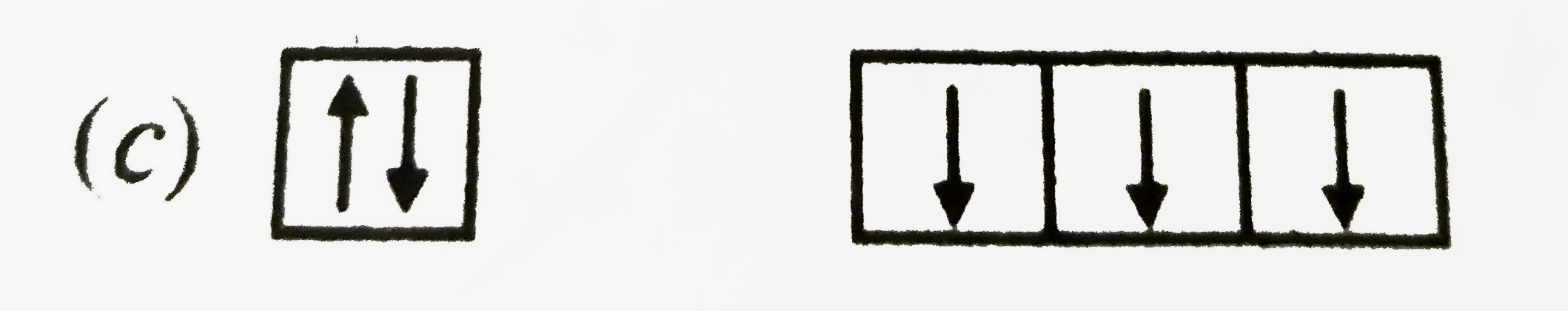Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The order of reactivity of alcohols with sodium metal is |
|
Answer» `3^(@) GT 2^(@) gt 1^(@)` In this REACTION, alcohol acts as an acud. The acidic CHARACTER of `1^(@), 2^(@)` and `3^(@)` ALCOHOLS is in the order `1^(@) gt 2^(@) gt 3^(@)`. |
|
| 2. |
The order of pK_(a) values of the following acids is |
|
Answer» `IV GT I gt III gt II` |
|
| 3. |
The order of leaving group ability is : |
|
Answer» `(i) gt (II) gt (iii) gt (iv)` ACIDITY : `F_3 C - SO_3 H gt MeSO_3 H gt AcOH gt MeOH`. Basicity : `underset (iv) (F_3)C^(Ө) lt underset (iii) MeSO_3^(Ө) lt underset (i) ACO^(Ө) lt underset (ii) MeO^(Ө)` Leaving group : `(iv) gt (iii) gt (i) gt (ii)`. |
|
| 4. |
The order of heat of fusion of T_(2),D_(2) and H_(2) is |
|
Answer» `T_(2) GT D_(2) gt H_(2)` |
|
| 5. |
The order of following reaction is : |
Answer» 
|
|
| 6. |
The order of enol content in the following molecules is |
|
Answer» `a GT B gt C gt d` |
|
| 7. |
The order of filling of electrons in the orbitals of an atom will be |
|
Answer» 3d, 4S, 4P, 4d, 5S |
|
| 8. |
The order of electron gain enthalpy (with negative sign) of F, CI, Br and I is… |
|
Answer» `F LT Cl lt BR lt I` |
|
| 9. |
The order of decreasing stability of the following cations is : CH_(3)CHCH_(3),CH_(3)CHOCH_(3),CH_(3)CHCOCH_(3) |
|
Answer» `III gt II gt I` GROUP is with DRAWING group Resonance stabilises more than HYPER conjugation |
|
| 10. |
The order of decreasing stability of the following carbanions is (i) (CH_(3))_(3)C^(-) (ii) (CH_(3))_(2)CH^(-) (iii) CH_(3)CH_(2)^(-) (iv) C_(6)H_(5)CH_(2)^(-) |
|
Answer» (i)GT(ii)gt(iii)gt(iv) |
|
| 11. |
The order of decreasing stability of the carbanions I) Cbar(H)_(3)"II)"CH_(3)-Cbar(H)_(2)"III)"CH_(3)-Cbar(H)-CH_(3)"IV)" overset(overset(CH_(3)-bar(C)-CH_(3))(|))(CH_(3)) |
|
Answer» `I GT II gt III gt IV` |
|
| 12. |
The order of decreasing stability of the carbanions (CH_3)_3overset(bar(..))C (CH_3)_2overset(bar(..))CH CH_3overset(bar(..))CH_2 C_6H_5(overset(bar(..))CH_2)_2 is |
|
Answer» `1 GT 2 gt 3 gt 4` |
|
| 13. |
The order of decreasing stability of the carbanions (CH_(3))_(3) C^(-) (I), (CH_(3))_(2) bar(C )H(II), CH_(3)CH_(2)(III), C_(6)H_(5)CH_(2)(IV) |
|
Answer» `I gt II gt III gt IV` |
|
| 14. |
The order of decreasingreactivity towards on electrophilic reagent for the following : (i)Benzene, (ii)Toluene , (iii)Chlorobenzoic acid , (iv)Phenol would be |
|
Answer» (IV) GT (II) gt (i) gt (III) |
|
| 15. |
The order of decreasing reactivity towards an electrophilic reagent, for the following: (i) Benzene (ii) Toluene (iii ) Chlorobenzene (iv) phenol would be |
|
Answer» `(i) gt (II) gt (iii) gt (IV)` |
|
| 16. |
The order of decreasing reactivity towards an electophilic reagent, for the following would be (a) Benzene (b) Toluene (c ) Chlorobenzene (d) Phenol |
|
Answer» `d GT B gt a gt C` |
|
| 17. |
Theorderof decreasingnegativeelectrongainenthalpyof O. S, Se, is |
|
Answer» `O GT SGT SE` |
|
| 18. |
The order of decreasing ionization enthalpy in alkali metals is |
|
Answer» `NagtLigtKgtRb` |
|
| 19. |
The order of decreasing ease of reaction with ammonia is |
|
Answer» ANHYDRIDES, ESTERS, ethers |
|
| 20. |
The order of decreasing ionisation enthalpy in alkali metals is |
|
Answer» NagtLigtKgtRb |
|
| 21. |
The order of correct bond energy of C-X bond is |
|
Answer» `C -CL GT C-I gt C-BR` |
|
| 22. |
The correct order of O-O bond length in O_(2), H_(2)O_(2) and O_(3) is |
|
Answer» `O_(2)gtH_(2)O_(2)gtO_(3)` |
|
| 23. |
The order of Canizzarro ' s reaction when base concentration is high. |
|
Answer» |
|
| 24. |
The order of bond strength among C-F, C-Cl, C-Br, C-1 is |
|
Answer» `C-F gt C-CL gt C-Br gt C-I` |
|
| 25. |
The order of basicity is (I) Ph-CONH_(2)""(II)Ph-NH_(2)""(III)Ph-CH_(2)-NH_(2)""(IV)p-OCH_(3)Ph-NH_(2) |
|
Answer» `II gt IV gt I gt III` |
|
| 26. |
The order of basicity of the following compounds is |
|
Answer» `I GT II gt IV gt III` |
|
| 27. |
The order of acidity of the H-atoms underlined in the following compounds is in the order- (I) Ph-Cul(H)_(2)-CH_(3)""(II) Ph-C-=Cul(H)""(III) Ph-CH=CulH_(2)""(IV) |
|
Answer» `IV GT II gt I gt III` |
|
| 28. |
The order of abundance of IIIA group elements is |
|
Answer» `Al GT Ga gt B gt TI gt "In "` |
|
| 29. |
The orbitals which have same number of nodes are |
|
Answer» 2S, 2p `:.` Orbitals having same VALUE of n have same no. of nodes |
|
| 30. |
The orbital overlapping is maximum in |
|
Answer» `Cl_2` `sigmap - p GT SIGMA s- p gt sigmas - s` |
|
| 31. |
The orbital without nodal planes is |
|
Answer» 1s |
|
| 33. |
The orbital diagram in which both the Pauli's exclusion principle and Hund's rule are violated is |
|
Answer»
|
|
| 34. |
The orbital diagram in which both the Pauli.s exclusion principle and Hund.s rule are violated, is: |
|
Answer»
|
|
| 35. |
The orbitalangularmomentum ofa p-electronis given as |
|
Answer» `(h)/(SQRT(2pi))` |
|
| 36. |
The orbital angular momentum of a p-electron is given as: |
|
Answer» `SQRT((3)/(2)) (h)/(PI)` Orbital angular momentum `= sqrt(l(l + 1)) (h)/(2pi) = sqrt(1(1 + 1)) (h)/(2pi)` `= SQRT2 (h)/(2pi) =(h)/(sqrt2pi)` |
|
| 37. |
The orbital angular momentum of a p-electron is given as |
|
Answer» `h/(sqrt2pi)` l=1 So =`h/(2pi)sqrt2 =h/(sqrt2pi)` |
|
| 38. |
The orbital angular momentum of 3p electrons is sqrt(x)(h)/(2pi). Then the value of x is ______. |
|
Answer» `=SQRT(l(l+1)) (h)/(2pi), l =1 ` for p - orbital. |
|
| 39. |
The orbital angular momentum of 3p electrons is : |
| Answer» Answer :D | |
| 40. |
The orange solid ono heating gives a colourless gas and green solid which can be reduced to the metal by aluminium powder. The orange and the green solies are, respectively, |
|
Answer» `(NH_(4))_(2)Cr_(2)O_(7)andCr_(2)O_(3)` `Cr_(2)O_(3)+2Aloverset(Delta)to2Cr+Al_(2)O_(3)` |
|
| 41. |
The option(s) with only amphotericoxides is(are) ? |
|
Answer» `CrO_(3),BeO,SnO,SnO_(2)` |
|
| 42. |
The orange coloured compound formed when H_(2) O_(2) is added to TiO_(2) solution acidified with conc H_(2) SO_(4) is |
|
Answer» `TiO_3` |
|
| 43. |
The orange coloured compound formed when H2O2 is added TiO2 lon acidified with conc H_(2)SO_(4) is |
|
Answer» `TiO_(30` |
|
| 44. |
The optimum pressure and temperature for the production of NH_3 by Haber's process is ………. |
|
Answer» 350 BAR, 760 K |
|
| 45. |
The optically active tartaric acid is named as D-(+)-tartaric acid because it has a positive. |
|
Answer» optical rotation and is DERIVED from D-glucose |
|
| 46. |
The optical rotation of a solution of pure natural camphor is found to be +5.76^(@) under the following conditions, concentration = 0.13g/ml, length of polarimeter = 1dm, wavelength = sodium D line, T=25^(@)C. The specific rotation of camphor is |
|
Answer» `+44.3^@` `((alpha) " = Observed angle of rotation" (THETA))/(c XX 1)` ` = (+576)/(0.13 xx 1) = + 44.3^@` |
|
| 47. |
The ONO bond angle is maximum in |
|
Answer» `NO_(3)^(-)` Structures 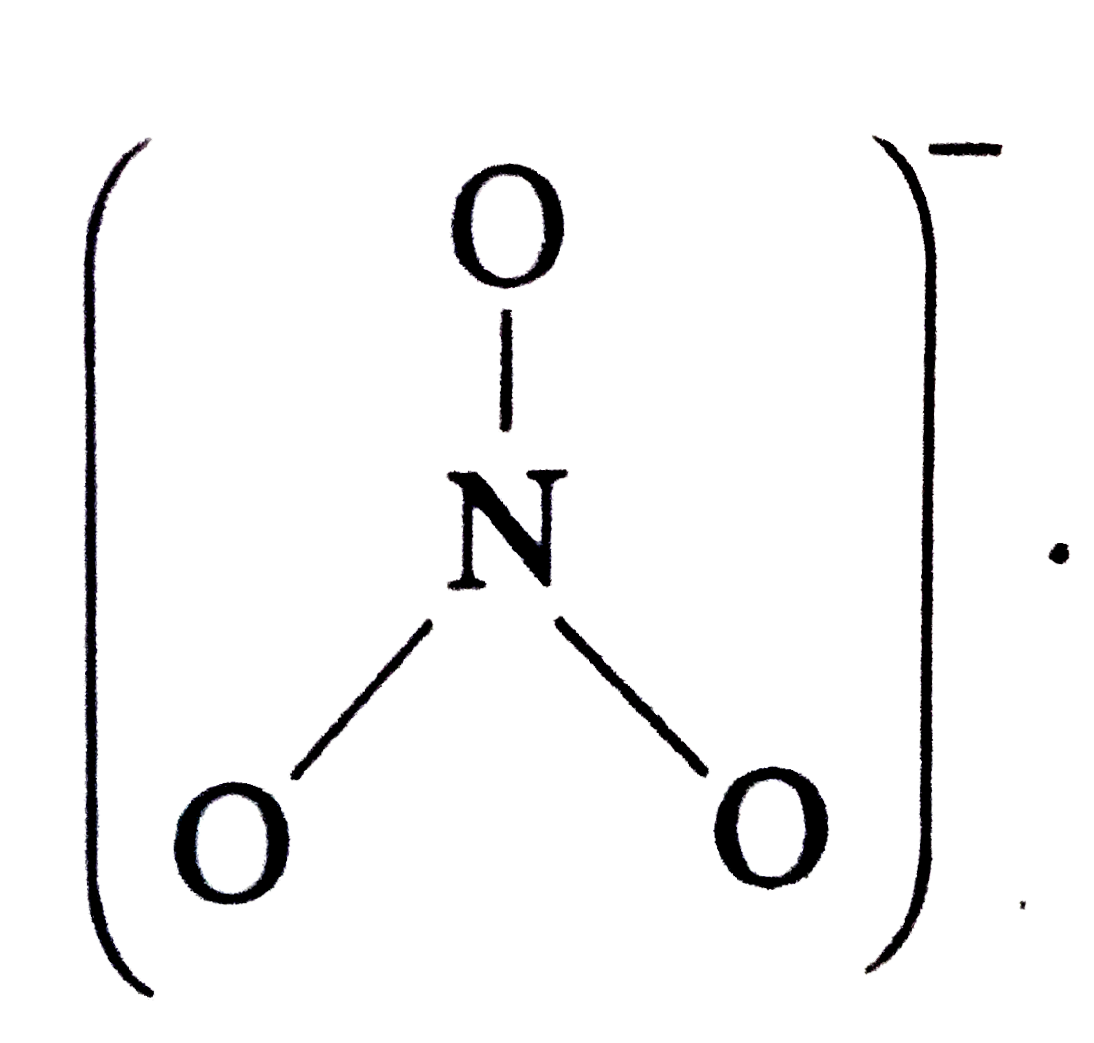 Hence , ONO BOND anlge `= 120^(@)` `NO_(2)^(+)` has no unshared electron . It has only bond PAIRS in two directions . Hence, SHAPE is linear `(underset(. .)overset(. .)O=overset(+)N=underset(. .)overset(. .)O)` with bond angle = `180^(@)` `NO_(2)` has one unshared electron whereas `NO_(2)^(-)` has one unshared electron pair . Hence , in `NO_(2)^(-)` , the repulsion on the bond pairs are more and angle is less 
|
|
| 48. |
The only motion exhibited by the constituent particles of a solid is ……………… |
|
Answer» |
|
| 49. |
The only metal which exist in nature as well as combined state is |
|
Answer» Al |
|
| 50. |
The only electron in the hydrogen atom resides under ordinary conditions on the first orbit. When energy is supplied, the electron moves to higher energy orbit depending on the amount of energy absorbed. When this electron returns to any of the lower orbits, it emits energy. Lyman series is formed when the electron returns to the lowest orbit while Balmer series is formed when the electron returns to second orbit. Similarly, Paschen, Brackett and Pfund series are formed when electron returns to the third, fourth orbits from higher energy orbits respectively (as shown in figure) Maximum number of lines produced when an electron jumps from nth level to ground level is equal to (n(n-1))/(2). For example, in the case of n = 4, number of lines produced is 6. (4 rarr 3, 4 rarr 2, 4 rarr 1, 3 rarr 2, 3 rarr 1, 2 rarr 1). When an electron returns from n_(2) to n_(1) state, the number of lines in the spectrum will be equal to ((n_(2) - n_(1))(n_(2)-n_(1) +1))/(2) If the electron comes back from energy level having energy E_(2) to energy level having energy E_(2) then the difference may be expressed in terms of energy of photon as E_(2) - E_(1) = Delta E, lambda = (h c)/(Delta E). Since h and c are constant, Delta E corresponds to definite energy, thus each transition from one energy level to another will prouce a higher of definite wavelength. THis is actually observed as a line in the spectrum of hydrogen atom. Wave number of the line is given by the formula bar(v) = RZ^(2)((1)/(n_(1)^(2)) - (1)/(n_(2)^(2))) Where R is a Rydberg constant (R = 1.1 xx 10^(7)) (i) First line of a series : it is called .line of logest wavelength. or .line of shortest energy.. (ii) Series limit of last of a series : It is the line of shortest wavelength or line of highest energy. Let v_(1) be the frequency of the series limit of the Lyman series, v_(2) bethe frequency of the first line of the Lyman series, and v_(3) be the frequency of the series limit of the Balmer series |
|
Answer» `v_(1) - v_(2) = v_(3)` |
|