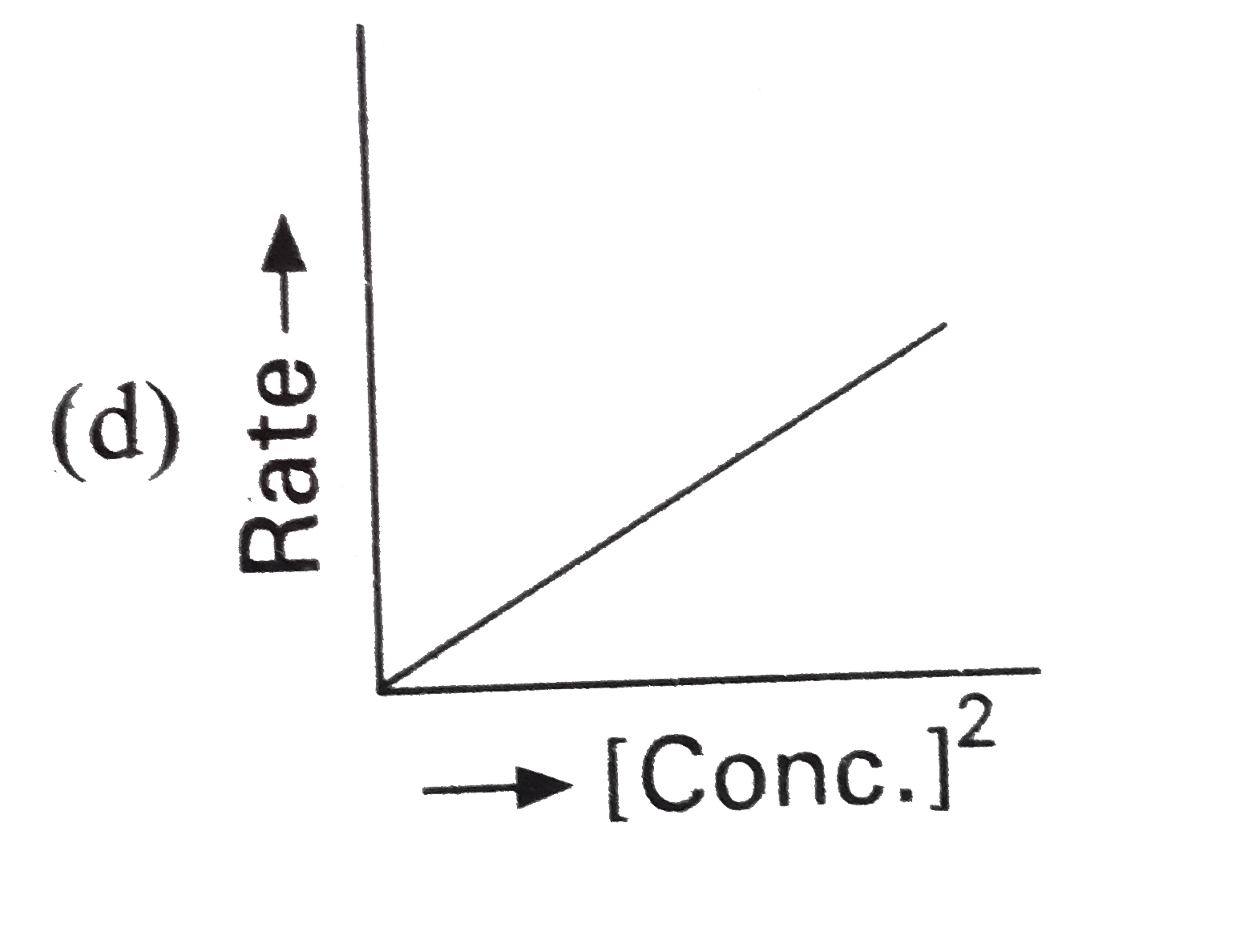Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What is Lindlar's catalyst ? Write its uses. |
| Answer» Solution :Lindlar.s catalyst : It is partially inert palladised CHARCOAL. This palladised charcoal is treated with sulphur compound or QUINOLINE like poisonous compound and made it inert. It is USED to have partial reduction of ALKYNE comopunds and form cis-alkene. | |
| 2. |
Which one of the following shows functional isomerism? |
|
Answer» ethylene |
|
| 3. |
when a tyre is pumped up rapidly, its temperature rises, would you expect the same effect if air were an ideal gas? |
|
Answer» SOLUTION :146.6 pm Given `b=0.0318Lmol^(-1)=4XX((4)/(3)pir^(3))` `thereforer=((bxx3)/(16pi))^(1//3)` |
|
| 4. |
Threeunpairedelectronof nitrogenatom can be examplainedby … |
|
Answer» Aufbauprinciple |
|
| 5. |
Write the oxidation number of Hg in amalgam. |
| Answer» SOLUTION :Oxidation number of MERCURY in AMALGAM is ZERO.Oxidation number of each constituent in an alloy or in an amalgam is zero. | |
| 6. |
What is the hybridisation of each carbon in H_(2)C = C = CH_(2) ? |
|
Answer» Solution :The gross structure of allenen `(C_(3)H_(4))` is : `H_(2) underset(underset(sp^(2))(darr))overset(1)( C)= underset(underset(sp)(darr))overset(2)(C )= underset(underset(sp^(2))(darr))overset(3)(C )H_(2)` The CARBON atom 1 and3 are `sp^(2)` hybridized since each ONE of then is joined by a double bond. Fn contrast, carbon atom 2 is sp-hybridized since it has two double bonds. Thus, the two `pi`-bonds in alkene like in acetylene are prependicular to each other are shown below.  `2p^(y) and 2p_(X)` orbitals of carbon `C_(2)` do not take part in HYBRIDIZATION on only `2p_(x)` take part so `C_(2)` has sp. |
|
| 7. |
The type of overlaping not observed in the formation of ethylene molecule is |
|
Answer» `rho SP^(2)-sp^(2)` |
|
| 8. |
What is meant by plasma state? Give and example. |
| Answer» Solution :Gaseous state of MATTER at very high TEMPERATURE containing gaseous ions and free ELECTRON is REFERRED to as the Plasma state e.g. Lightning. | |
| 9. |
What would be the major product in the following reactions ? |
|
Answer» |
|
| 10. |
Which hybird orbitals are used by carbon atoms in the following molecules?CH_3-CH_2-OH |
Answer» SOLUTION :
|
|
| 11. |
Water gas is 3 times move efficient than producer gas as fuel. Why? |
| Answer» SOLUTION :Both the main components of water gas are combustible. But in PRODUCER gas about two-thirds is nitrogen and is non-combustible. The calorific value of water gas is `13000 KJM^(-3)` but of producr gas is less than `4000 kJm^(-3)`? | |
| 12. |
What is osmosis? |
| Answer» SOLUTION :Osmosis is a SPONTANEOUS PROCESS by which the SOLVENT molecules pass through a semipermeable MEMBRANE from a solution of lower concentration to the solution of higher concentration. | |
| 13. |
Which of the following metal reacts with cold water as well as hot water |
|
Answer» Ca |
|
| 14. |
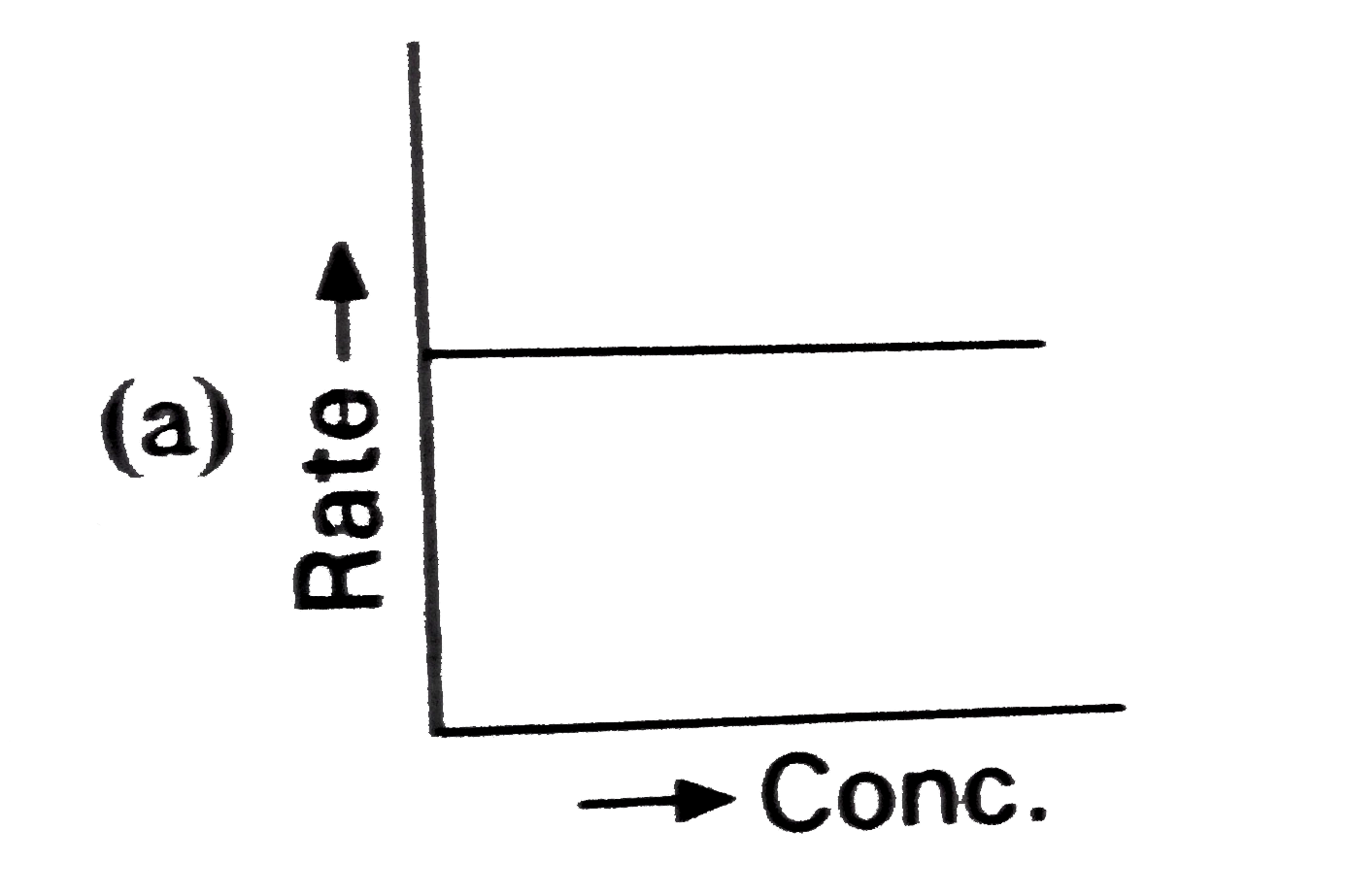
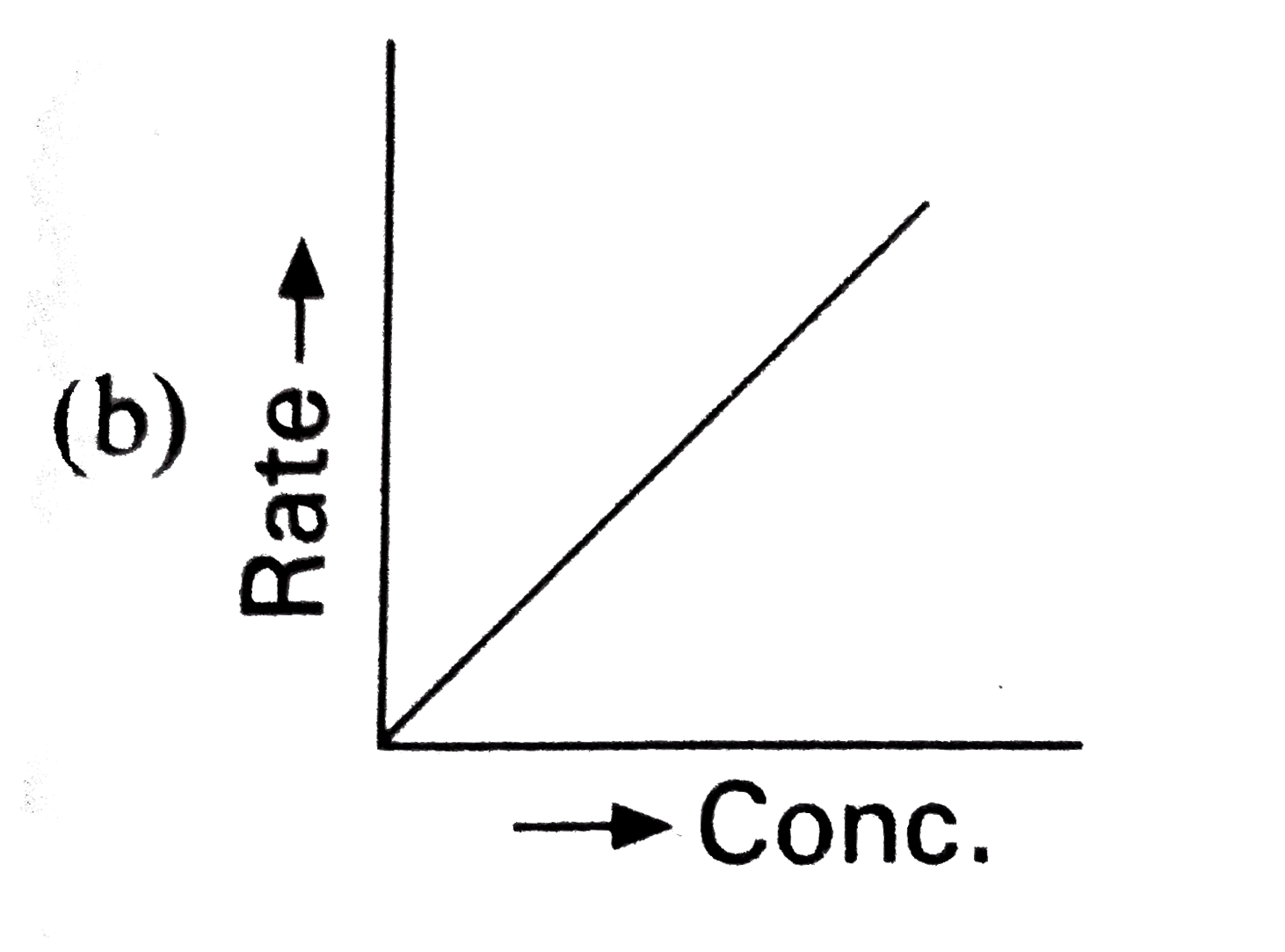
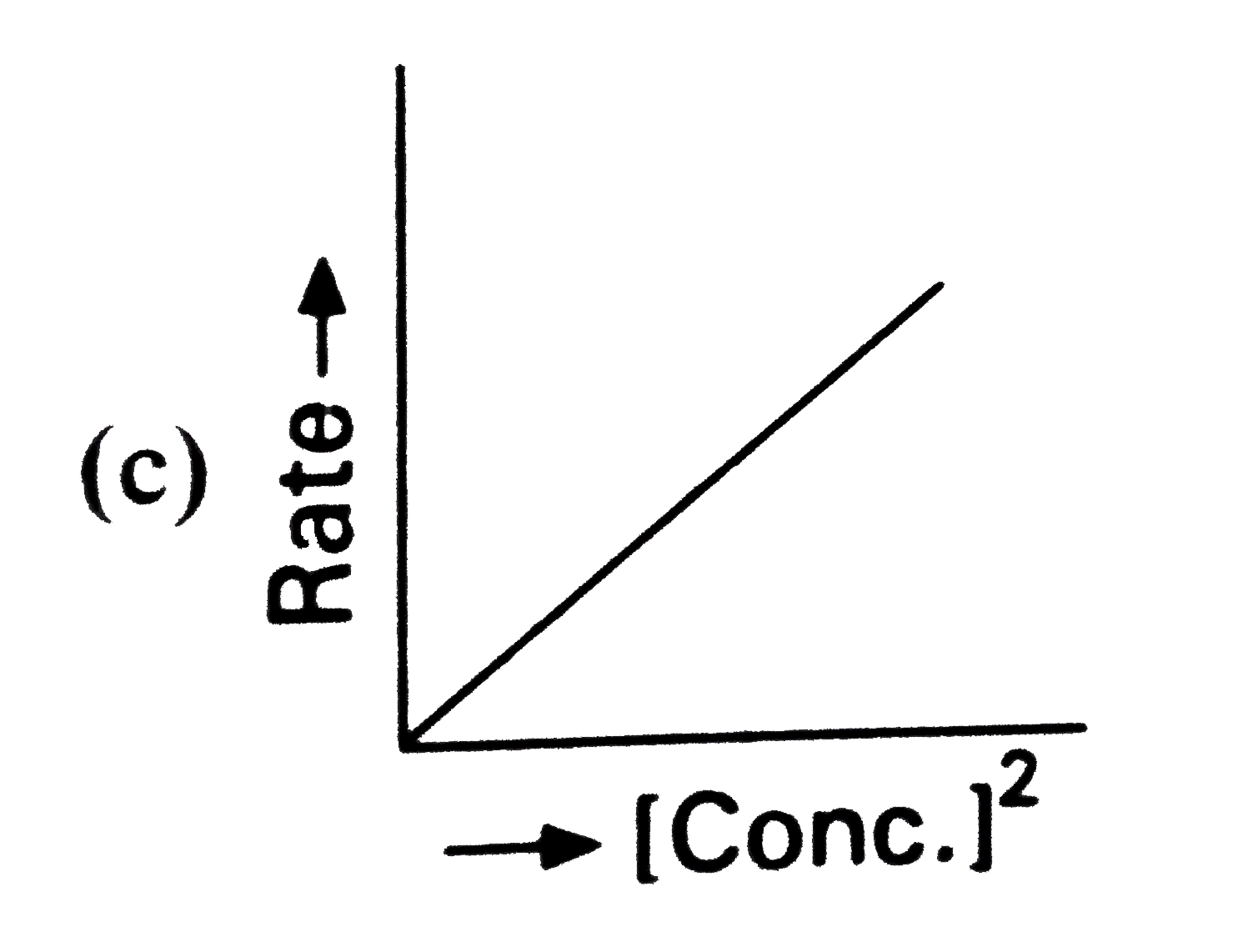
Which of the following represents zero order reaction ? |
|
Answer»
|
|
| 15. |
What effect does branching of an alkane chain has on its boiling point? |
| Answer» Solution :The boiling point of an alkane DECREASES with increase in branching. This is because the branching MAKES the molecule more compact and decreases its surface area. This decreases the MAGNITUDE of van DER Waals. forces existing between the molecules. On ACCOUNT of this, boiling point decreases. | |
| 16. |
What is functional group ? Give example |
|
Answer» SOLUTION :The functional group may be defined as an atom or group of atoms joined in a specific manner which is RESPONSIBLE for the characteristic chemical properties of the organic compounds Examples: (i) `(-OH)` hydroxyl group i.e., `CH_(3)OH` (ii) `(-CHO)` Aldehyde groupi.e., `CH_(3)CHO` (III) `(-COOH)` Carboxylic acid group `CH_(3)COOH`  Some IMPORTANT functional group are of under (a) Carbon -carbon multiple bond containing functional group:  (b) Atoms other then carbon CONNECTED with carbon such functional group:  
|
|
| 17. |
What is the compressibility facto (Z) for 0.02 mole of a van der Waal's gas at pressure of 0.1 atm. Assume the size of gas molecules is neligible. Given : RT = 20 L. atm "mol^(-1) and alpha = 1000 atm L^(2) "mol^(-2) |
|
Answer» 2 `0.1 V^2 - 0.4 V + 0.4 = 0`. `= V^2 - 4V + 4 = 0 implies V = 2L` `Z = (PV)/(nRT) = (0.1 xx 2)/(20 xx 0.02) = 0.5`. |
|
| 18. |
What is the difference between the terms orbit and orbital ? |
Answer» SOLUTION :The DIFFERENCE between ORBIT and ORBITAL are as below
|
|
| 19. |
Which one of the following is an isobar of ""_(6)C^(14) ? |
| Answer» Answer :C | |
| 20. |
Which one of the following is called benzylchloride? |
|
Answer» `C_(6)H_(5)CH_(2)Cl` |
|
| 21. |
Which of the following statements is/are not correct ? |
|
Answer» Aluminium wire is used in Beilstein test (d) Estimated magnesium pyrrophosphate `(Mg_(2)P_(2)O_(7))`. |
|
| 22. |
The sodium fusion extract of an organic compound on acidification with acetic and addition of lead acetate solution gives a back precipitate. The organic compound contains : |
|
Answer» NITROGEN |
|
| 23. |
What is the basic theme of orgainisation in the periodic table? |
| Answer» Solution :It is to simplify and systematise the study of the PROPERTIES of all the elements and MILLIONS of their compounds. On the basis of similarities in chemical properties, the various elements have now been divided into different groups. This has MADE the study simple because the properties of elements are now STUDIED in the form ofagroups rather than INDIVIDUALLY. | |
| 24. |
The successive ionisation enthalpies of an element M are 5.98, 18.82, 28.44, 119.96, 153.77, ...... e V atom What is the formula of chloride of M? |
|
Answer» Solution :Observing the `I_1, I_2, I_3, I_4, I_5`, .... it is NOTICED that there is a sudden jump from `I_3` and `I_4` This observation gives the idea that the element has 3 ELECTRONS in the OUTER most shell. `M^(3+)` state is STABLE and valency is 3. Formula of CHLORIDE of `M`is `MCI_3` |
|
| 25. |
Which of the following is the ore name related to 'Mg' |
|
Answer» Brucite Anhydrite `CaSO4` Baryta `BaSO_(4)` |
|
| 26. |
Which of thefollowing is not an air pollutant? |
|
Answer» `N_2` |
|
| 27. |
What is the maximum number of electrons in a element possible ? |
| Answer» SOLUTION :DOUBLE NUMBER of ELECTRONS are PRESENT than sub-orbitals in an element. | |
| 28. |
Which one of the following compounds, decolourises cold alkaline potassium permanganate solution? |
|
Answer» `C_(2)H_(6)` |
|
| 29. |
Two containers, X and Y at 300K and 350K with water vapour pressures 22 mm and 40 mm respectively are connected, initially closed with a valve. If the valves opened, |
|
Answer» the FINAL pressure in each container is 31 mm In container `Y, l to G` to maintain same V.P In container `X, g to l` to maintain same V.P . |
|
| 30. |
Total number of lone pairs of electrons in I_(3)^(-) ion is |
|
Answer» 3 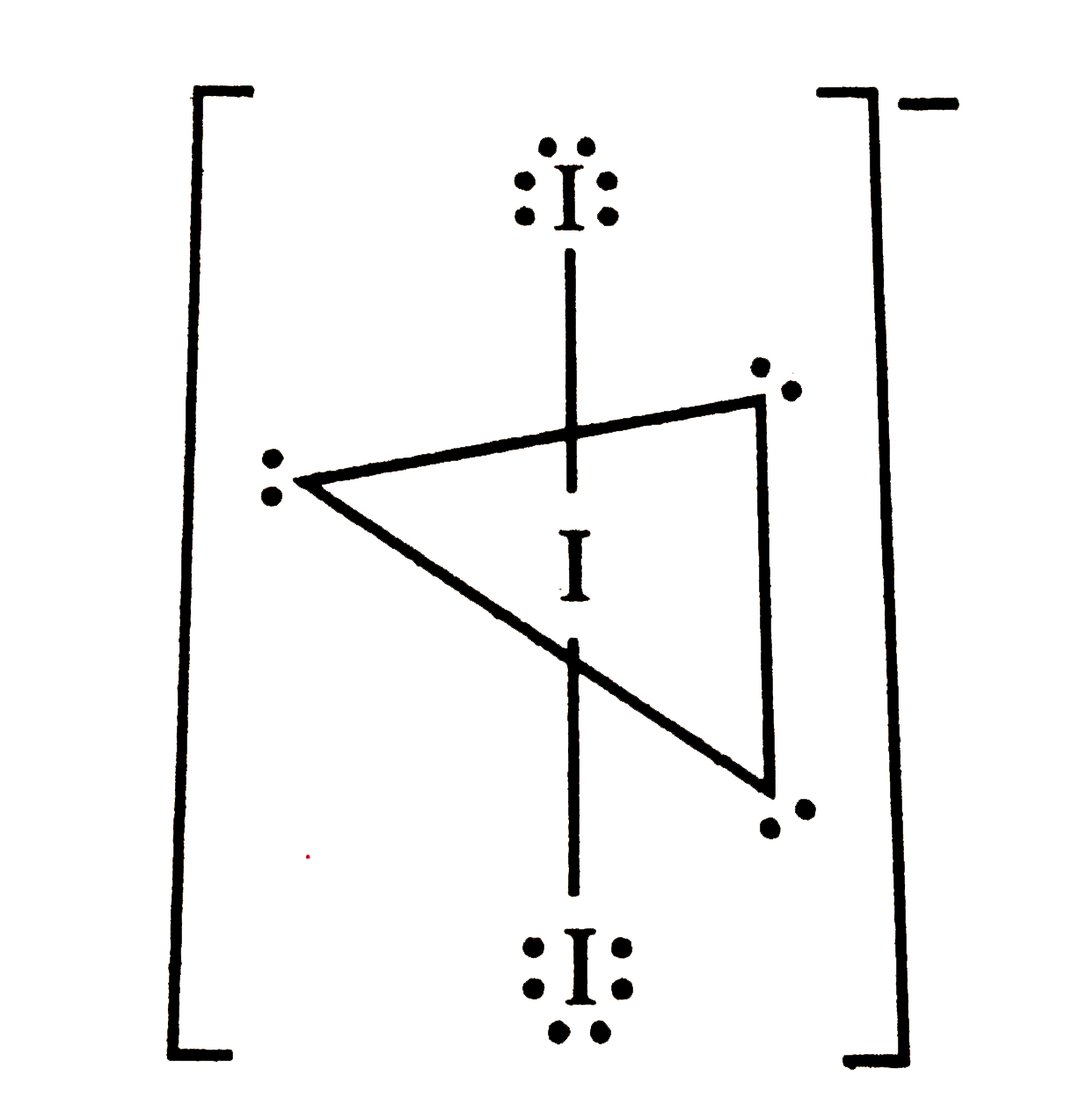 THEREFORE, total number of lone pairs of ELECTRONS in `I_(3)^(-)` ion is 9. |
|
| 31. |
Which of the following salt is not produce from the solvey ammonia soda process ? |
|
Answer» `Na_(2)CO_(3)` |
|
| 32. |
Which physical property is lower for alkaline earth metals as compared to alkali metals of the same period ? |
|
Answer» Hardness |
|
| 33. |
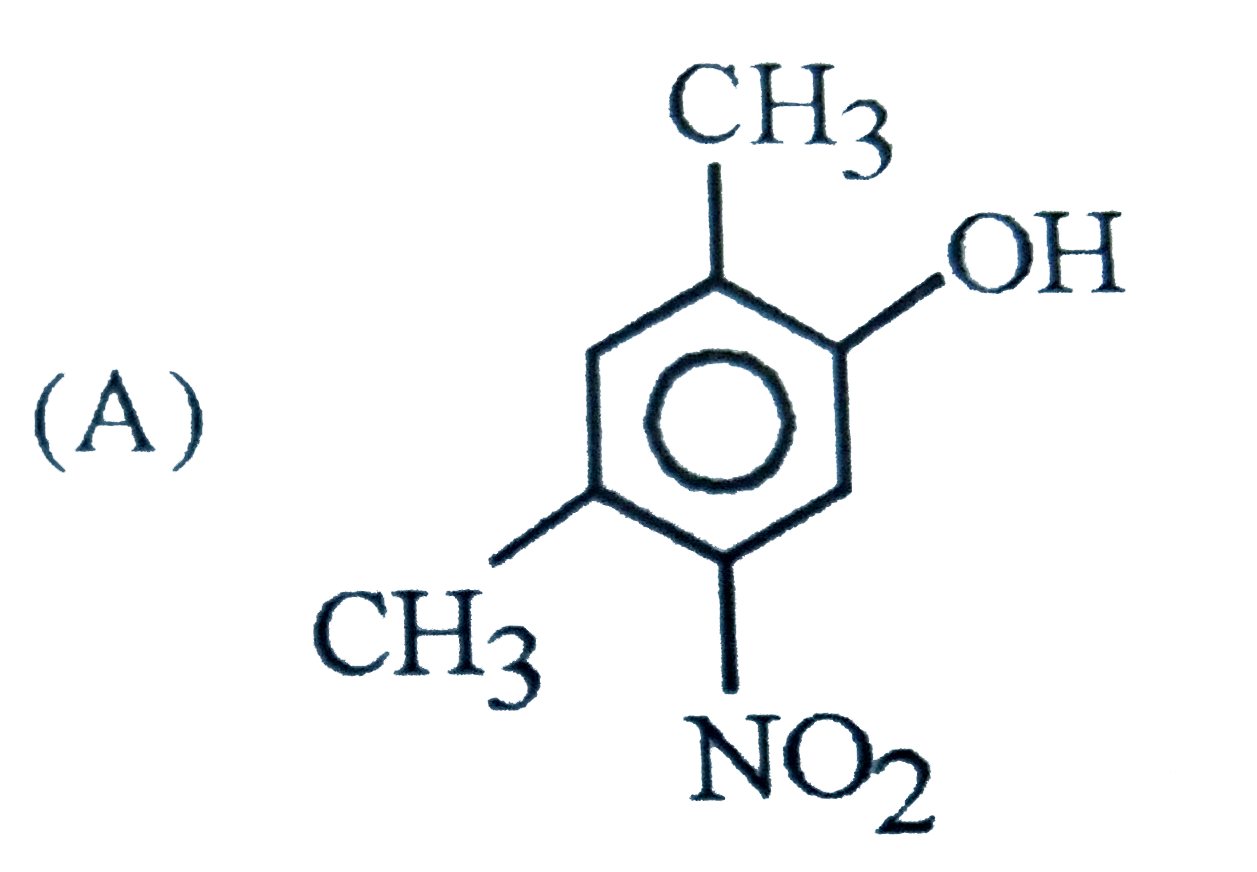
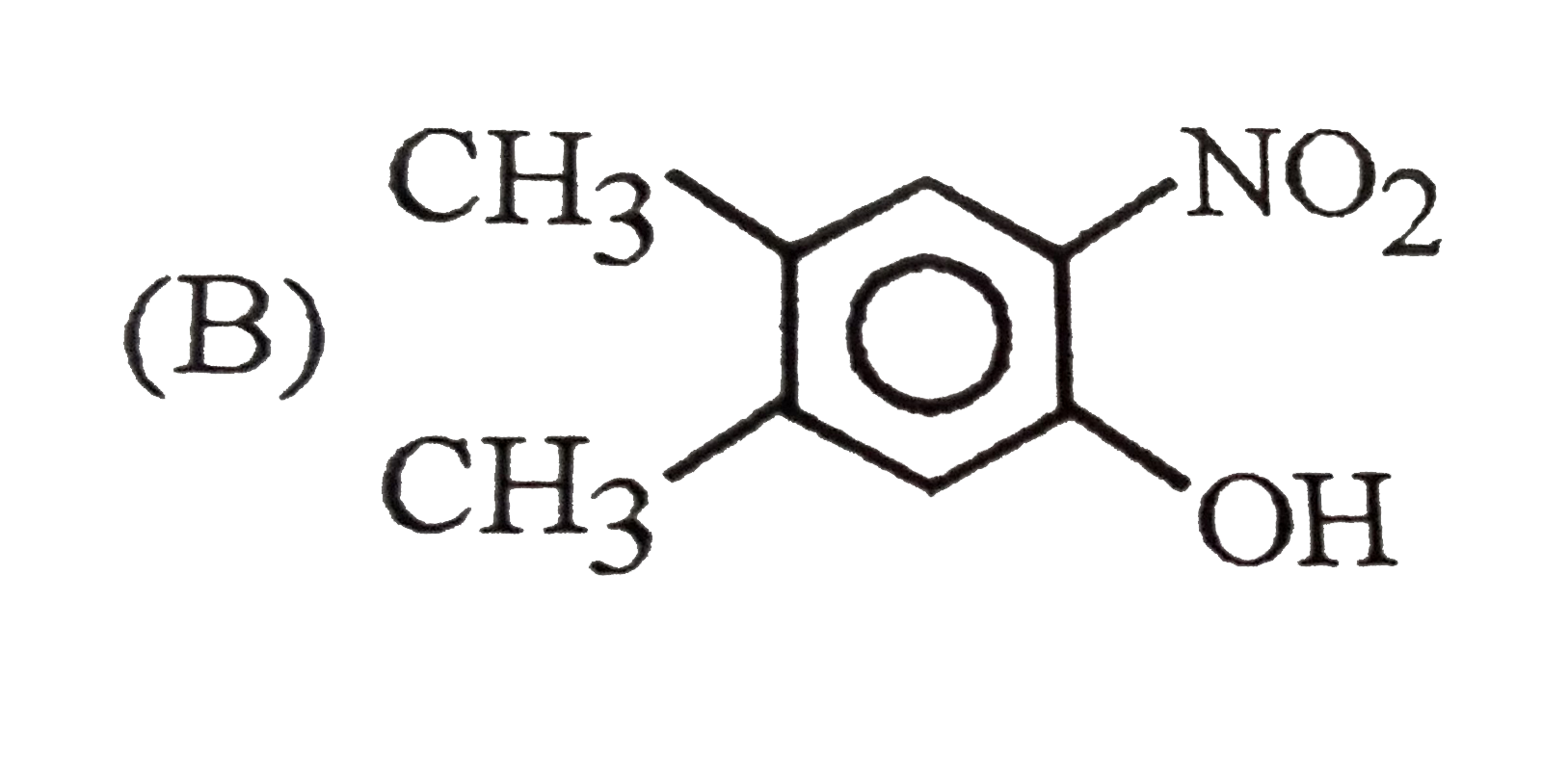
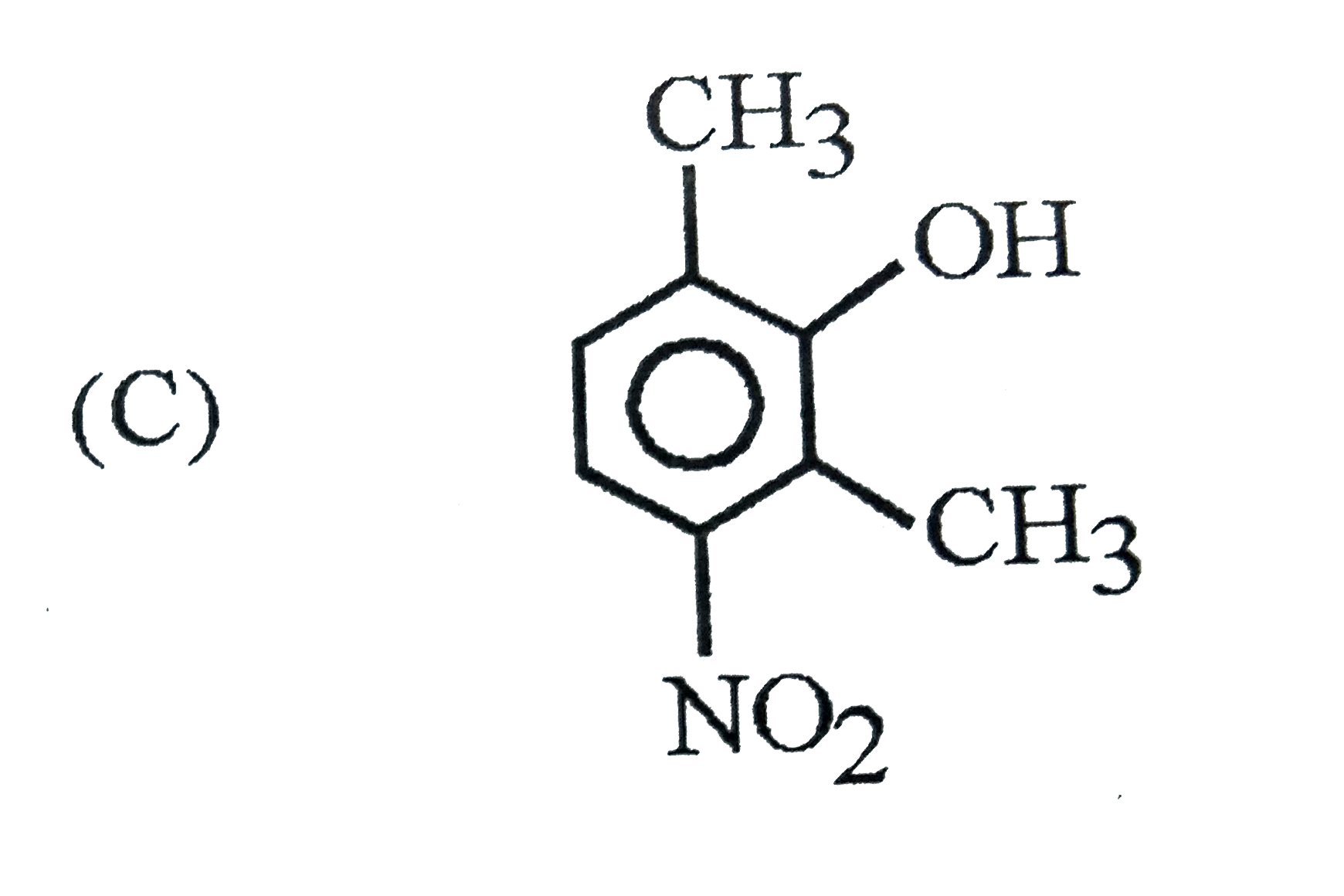
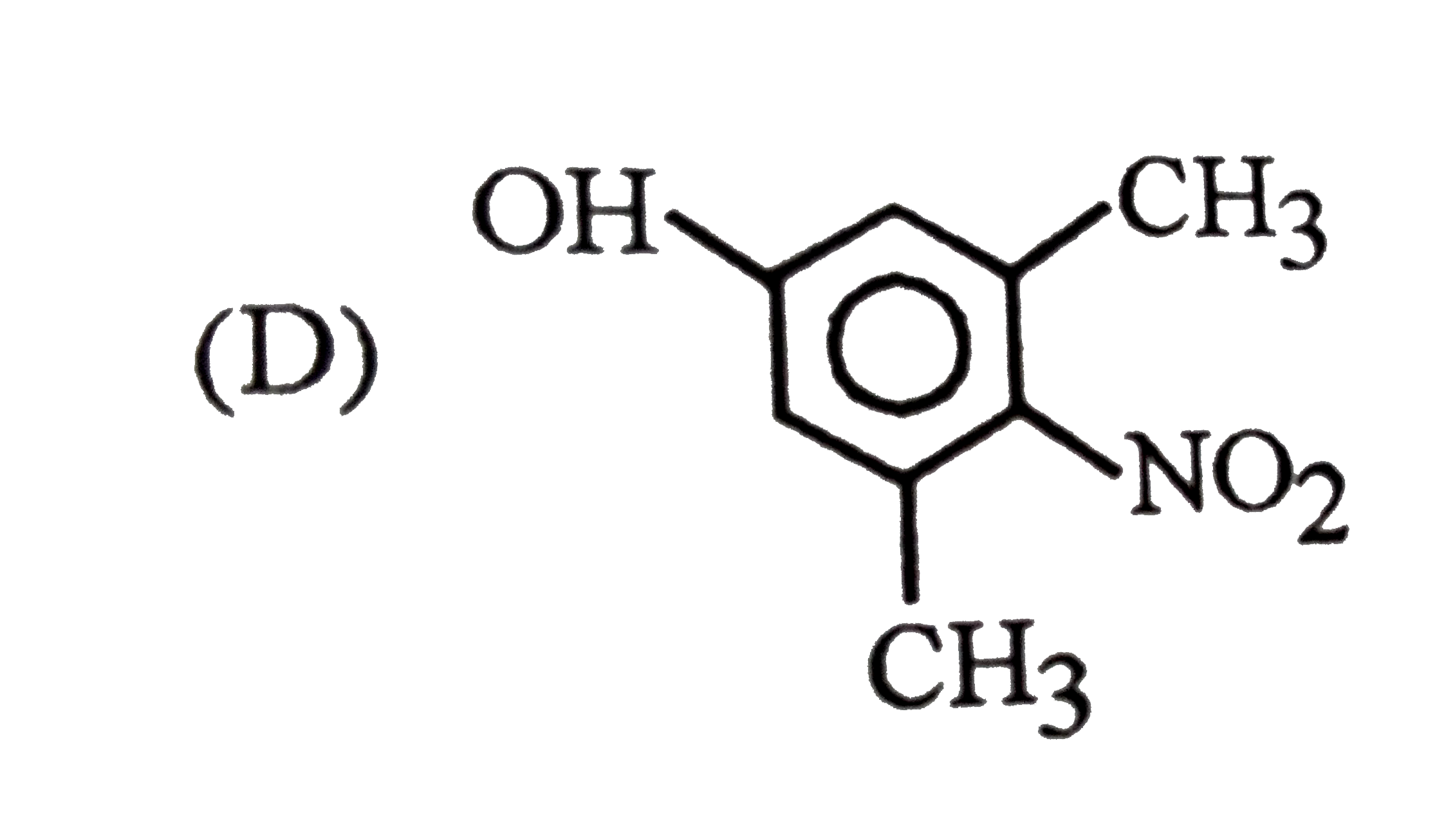
Which of the following phenol is strongest acid |
|
Answer»
|
|
| 34. |
Which is the function of sodium-potasssium pump? |
|
Answer» MAINTENANCE of ion balance |
|
| 35. |
Which of the following is (are) not an air pollutant (s) |
|
Answer» CO |
|
| 36. |
When alpha particles are sent through a thin metal foil, most of them go straight through the foil because: |
|
Answer» alpha PARTICLES are MUCH heavier than electrons |
|
| 37. |
What is the condition spontaneity in terms of free energy change? |
|
Answer» SOLUTION :(i)If`DeltaG`is negative, process is SPONTANEOUS. (II)If `DeltaG` is positive, process is non-spontaneous. (iii) If `DeltaG=0`, the process is in equilibrium |
|
| 38. |
Which of the following combination can be used to synthesise isopropyl alcohol ? |
|
Answer» `CH_(3)MgI` and `CH_(3)COCH_(3)` 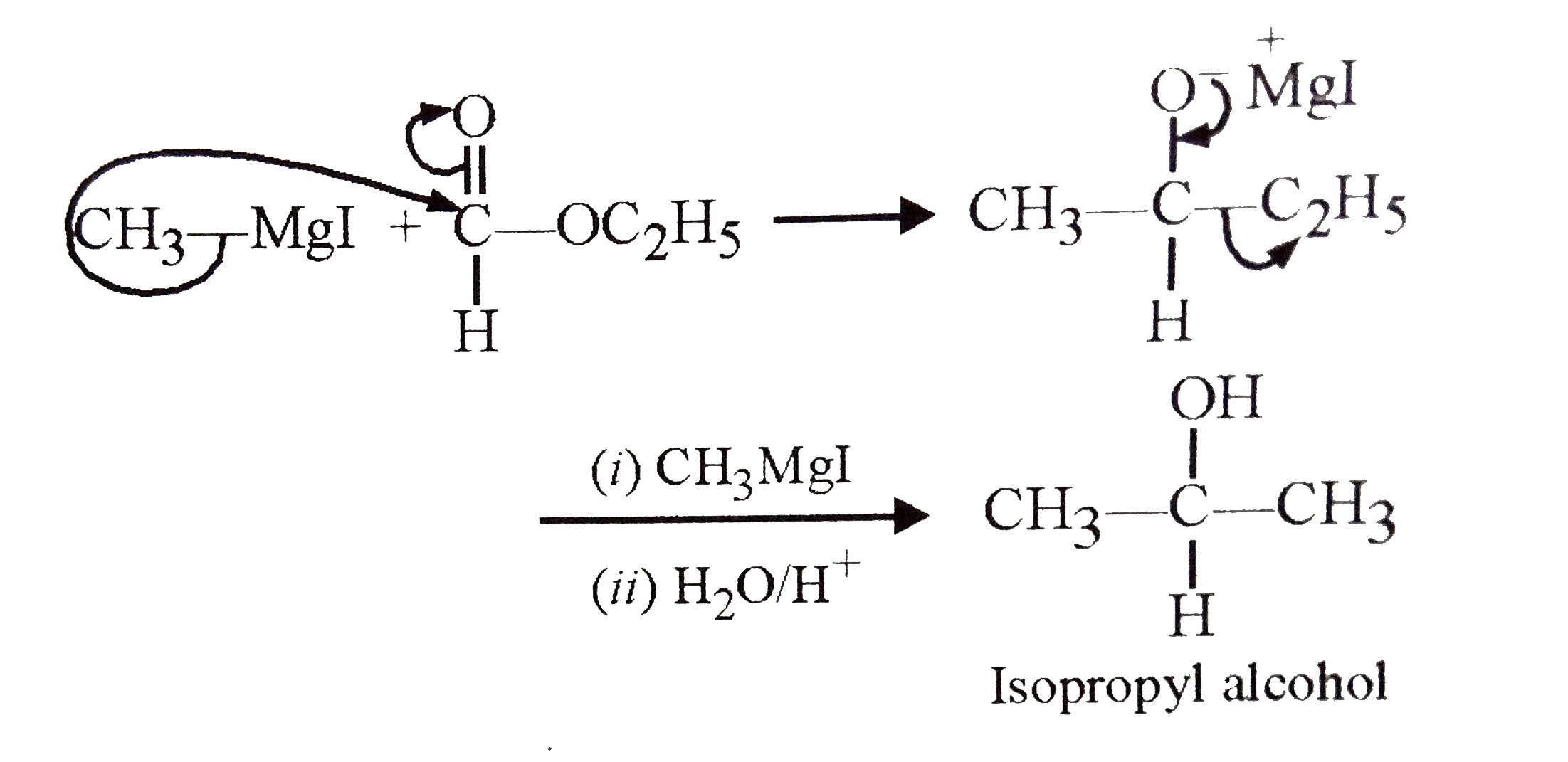
|
|
| 39. |
Which of the following electronic configura tions represent the most metallic element ? |
|
Answer» `[N E]3s^2 3p^5` |
|
| 40. |
Two immiscible liquid present in a bottle can be separated by: |
|
Answer» SEPARATING funnel |
|
| 41. |
The rice is cooked earlier in pressure cooker because |
|
Answer» boiling point increases with INCREASING pressure |
|
| 42. |
Write relation of value of Q_c and K_c and direction of reaction. |
|
Answer» Solution :`Q_c LT K_c`, So REACTION will be forward, Products is increase. `Q_c gt K_c`, So, reaction will be REVERSE (Right to Left ) . Products is DECREASE. `Q_c=K_c`, So, reaction will be in equilibrium. The reaction is not forward or reverse. |
|
| 43. |
What is sp^(2) hybridization ? Give characteristics of it. |
|
Answer» Solution :Definition: ONE ns and THREE np orbitals of an atom having less difference in energy combine and form four equivalent orbitals having similar shape is called `sp^(3)` hybridization and form orbitals are called `sp^(3)` hybridization and form orbitals are called `sp^(3)` hybrid orbitals. Characteristics : there is 25% s-character and 75% p-character in each sp hybrid orbital The four `sp^(3)` hybrid orbitals so formed are directed towards the four corners of tetrahedron. The angle between `sp^(3)` hybrid orbital is `109^(@)`28..  ll four `sp^(3)` hybrid orbitals OVERLAP with s, p or hybrid orbitals of other atom on inter molar AXIS sigma bond arrange at `109.5^(@)` in three dimension & remain tetrahedral in shell. |
|
| 44. |
Which of the following is are resolvable: |
|
Answer»
Resolution is aprocess bywe separate entionmers and we know that entiomers are individually active so CHEAK POS and COS . 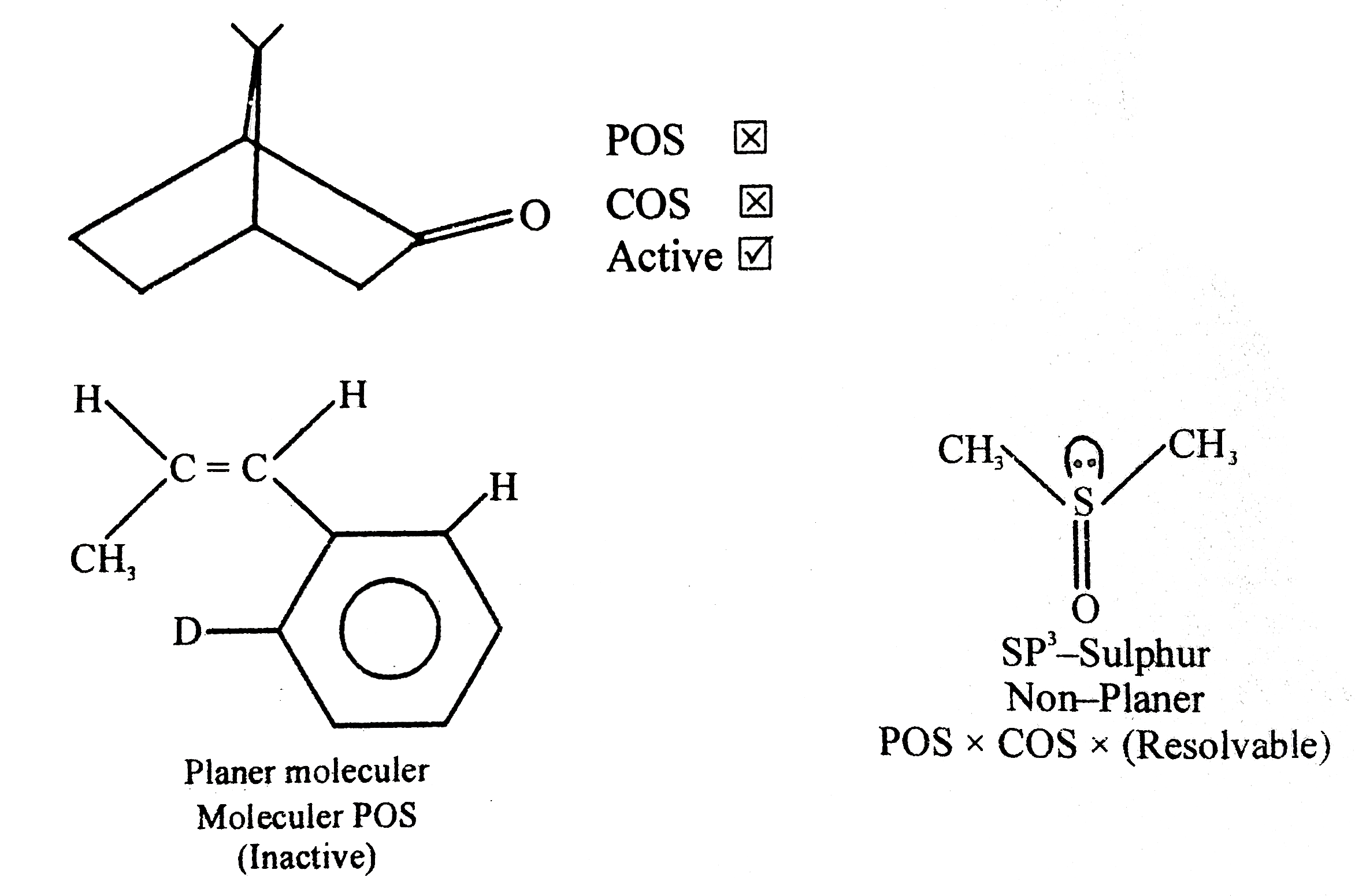
|
|
| 45. |
Why are solids incompressible ? |
|
Answer» |
|
| 46. |
What is the value of Dipole moment (mu) for trans-but-2-ene ? |
|
Answer» 2.5 D |
|
| 47. |
Though the two ends of a glass tube of length 200 cm, hydrogen chlorine gas and ammonia are allowed to enter. At what distance ammonium chloride will first appear ? |
|
Answer» Solution :By Graham's law of diffusion, `(r_(NH_(3)))/(r_(HCl))=sqrt((M_(HCl))/(M_(NH_(3))))=sqrt((36.5)/(17))=sqrt(2.147)=1.465`. Thus, `NH_(3)` travels 1.465 times FASTER than HCl. In other words, `NH_(3)` will travel 1.465 cm in the same time which HCl travels 1 cm. LENGTH of the tube =200 cm. `:. " Distance TRAVELLED in the tube by "=(1.465)/(1.465+1)xx200=118.9 cm` Thus, `NH_(Cl)` will first appear at a distance of 118.9 cm from `NH_(3)` end 81.1 cm from HCl end. |
|
| 48. |
Which one of the following has tetrahedral shape? |
|
Answer» `NH_(4)^(+)` |
|