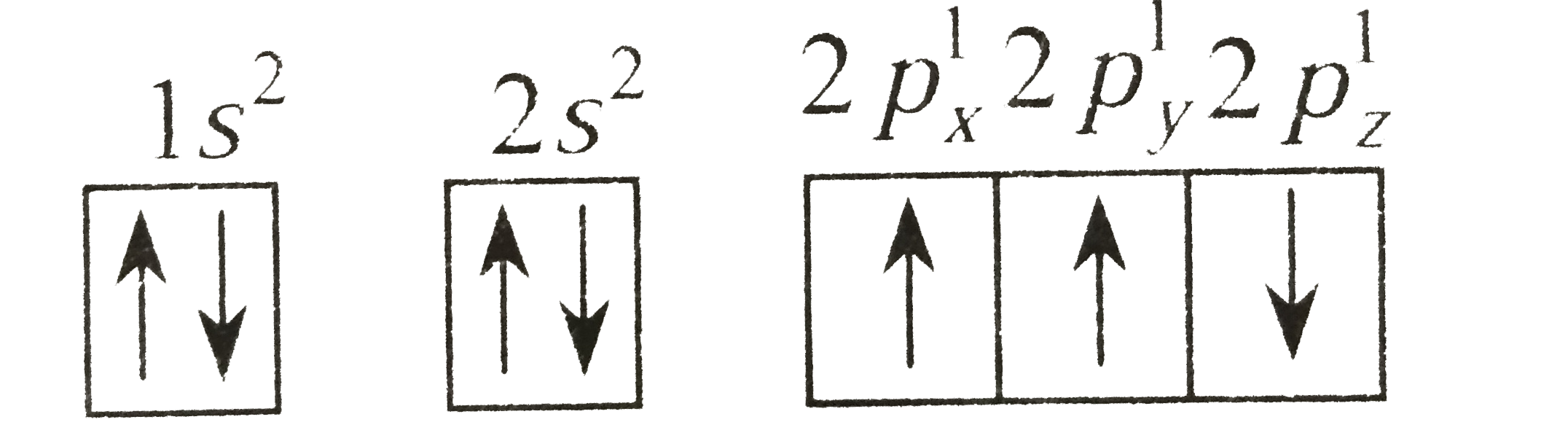Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which is correct increasing order of their tendency of the given elements to from M^(3-) ion? |
|
Answer» `BigtSbgtAsgtPgtN` |
|
| 2. |
Which of the following reactions taking place in the blast furnace during extraction of iron is endothermic . |
|
Answer» `CaCO_(3) to CAO + CO_(2)` |
|
| 3. |
Total number of benzenoid aromatic isomers having formula C_(7)H_(7)Cl : |
|
Answer» 4 |
|
| 4. |
What is inorganic benzene and how is it obtained? |
| Answer» SOLUTION :`B_3 N_3 H_6` | |
| 5. |
What characteristics do you except from an electron -deficient hydride with respect to its structure and chemical reactions ? |
|
Answer» Solution :Electron-deficient hydrides do not have sufficient number of electrons to form normal covalent bonds. Therefore , to mak up this defiency , they generally exist in polymeric forms such as `B_(2)H_(6)` , `B_(4)H_(10)`, `(AlH_(3))_(n)` etc. further to make up this deficiency of electrons , they react with many metals, non-metals and their compounds. Hence, electron-deficient hydrides are very REACTIVE as shown below : `B_(2)H_(6)(g) + 3O_(2)(g) to B_(2)O_(3)(g) + 3H_(2)O(g)` `NaH (s) + B_(2)H_(6)(g) overset("Diethyl ether")to underset("Sod. borohydride")(2Na^(+) [BH_(4)]^(-)(s)` Being electron -deficient compounds, they act as Lewis acids, and thus from complexes with Lewis bases. For example. `underset("Diborane")(B_(2)H_(6))+underset("Trimethylamine")(2NMe_(3)) to underset("COMPLEX")(2H_(3)B larr NMe_(3))` `B_(2)H_(6) + underset("Carbon monoxide")(2:CO) to underset("Complex")(2H_(3)B larr CO)` |
|
| 6. |
What is the bond enthalpy of Xe-F bond? XeF_(4)(g) rarr Xe^(+) (g) + F^(-)(g) + F_(2) (g) + F(g), Delta_(f)H=292kcal/mol. Given that I.E of Xenon =279k Cal/mole, B.E of F_(2)= 38 k Cal/mol. E.A of F=85k Cal/mole. |
|
Answer» 24k Cal/mol `XeF_(4(g)) rarr Xe_((g))^(+) + F_((g))^(-) + F_(2(g)) + F_((g)) , Delta H_(1)` …(1) `Xe_((g)) rarr Xe^(+) + e^(-), Delta H_(2)` …(2) `F_(2(g)) rarr 2F_((g)) , Delta H_(3)` …(3) `F_((g)) + e^(-) rArr F_((g))^(-), Delta H_(4)` ...(4) From the above reactions we will giet the following reaction. `Xe_((g)) + 2F_(2(g)) rarr XeF_(4(g))`... (5) eq.(5) = eq.(2) + eq.(3) + eq(4)- eq.(1) `Delta H_(5) = 279 + 38- 85 - 292` `= -60 = 2` (B. E of `F_(2)`) - 4 (B.E of `Xe- F`) `=60 = 2(38) - 4(Xe - F)` (Xe-F) Bond energy = 34 K.Cal |
|
| 7. |
The standard heat of formation of sodium ions in aqueous solution from the following data : Heat of formation of NaOH(aq) at 25^(@)C = - 470.7 KJ : Heat of formation of OH^(-1) at 25^(@)C = -228.8 KJ is : |
|
Answer» `-251.9KJ` |
|
| 8. |
Under what condition of temperature and pressure the formation of some hydromen from molecul hydrogen will be favoured most |
|
Answer» High temperature and high PRESSURE |
|
| 9. |
Two elements X and Y have the following configuration X=1s^(2)2s^(2)2p^(6)3s^(2)3p^(6)4s^(2) Y=1s^(2)2s^(2)2p^(6)3s^(2)3p^(5) The compound formed by the combination of X and Y will be |
|
Answer» `XY_(2) ` |
|
| 10. |
What is the value of normal boiling point and standard boiling point of water? |
| Answer» Solution :When the external pressure is equal to one atmosphere pressure, the boiling POINT is called NORMAL boiling point. When the external pressure is 1 bar,it is called STANDARD boiling point. | |
| 11. |
What is correct order of ionisation enthalpy of Li, Be, B, C of first period and Na, Mg, Al, Si of second period ? |
|
Answer» `Li lt B lt Be lt C " and " Na lt Al lt Mg lt Si` ` :. B lt Be, Al lt Mg` ` :. B ` and Al having half filled `p^(3)` configuration. |
|
| 12. |
Two isomers of formula C_4H_9 Br are Aand B, A on reaction with alcoholic KOH gives of molecular formula C_4H_8 by E_1 reaction. B on reaction with alcoholic KOH gives D and E as products by Saytzeff's rule. Identify A, B, C, D, E. |
|
Answer» Solution :(i) `C_4H_9Br`: Two isomers may be there : `(A) CH_3 - UNDERSET(Br)underset(|)overset(CH_3) overset(|)C - CH_3` Text butyl bromide and `CH_5 - underset(Br) underset(|)CH - CH-2 -CH_3 ALPHA`-Bromobutane (ii) A on reaction with ALCOHOLIC KOH gives iso BUTYLENE C as the product.  (iii) 2-bromobutane B on reactiön with alcoholic KOH follows Saytzeff.s rule to give a MIXTURE of olefins in different amounts.  
|
|
| 13. |
Which of the following rules could explains the presence of three unpaired electrons in N-atoms ? |
|
Answer» Hund.s RULE |
|
| 14. |
Diamond And Graphite |
| Answer» SOLUTION :`sp^3 , sp^2` | |
| 15. |
Why alloy of Pt-lr was chosen for SI unit (mass & length) ? |
|
Answer» It is cheap metal |
|
| 16. |
Tolerable limits of lead and fluorides in drinking water according to international standard are respectively |
|
Answer» 50ppm and 3ppm |
|
| 17. |
Total number of pi bonds in the contaminent, MIC, which is responsible for Bhopal gas Tragedy is ____. |
|
Answer» |
|
| 18. |
Which relations between equivalent weight (E) and Molecular weight (M) of reactant are correct for the given change? |
|
Answer» `FeCl_(2))` into `Fe(OH)_(2),E=(M)/(2)` EQ wt if `FeCl_(2)=(M)/(2)` `overset(+3)(Fe_(2))(SO_(4))_(3)rarr""^((+2))Fe^(+2)` Eq wt of `Fe_(2)(SO_(4))_(3)=(M)/(2)` |
|
| 19. |
Total number of lone pair of electrons in I_(3)^(-) ion is - |
|
Answer» SOLUTION :`AX_(2)` (i) shape-linear, e.g., `BeCl_(2)` (ii) shape-V-shaped e.g., `C Cl_(2)` (iii) shape-V-shaped e.g., `H_(2)O` `AX_(3)` (i) shape-trigonal planar, e.g., `BF_(3)` (ii) shape-pyramidal, e.g., `NH_(3)` (iii) shape-T-shaped, e.g., `ClF_(3)`. |
|
| 20. |
The system shown in the figure is in equilibrium, where A and B are isomeric liquids and form an ideal solution at TK. Standard vapour pressures of A and B are P_(A)^(0) and P_(B)^(0), respectively, at TK. We collect the vapour of A and B in two containers of volume V, first container is maintained at 2 T K and second container is maintained at 3T//2. At the temperature greater than T K, both A and B exist in only gaseous form. We assume than collected gases behave ideally at 2 T K and there may take place an isomerisation reaction in which A gets converted into B by first-order kinetics reaction given as: Aoverset(k)rarrB, where k is a rate constant. In container (II) at the given temperature 3T//2, A and B are ideal in nature and non reacting in nature. A small pin hole is made into container. We can determine the initial rate of effusion of both gases in vacuum by the expression r=K.(P)/(sqrt(M_(0))) where P= pressure differences between system and surrounding K= positive constant M_(0)= molecular weight of the gas If vapours are collected in a container of volume 8.21 L maintained at 3 T//2K, where T=50 K, then the ratio of initial rate of effusion of gases A and B is given as |
|
Answer» <P>`2 : 1` |
|
| 21. |
The system shown in the figure is in equilibrium, where A and B are isomeric liquids and form an ideal solution at TK. Standard vapour pressures of A and B are P_(A)^(0) and P_(B)^(0), respectively, at TK. We collect the vapour of A and B in two containers of volume V, first container is maintained at 2 T K and second container is maintained at 3T//2. At the temperature greater than T K, both A and B exist in only gaseous form. We assume than collected gases behave ideally at 2 T K and there may take place an isomerisation reaction in which A gets converted into B by first-order kinetics reaction given as: Aoverset(k)rarrB, where k is a rate constant. In container (II) at the given temperature 3T//2, A and B are ideal in nature and non reacting in nature. A small pin hole is made into container. We can determine the initial rate of effusion of both gases in vacuum by the expression r=K.(P)/(sqrt(M_(0))) where P= pressure differences between system and surrounding K= positive constant M_(0)= molecular weight of the gas Vapours of A and B are passed into a container of volume 8.21 L, maintained at 2T K, where T=50 K and after 5 min, moles of B=8//3. The pressure developed into the cotainer after two half lives is |
|
Answer» `3 ATM` `UNDERSET(8//3)(A)overset(K)rarrunderset(4//3)(B)` `t=5 min (8)/(3)-x (4)/(3)+x=(8)/(3)impliesx=(4)/(3)` `=4//3implies t=t_(1//2)=5 min` `implies` At `t=10 min underset(x//3)(A)overset(K)rarrunderset(10//3)(B)` `implies P=(4xx0.0821xx100)/(8.21)=4 atm` |
|
| 22. |
The system shown in the figure is in equilibrium, where A and B are isomeric liquids and form an ideal solution at TK. Standard vapour pressures of A and B are P_(A)^(0) and P_(B)^(0), respectively, at TK. We collect the vapour of A and B in two containers of volume V, first container is maintained at 2 T K and second container is maintained at 3T//2. At the temperature greater than T K, both A and B exist in only gaseous form. We assume than collected gases behave ideally at 2 T K and there may take place an isomerisation reaction in which A gets converted into B by first-order kinetics reaction given as: Aoverset(k)rarrB, where k is a rate constant. In container (II) at the given temperature 3T//2, A and B are ideal in nature and non reacting in nature. A small pin hole is made into container. We can determine the initial rate of effusion of both gases in vacuum by the expression r=K.(P)/(sqrt(M_(0))) where P= pressure differences between system and surrounding K= positive constant M_(0)= molecular weight of the gas If partial vapour pressure of A is twice that of partial vapour pressure of B and total vapour pressure 2 atm at T K, where T=50 K and V=8.21 L, then the number of moles of A and B in vapour phase is: |
|
Answer» `(8)/(3),(4)/(3)` 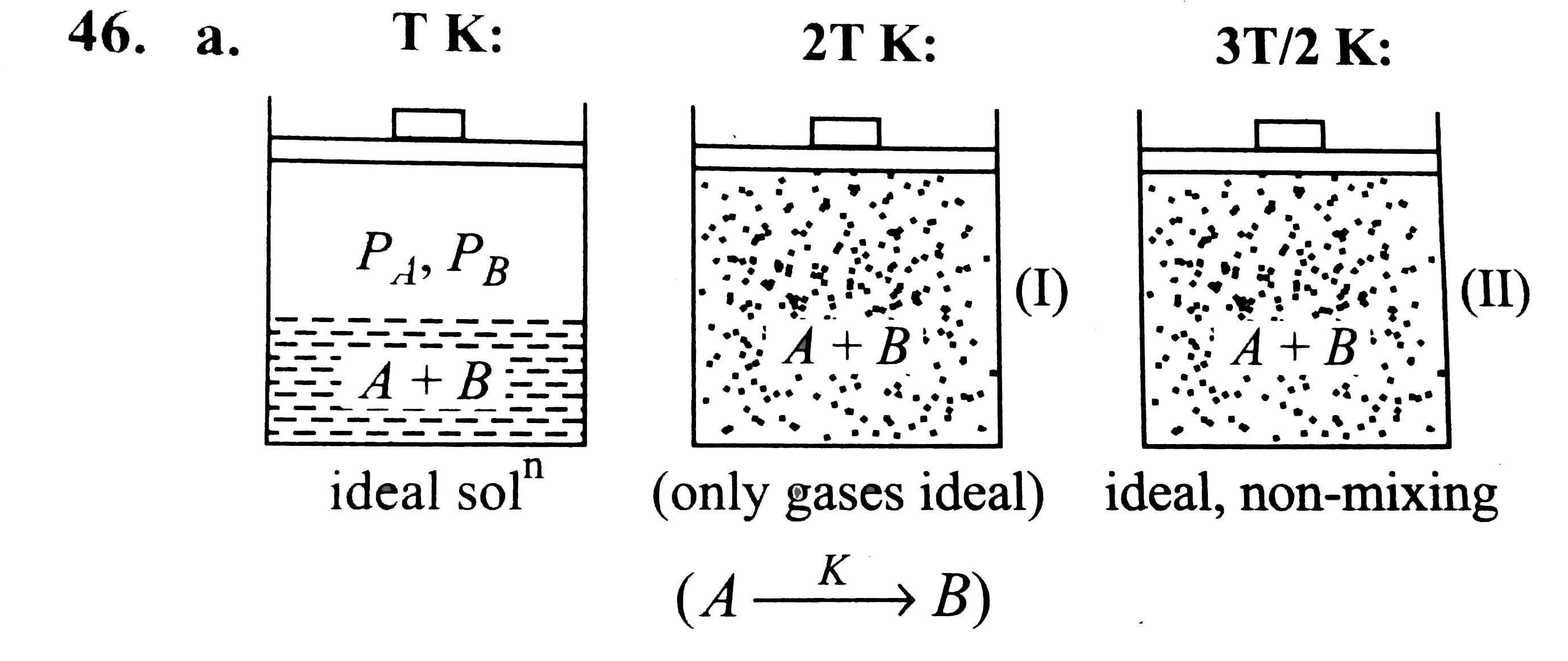 `(n_(A))/(n_(B))=(P_(A))/(P_(B))=2`, Total `V.P.=2 ATM`, `PV=nRTimplies 2xx8.21=(n_(A)+n_(B))xx0.0821xx50` `implies n_(A)+n_(B)=4` |
|
| 23. |
Tin and lead are mainly occur as which ore ? |
| Answer» SOLUTION :Tin OCCURS MAINLY as cassiterite, `SnO_2` and lead asgalena, PBS. | |
| 24. |
What is the use of ZSM-5 ? |
|
Answer» Isomerism in PETROLEUM |
|
| 25. |
Which statement is false for biological importance of K^(+)ions? |
|
Answer» They PRODUCE ATP by oxidation of glucose |
|
| 26. |
Which of the following bond has highest enthalpy |
|
Answer» `N -= N ` |
|
| 27. |
Write the structure of (i) p-dinitrobenzene (ii) o-dichlorobenzene |
Answer» SOLUTION :(i) 
|
|
| 28. |
What is the lowest energy of the spectral line emitted by the hydrogen atom in the Lyman series? (h = Planck constant, c = Velocity of light, R = Rydberg constant) |
|
Answer» `(5hcR)/(36)` |
|
| 29. |
Which of the following statements is wrong |
|
Answer» Benzene does not decolourise alkalin potassium permanganate solution |
|
| 30. |
Which reactions are difficult for alkane ? |
| Answer» Solution :(i) Reaction with acid (ii) Reaction with BASE (iii) With OXIDIZING agent and (IV) With reducing agent etc….are not easily performed by ALKANE so alkane is inert. | |
| 31. |
The elements in which of the following have most nearly the same atomic radius |
| Answer» Answer :D | |
| 32. |
Which one of the following is equal to 1 Joule? |
|
Answer» `NM^(-1)` |
|
| 33. |
Whichone is morecloser to nucleusfrom2pand 3p ? |
| Answer» SOLUTION :2P ismorecloserto nucleusthan 3P | |
| 34. |
Which is a better name for pure H_(2), diprotium or dihydrogen? |
| Answer» SOLUTION :The name diprotium refers to pure `H_(2)` but DIHYDROGEN refers to a isotopic MIXTURE with NATURAL abundanceof H and D (i.e. 99.9844% :0.0156 %). | |
| 35. |
Which of the following reagent is used to distinguish between halogens (CI, Br, I) in an organic compound? |
| Answer» Answer :D | |
| 36. |
What is the difference in NO_(2) and NO_(2)^(-) ? |
|
Answer» Solution :TOTAL valence ELECTRON in `NO_(2)` is 5 + 12 = 17 but total valence electron in `NO_(2)^(-) ` is 5 + 12 + 1 = 18 In `NO_(2)` total 17 electrons which are ODD so octet rule in not follows but in `NO_(2)^(-)` octet rule is follow. 
|
|
| 37. |
Which is the correct increasing acidity order of oxo acids ? |
|
Answer» `HOCLO LT HOCL lt HOClO_3 GT HOClO_2` |
|
| 38. |
Which law of thermodynamics is known as a defination of temperature ? |
| Answer» Solution :ZEROTH LAW of THERMODYNAMIC is known as defination of TEMPERATURE. | |
| 39. |
What are gem dihalides how will youprepare propyne form gem dihalides |
Answer» SOLUTION :compoundscontainingtwohalogenatomson thesamecarbonatomare calledgemdihalideOn heating1,1- dichloropanewithalcoholicKOHit will givepropyne. 
|
|
| 40. |
Whenever a reaction between an oxidsing agent nad a reducing aent is carried out a compound of lower oxidation state I is formed if the reducing agent is in excess and a compound of higher oxidation state is formed if oxidising agent is in excess justify this statement giving three illustrations |
|
Answer» <P> Solution :(i)C is reducing agent while `O_(2)` is n oxidising agent if excess of carbon is burnt in a limitedsupport of `O_(2)` CO formed in which the OXIDATION state of C is +2 if however excess of `O_(2)` is used the initial formed CO gets oxidised to `CO_(2)`in oxidation state of C is +4`2C(s) +O_(2)(g)rarr 2CO(g),C(s)+O_(2)(g)rarrCO_(2)(g)` (ii) `P_(4)` is a reducing agent while `CI_(2)`is usedthe initally formed `PCI_(3)` react further to form `PCI_(5)`in which the oxidation state of p is +5 `P_(4)(s)+6CI_(2)(g)rarr4overset(+3)PCI_(3), P_(4)(s)10 CI_(2)rarr4 overset(+5)PCI_(5)` (iii) NA is a reducing agent while `O_(2)` is an oxidising agent when excessof Na is used sodium oxide formed in the oxidation state of O is -1 which is higher than -2 `4NA(s)+O_(2)(g)rarrNa_(2)O(s), 2NA(s)+2O_(2)(g)rarrNa_(2)O_(2)(s)` |
|
| 41. |
Which one is a wrong statement |
|
Answer» Total ORBITAL ANGULAR momentum of ELECTRON in 's ' orbital is equal to zero 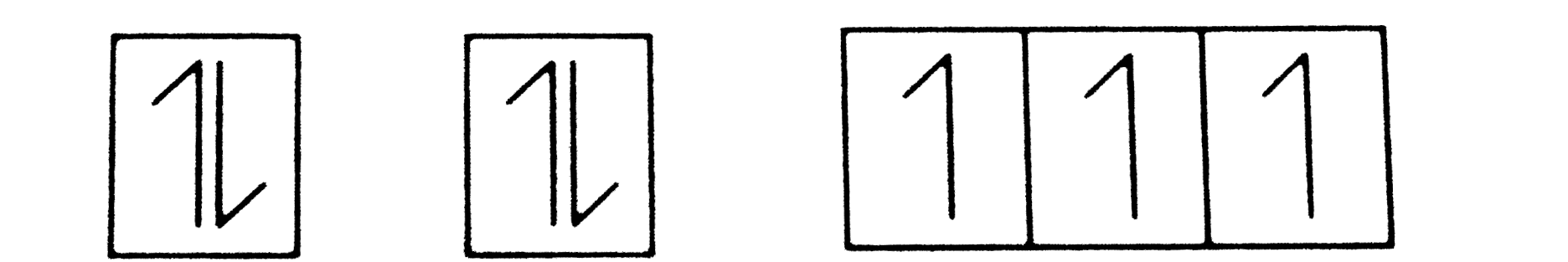
|
|
| 42. |
which cation cannot be identified by flame test ? |
|
Answer» `NA^(+)` |
|
| 43. |
Which has maximum number of molecules? |
|
Answer» 7 G `N_(2)` |
|
| 44. |
Which is the amorphous form of carbon ? |
|
Answer» Coke |
|
| 45. |
Which quantum number reveal information about the shape energy, orientation and size of orbitals ? |
|
Answer» SOLUTION :i.Shape-Azimuthal quantum number II. Energy-Principal quantum number iii. Orientation- Magnetic quantum number IV. Size of orbital-Principal quantum number |
|
| 46. |
Which product is formed when the compoundis reated with concentrated aqueous KOH solution ? |
|
Answer»
|
|
| 47. |
Which of the following free radical is the most stable? |
|
Answer» Tertiary |
|
| 48. |
Write structural formulae for compounds names as- (a) 1-Bromoheptane(b) 5-Bromoheptanoic acid |
|
Answer» Solution :`underset("1-Bromoheptane")(overset(7)(C)H_(3)-overset(6)(C)H_(2)-overset(5)(C)H_(2)-overset(4)(C)H_(2)-overset(3)(C)H_(2)-overset(2)(C)H_(2)-overset(1)(C)H_(2)BR)` (B) `{:(overset(7)(C)H_(3)-overset(6)(C)H_(2)-overset(5)(C)H-overset(4)(C)H_(2)-overset(3)(C)H_(2)-overset(2)(C)H_(2)-overset(1)(C)-OH),("|""||"),(""Br""O),("5-Bromoheptanoic acid"):}` |
|
| 49. |
What does the chemical formula of an ionic compound represent ? |
| Answer» Solution :The chemical formula of an ionic compound REPRESENTS simply a unit of the lattice. It does not represent a molecule of the compound. The unit of lattice is called formula unit. One MOLE of ionic compound represents one mole of formula units and the mass of one mole of an ionic compound in GRAMS is known as the gram formula mass or gram formula WEIGHT. | |