Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which is (are) correct for ideal gas ? |
|
Answer» `((deltaU)/(deltaT))_(P)=0` |
|
| 2. |
The volume of a gas X and chlorine diffusing during the same time are 35 mL and 29 mL respectively. If molecular mass of chlorine is 71, calculate the molecular mass of gas X. |
|
Answer» Solution :SUPPOSE the molecular weight of the gas X is `M_1` . ACCORDING to Eq. 5.25, we have `V_1/V_2 =sqrt(M_2/M_1) ""(t_2and t_2` being the same) In the present CASE, `V_1 = 35 mL, V_2 = 29 mL, M_1 = ? " and " M_2 = 71` `:.35/29 sqrt(71/M_1) " or" M_1 = 48.74` `:.`The Molecular weight of the gas `X =48.74`. |
|
| 3. |
Wavelength of an electron is 5Å. Velocity of the electron is |
|
Answer» `1.45 XX 10^(8) cm//s` |
|
| 4. |
Which period mostly include man made radioactive elements? |
|
Answer» 4 period |
|
| 5. |
What is Wurtz reaction? Give example. |
|
Answer» Solution :When ALKYL halides are heated with sodium metal in ether medium hgiher alkenes are formed. This reaction is KNOWN as Wurtz reaction and employed for the synthesis ofhigher alkanes containig even number of carbon atoms. `underset("bromethane")(CH_(3)-Br+2Na)+Br-HCH_(3)underset("heat")OVERSET("dryether")(to)underset("ethane")(CH_(3))-CH_(3)+2NaBr` |
|
| 6. |
What is the molar solubility of AgCI_((g)) in 0.100M NH_(3(aq.)) ? Given K_(SP) of AgCI = 1.8xx10^(-10), K_(f) of [Ag(NH_(3))_(2)]^(+)= 1.6xx10^(7) . |
|
Answer» |
|
| 7. |
What is the role of chloromethane and dibromoethane when added to gasoline along with tetraethyl lead ? |
| Answer» SOLUTION :They CONVERT PBO PRODUCED into VOLATILE `PbCl_2` and `PbBr_2` | |
| 8. |
Write the tautomers of the following compounds. |
|
Answer» |
|
| 9. |
Write IUPAC names of the products obtained by addition reactions of HBr to hex-1-ene (i)in presence of peroxide and (ii)in the absence of peroxide. |
|
Answer» Solution :(i)In PRESENCE of peroxides, anti-Markovnikov's addition occurs and 1-bromohexane is FORMED . <BR> `underset"Hex-1-ene"(CH_2=CH-CH_2-CH_2-CH_2-CH_3+HBr ) underset"(Anti-Mark. addn.)"OVERSET"PEROXIDE"to underset"1-Bromohexane"(undersetunderset(Br)|CH_2-CH_2-CH_2-CH_2-CH_2-CH_3)` (ii)In absence of peroxides, Markovnikov's addition occurs and 2-bromohexane is formed . `underset"Hex-1-ene"(CH_2=CH-CH_2-CH_2-CH_2-CH_3+HBr ) underset"(Mark. addn.)"overset"No peroxide"to underset"2-Bromohexane"(CH_3-undersetunderset(Br)|CH-CH_2-CH_2-CH_2-CH_3)` |
|
| 10. |
Which one of the following is the wrong statementabout the liquid? |
|
Answer» It has INTERMOLECULAR force of attraction |
|
| 11. |
Which bond angel is same in H_(2)^(+) He_(2)^(-) and He_(2)^(2-)? |
Answer» SOLUTION :BOND ORDER is same in `H_(2)^(+) and He_(2)^(-)`. 
|
|
| 12. |
Which is considered as to be earth protective umbrella? Why? |
|
Answer» Solution :(i) At high altitude in the atmosphare consists of a layerof ozone `(O_(2))` Which acts as unbrella for harmful UV radiations. Ozone is considered to be EARTH protective umbrella. (ii) It protects us from harmful effects of UV-radiations of the SUN such as skin cancer. (III) Ozone layer prevent the UV radiations to reach the earth surface So it acts as an umbrella for the earth. |
|
| 13. |
What is not true about the following reaction RCOCl + H_(2) overset(Pd//BaSO_(4) , S)rarr RCHO + HCl |
|
Answer» It is ROSENMUND's reaction |
|
| 14. |
Write short notes on the the following : (i) Raschig process (ii) Dows Process (ii) Darzens process |
Answer» Solution :(i) Raschig process: Chlorobenzene is commercially prepared by passing a mixture of benzene vapour in air and HCL over heated cupric CHLORIDE.  (ii). Dow.s process: Chlorobenzene is boiled with Sodium hydroxide to get Phenol. This reaction is called Dow.s process. `UNDERSET("Chloro benzene")(C_6H_5Cl) + NaOH underset("350 atm")overset(350^@C)tounderset("Phenol")(C_6H_5OH) + NaCl` (iii) Darzens process: Ethanol reacts with `SOCl_2` in the presence of PYRIDINE to form chloroethane. This reaction is called Darzens process. `underset("Ethanol")(CH_3-CH_2OH) + SOCl_2 overset("Pyridine")to underset("Chloroethene")(CH_3CH_2Cl) + SO_2 UARR + HCluarr` |
|
| 15. |
Which of the following are non functional in human beings? |
|
Answer» METHYL propyl ETHER and diethyl ether |
|
| 16. |
What are the uses of sodium and its compounds? |
|
Answer» Solution :Sodium is used to make `Na//Pb` alloy NEEDED to make `Pb(Et)_(4)` and `Pb(Me)_(4)` These organolead compounds were used as anti-knock additives to PETROL in early days (ii) Liquid sodium metal is used as a coolant in fast breeder nuclear reactors |
|
| 17. |
What is concentration of solution ? |
|
Answer»
|
|
| 18. |
The relative abundance of three isotopes of carbon C^(12), C^(13) and C^14 are 98.892%, 1.108% and 2 xx 10^(-10)% respectively. If the relative atomic masses of these isotopes are 12.00, 13.00335 and 14.00317 amu respectively, find the average atomic mass of carbon. |
|
Answer» SOLUTION :Average atomic mass of carbon can be obtained by averaging the relative atomic MASSES of its isotopes in the ratio of their abundance. Thus, Average atomic mass `(12.000 XX 98.892 + 13.00335 xx 1.108+ 14.00317 xx 2 xx 10^(-10))/(98.892 + 1.108 +2 xx 10^(-10))` `=12.011` amu |
|
| 19. |
Which of the following elements has [Ar]3d^10 4s^2 4p^1electronic configuration ? |
|
Answer» B |
|
| 20. |
Write the expression of K_(c) for the reaction N_(2)(g)+3H_(2)(g)hArr2NH_(3)(g). Give units of K. |
| Answer» Solution :`K=([NH_(3)]^(2))/([N_(2)][H_(2)]^(3))=((MOLL^(-1))^(2))/((molL^(-1))(molL^(-1))^(3))=L^(2)mol^(-2)` | |
| 21. |
The reagent(s) required for the following conversion are : |
|
Answer» (i) `NaBH_(4)` (ii) Raney `Ni//H_(2)` (ii) `H_(3)O^(+)` |
|
| 22. |
X- ray diffraction studies show that copper crystallizes in an fcc unit cell with cell edge of3.608 xx 10^(-8)cm. In a separate experiment, copper is deteremined to have a density of 8.92 g/cm^(3)calculate the atomic mass of copper. |
|
Answer» Solution :` P = (Z xx M)/(a^(3) xx N_(0))orM = ( p xx a^(3) xx N_(0))/Z` for fcc LATTICE , Z= 4. Hence , ` M = (( 8.92 " g cm" ^(-3))( 3.608 xx 10^(-8) cm) ^(3)( 6.022 xx 10^(23)" atoms mol"^(-1))) = 63.1" g mol ^(-1)` Atomic MASS of copper = `63.1 "g mol"^(-1)` |
|
| 23. |
What is the pyrolysis of alkane ? Give reaction. |
|
Answer» Solution :Defination higher alkanes on heating to higher temperature decompose into lower alkanes, alkenes etc. Such a decomposition reaction into smaller fragements by the application of heat I called pyrolysis or craking. Higher alkane `underset("Pyrolysis")overset("Higher temperature")rarr (("Lower alkane"),("alkane mixture"))` Example -1 : Pyrolisis of hexane  Example -2 : Pyrolysis of alkynes is believed to be a free radical reaction. Preparation of oil gas or petrol gas from kerosene oil or petrol involves the principle of pyrolysis. For example, dodecane, a constituent of kerosene oil on heating to 973K in the presence of platinum, palladium or nickel gives a mixture of heptane and pentene. `underset("Kerosene")underset("Dodecabe")(C_(12)H_(26))underset(973 K)overset("Pd, PT, Ni")rarr underset("(Alkane)")underset("Heptane")(C_(7)H_(10)) + underset("(Alkene)")underset("Pentene")(C_(5)H_(10))+ underset("Products")("Other")` Thermal combusiton of alkane fission is free radical type reaction. THis type of mixer of HYDROCARBON is mixture of less carbon (Alkane + Alkene) |
|
| 24. |
Which of the following has maximum -I effect |
| Answer» SOLUTION :N/A | |
| 25. |
'X' +AgNO_(3) rarr 'Y' (yellowish white ppt) 'Y' is soluble is nitricacid and ammonia. The ppt. becomes yellowish brown upon addition of excess reagent owing to the formation of 'Z'.If aqueous suspension of 'Y' is boiled then it gives the same product 'Z'. Here 'X' and 'Y' are: |
|
Answer» `CO_(3)^(2-)` and `Ag_(2)O` |
|
| 26. |
'X' +AgNO_(3) rarr 'Y' (yellowish white ppt) 'Y' is soluble is nitricacid and ammonia. The ppt. becomes yellowish brown upon addition of excess reagent owing to the formation of 'Z'.If aqueous suspension of 'Y' is boiled then it gives the same product 'Z'.Here 'Z' is: |
|
Answer» `Ag_(2)CO_(3)` |
|
| 28. |
XeF_2 is isostructural with ........... |
|
Answer» `SbCl_2` |
|
| 29. |
Write short notes on Ionic Hydrides and give suitable examples. |
|
Answer» Solution :IONIC (Saline) Hydrides: These are hydrides composed of an electropositive metal, generally, an alkali or alkaline-earth metal, except beryllium and magnesium, formed by transferring of electrons from metal to hydrogen atoms. They can be PREPARED by the REACTION of ELEMENTS at about `400^(@)C`. Theseare salt-like, high-melting, WHITE, crystalline solids having hydride ions `(H^-)` and metal cations `(Mn^+)` `2Li(s)+H_2(g)to2LiH(s)` `2Ca(s)+H_2(g)to2CaH_2(s)` |
|
| 30. |
Which of the following can stabilize gold sol from coagulation by NaCl solution:- |
|
Answer» `FE(OH)_(3)` |
|
| 31. |
Which of the following is correct regarding stability of conformations of n-butane about C_(2)-C bond Anti (I)gauche (II)partially eclipsed (III)fülly eclipsed (IV) |
|
Answer» I gt II gt III gt IV |
|
| 32. |
What is the volime of a piece of silver which weighs 21.5639 g. The density of silver is 10.4 g//cm^(3) ? |
|
Answer» |
|
| 33. |
What are ultramines ? |
|
Answer» SOLUTION :Ultramarines are aluminosilicates but do not contain water of hydration. Some extra anioins, like `CI^(-), S_(2)^(2-) and SO_(4)^(-2)`, MAY also be present in the cavities. Example of ultramarines are sodalite, ` Nag(AIO_(2))_(6)(SiO_(2))_(6)CI_(2)` , ultramarine, ` Na_(8) (AIO)_(2))_(6)(SiO_(2))_(6)S_(2)` and nosean, `Na_(8)(AIO_(2))_(6)(SiO_(2))_(6)SO_(4)`, Many ultramarines are coloured and HENCE as pigments. |
|
| 34. |
The yield of product in the reaction 2A_((g)) + B_((g)) hArr 2C_((g)) + Q K J would be lower at : |
|
Answer» LOW TEMPERATURE and low PRESSURE |
|
| 35. |
What is electrovalent (Ionic) bond ? Explain with any two example. |
|
Answer» Solution :Electrovalent Oonic) bond : The bond formed, as a result of the electrostatic attraction between the positive and negative ions was termed as the electrovalent bond. Example-I : IONIC bond formation in sodium chloride (NaCl) Na (Alkali metal) is highly electropositive metal and form Na+ ion by loss of one electron and attain stable noble gas electronic configuration `underset([Ne]3s^(1))(Na) rarr underset([Ne])(Na^(+)) + e^(-)` . Chlorine is the highly electronegative halogens. It convert into `Cl^(-)`negative ion by gain one electron of sodium ion. This `Cl^(-)` ion attain stable electron configuration of noble gas AR `(3s^(2)3p^(6))`. `underset([Ne] 3s^(2) 3p^(5)) + e^(-) rarr "" underset([Ar] " or " [Ne] 3s^(2) 3p^(6))(Cl^(-))` Thus, `Na^(+)` positive ion and `Cl^(-)` negative ion both have stable outer shell configuration of eight (octet) electrons. And between them ionic (electrovalent) bond is formed. `Na^(+) + Cl^(-) rarr " NaCl or " [Na^(+) Cl^(-)]` Example-2 : Ionic bond (electrovalent) formation in `CaF_(2)`. The calcium loses TWO electrons to attain the stable noble gas configuration of argon and converted into `Ca^(2+)`calcium. `underset([Ar]4s^(2))(Ca) rarr underset([Ar])(Ca^(2+)) + 2e^(-)` The halogen atom fluorine can gain highly electronegative one electron to require the stable outer shell configuration and convert into fluoride ion. `underset([He]2s^(2) 2p^(5))(F) +e^(-) rarr underset([He]2s^(2) 2p^(6)"or"[Ne])(F^(-))` The positive ion `Ca^(2+)` and negative ion `F^(-)` ion are held TOGETHER by strong electrostatic force of attraction and formed electrovalent bond containing `CaF_(2)`. `Ca^(2+) + 2F^(-1) rarr CaF_(2) ` |
|
| 36. |
which of the following is used to make visible upper tract in an X-ray photograph? |
|
Answer» Milkshake |
|
| 37. |
Which of the following flux is used to remove acidic impurities in metallurgical processes ? |
|
Answer» Silica |
|
| 38. |
When BaCl_(2) is added to an aqueous salt solution , a white ppt. is obtained . The anion amoung CO_(3)^(2-) , SO_(3)^(2-) and SO_(4)^(2-) that was present in the solution can be |
|
Answer» `CO_(3)^(2-)` but not any of the other two `BaCl_(2) + CO_(3)^(2-) or SO_(3)^(2-) or SO_(4)^(2-) to ` `(BaCO_(3) or BaSO_(4) or BaSO_(4))/("Insoluble in" H_(2)O) + 2 Cl^(-)` Thus , any one of them may be present and hence option (d) is CORRECT . |
|
| 39. |
What is the chemical composition of Sorel's cement? |
| Answer» SOLUTION :`MgCl_2 . 5MgO . xH_2O` | |
| 40. |
Xg of Ag was dissolved in HNO_3 and the solution was treated with excess of NaCl when 2.87g of AgCl was precipitated. The value of X is |
|
Answer» 1.08g |
|
| 41. |
The unit cube Length for LICL (NACL structure) is 5.14 Å. Assuming anion-anion contact, calculate the ionic radius for chloride ion. |
|
Answer» `THEREFORE BC=sqrt(AB^2+AC^2)=sqrt((2.57)^2+(2.57)^2)=3.63` RADIUS of `Cl^-` ion=`1/2` x 3.63 Å =1.81 Å 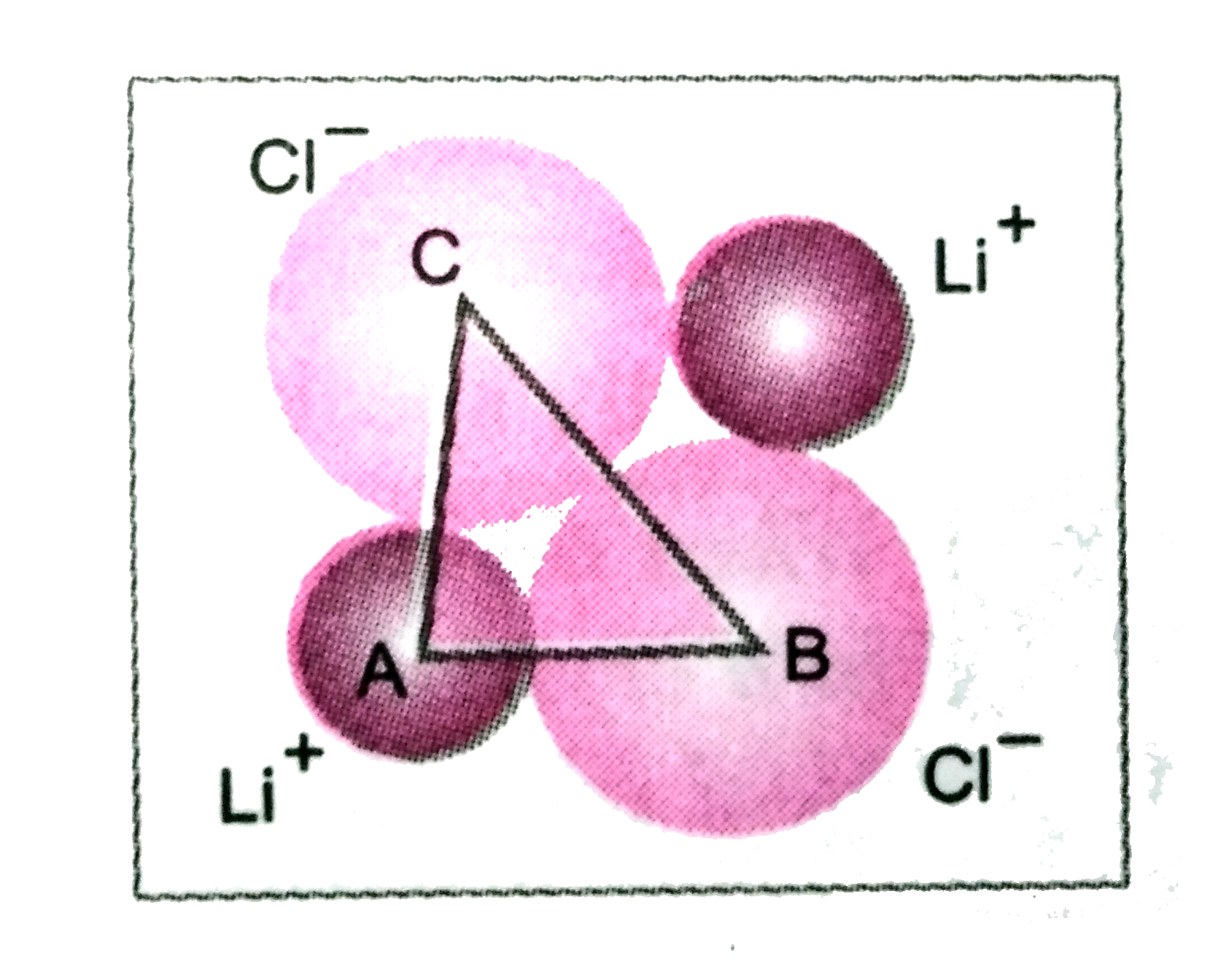
|
|
| 42. |
The solubility of BaSO_4 is 5 mol L^(-1) then what is the solubility product ? |
|
Answer» Solution :`{:(BaSO_(4(s)) hArr, Ba_((aq))^(2+) +, SO_(4)^(2-)),(,SM, SM):}` `K_(sp)=[Ba^(2+)][SO_4^(2-)]=(S)(S)=S^2` `=(5)^2=25 ("MOL L"^(-1))^2` |
|
| 43. |
Which pairs of ions are responsible for hardness of water ? |
| Answer» SOLUTION :`Ca^(2+)` and `MG^(2+)` LIKE pairs of IONS are responsible for hardness of water. | |
| 44. |
Which one of the following elements has the highest ionization energy. |
|
Answer» `[He] 3s^(2) 3p^(1)` |
|
| 45. |
What is intermolecular force and van der Waals forces ? Which forces are not included in it ? Explain its types and uses ? |
|
Answer» SOLUTION :Inter Molecular Forces : Inter molecular forces are the forces of attraction and replusion between interacting particles. This term does not inclde the electrostatic forces that exist between the two oppositely charged ions the forces that hold atoms of a MOLECULE together i.e., covalent bonds. van der Waals Forces : Attractive intermolecular forces are KNOWN as van der Waals forces. In honour of Dutch SCIENTIST Johannes van der Waals (187 - 1923), who explained the deviation of real gases from the ideal behaviour through these forces. Under forces are not considered as van der Waals forces. (i) Attraction force between an ion and dipole (ii) Forces present between Atoms of molecule of covalent bond. (iii) Attractive forces between an ion and a dipole are known as ion - dipole forces. Van der Waals forces vary considerably in magnitude. (i) dispersion forces of London forces. (ii) dipole - dipole forces. (iii) dipole - induced dipole forces. Deviation between ideal gas and real gas is EXPLAIN by the help of van der Waals forces. |
|
| 46. |
What are Fumes ?Given one example. |
|
Answer» Solution :Fumes are one of the non-viable particulate pollutants air. They are obtain by CONDENSATION of vapour released during sublimation, distillation , BOILING and CALCINATION. For Example, ORGANIC solvents, metals and metalic OXIDES form fumes particles. |
|
| 47. |
Which is, a meso compound? |
|
Answer» (2R, 3R) - 2,3-Dibromobutane |
|
| 48. |
What are electron deficient compounds? Are BCl_3 and SiCl_4 electron deficient? |
|
Answer» |
|
| 49. |
Which mixture is separated by physical method ? |
|
Answer» |
|
| 50. |
What are competive electron transfer reaction ? Give example. |
|
Answer» Solution :These are the reactions in which redox reactions take place in DIFFERENT vessels and it is an indirect redox reaction. There is a competition for the RELEASE of electrons among different metals. Example: Zn releases electrons to Cu and Cu releases electrons to silver and so on `Zn_((s))+Cu^(2+)rarrZn_(aq)^(2+)+Cu_((s))` (Here Zn - oxidised : `Cu^(2+)` - REDUCED) `Cu_((s))+2Ag^(+) rarr Cu_(aq)^(2+)+2AG_((s))` (Here Cu - oxidised, `Ag^(+)` - reduced ) |
|