Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Arrange the steps in sequence for the manufacture of cement. (a)Mixture is pulverized using a pulverizer. (b)Limestone and clay are mixed in a definite proportion. (c)Mixture is heated to1500^(@) C " to " 1600^(@)C to form clinkers. (d) Clinkers are pulverizedto form fine powder and a small quantity of gypsum is added. |
|
Answer» a c B d (b) Mixture is pulverized USING a pulverizer. (c) Mixture is heated to ` 1500^(@) c " to " 1600^(@) C` to from CLINKERS. (d) Clinkers are pulverized to form fine powder and a SMALL quantity of gypsum is added. Choice |
|
| 2. |
Arrange the steps in sequence for the manufacture of borosilicate glass. (a) Addition of ingredients like red lead, aluminium oxide, manganese oxide, chromium oxide etc. (b)Addition of broken pieces of glass (c)Mixing up of sand, limestone, sodium carbonate, aluminium oxide and borax in required proportion (d)Melting mixture in a glass tank furnace or pot furnace (e ) Annealing process (f) Blowing of glass |
|
Answer» cbadfe (ii) addition of BROKEN pieces of glass (iii) Addition of ingredients like red lead, aluminium oxide, manganes oxide, chrominum oxide ETC. (iv)Melting mixture in a glass tank furnace orpot furnace. (v) Blowingof glass (VI) Annealing process |
|
| 3. |
Arrange the steps in sequence for the manufacture of cement. (a)Mixture is pulverized usinga pulverizer. (b)Limestone and clay are mixed in a definite proportion. (c)Mixture is heated to1500^(@)C" to "1600^(@)C to form clinkers. (d) Clinkers are pulverized to from fine powder and a small quantity of gypsum is added. |
|
Answer» acbd (II) Mixture is pulverized using a pulverizer . (iii) Mixture is HEATED to ` 1500^(@)C"to" 1600^(@)C` to from clinkers. (iv) Clinkers are pulverized to form fine powder and a small quantity of gypsum is added. |
|
| 4. |
Arrange the steps in sequence for the manufacture of borosilicate glass. (a)Addition of ingredients like red lead, aluminium oxide, manganese oxide, chromium oxide etc (b)Addition of broken pieces of glass (c)Mixing up of sand, limestone, sodium carbonate, aluminium oxide and borax in required proportion (d) Melting mixture in a glass tankfurnace or pot furnace (e ) Annealing process (f) Blowing of glass |
|
Answer» c b a d f e (b) Addition of broken pieces of glass (c) Addition of INGREDIENTS like red lead, aluminium oxide manganese oxide, chromium oxide etc (d) Melting mixture in a glass TANK furnace or pot furnace. (f) BLOWING of glass (e) Annealing process |
|
| 5. |
Arrange the steps in a sequence for the removal of microorganisms during the purification of drinking water. (a) Lime feeder(b) Mechanical mixing(c ) Alum feeder(d) Sedimentation tank(e) Passing through gravel and sand (f) Chlorine feeder |
|
Answer» C a b d e f (iii) Mechanical MIXING(iv) Sedimentation TANK (v) Passing through gravel and SAND(vi)Chlorine feeder |
|
| 6. |
Arrange the steps in a sequence for the conversion of atmospheric water vapour into underground water. (a) Infiltration in recharge area (b) Water table (c ) Infiltration in zone of aeration (d) Precipitation(e) Aquifer |
|
Answer» d a c b e (ii) Infiltration in ZONE of aeration (iii) Infiltration in RECHARGE AREA (iv) Water table (v) Aquifer |
|
| 7. |
Arrange the steps in a sequence for the formation of rainfall.(a) Formation of micelles(b) Condensation(c ) Evaporation(d) Formation of clouds (e) Precipitation |
|
Answer» c E d b a (ii) Condensation (III) Formation of micelles (IV) Formation of CLOUDS (v) Precipitation |
|
| 8. |
Arrange the staps in sequence for the manufacture fo glass. |
|
Answer» Addition of CULLET (ii) Additon of cullet (iii) Addition of colour pigments (iv) Annealing |
|
| 9. |
Arrange the process in sequence for the purification of drinking water (a) Lime feeder, alum feeder(b) Filtration(c ) Mechanical mixing(d) Chlorination |
|
Answer» abcd (iii) FILTRATION(IV) CHLORINATION |
|
| 10. |
Arrange the relevant steps in correct sequence for themanufacture of sodium chloride from sea water. (a) Making of hot saturated solution (b) Evaporation (c) Recrystallization (d) Collection of sea water (e ) Addition of small quantity of sodium iodide or potassium iodide |
|
Answer» d E b a (b) Evaporation (a) MAKING of hot saturated solution (c) RECRYSTALLIZATION |
|
| 11. |
Arrange the points in sequence to draw the geometrical representation of an atom which is resprecented as _(Z)X^(A).(a)Calculation of the numberof protons and neutrons.(b)Identification of the atomic number and mass number of the given element.( c) Placement of the electrons in shells aroundthe nucleus according to the electronic configuration.(d)Wirting the electronc configuration of the atom.(e) Protons and neutrons are shown in nuleus. |
|
Answer» baedc (ii) Calculation of the number of protons and NEUTRONS. Protons and neutrons are SHOW in nucleus. (III) Protons and neutrons are show in nucleus. (iv) Placement of the ELECTRONS in SHELLS around the nucleus according to the electronic configuration. |
|
| 12. |
Arrange the points in sequence to draw the geometrical representation representation of_(20)Ca^(40).(a) Protons and neutrons are shown in the nucleus.(b) Writing the electronic configuration of the atom.(c ) Arrange the electrons in shells around the nucleus following the electronic configuration.(d) Calculation of the number of protons and neutrons.(e ) Identification of the atomic number and mass number of the atom. |
|
Answer» edcba (ii) Calculation of the number of protons and NEUTRONS. (iii)Protons and neutrons are shown in the nucleus. (iv) Writing the ELECTRONIC configuration of the atom. (v)ARRANGE the electronis in SHELLS around the nucleus following the electronic configuration. |
|
| 13. |
Arrange the pairs in sequence in decreasing order of basicity of an acid and acidity of a base followed by the normal salts formed by the given acid base pairs in the same order respectively. (a) Acetic acid, ammonium hydroxide (b) Oxalic acid, barium hydroxide (c) Phosphoric acid, aluminium hydroxide (d) Barium oxalate (e) Ammonium acetate (f) Aluminium phosphate |
|
Answer» adcfe (ii) OXALIC acid, barium hydroxide (iii) ACETIC acid, ammonium hydrozide (iv) Ammonium ACETATE (v) Barium oxalate (vi) Aluminium phosphate |
|
| 14. |
Arrange the pairs in sequence in decreasing order of base and increasing order of basicity of an acid and the salts formed between an acid and base with equal basicityand acidity in their increasing order respectively. (a) Sodium chloride (b) Barium hydroxide, sulphuric acid (c) Ferric phosphate (d) Ferric hydroxide, hydrochloric acid (e) Barium sulphate (f) Sodium hydroxide, phosphoric acid |
|
Answer» dbfaec (iii) SODIUM hydroxide, PHOSPHORIC acid. (iv) Sodium chloride (v) Barium SULPHATE (vi) Ferric phosphate |
|
| 15. |
Arrange the layers of the atmosphere as per the following terms related in a sequence order respectively. Inversion layer, convertion currents, absorbs harmful radiations and burning up of meteorites (a) Troposphere (b) Stratophere ( c) Exophere (d) Mesosphere (e ) Ionophere |
|
Answer» decab (II) Troposphere (III) IONOSPHERE (iv) Exosphere (V) Mesophere |
|
| 16. |
Arrange the given compounds inthe increasing order of carbon atoms present in the compounds (a) Methane (b) Suger (c) Glucose (d) Starch |
|
Answer» `bltclt DLT a` (c) Glucose (b) Sugar (d) STARCH |
|
| 17. |
Arrange the general steps for the purification of water in a sequence. (a) Filtration(b) Chlorination (c ) Sedimentation (d) Addition of chemicals |
|
Answer» a C b d (ii) Addition of CHEMICALS (iii) FILTRATION (IV) Chlorination |
|
| 18. |
Arrange the following terms in sequence from source to supply. (a) Chlorination (b) Sedimentation (c ) Filtration(d) Storage water tank (e) Lakes |
|
Answer» ecadb (ii) Sedimentation (iii) FILTRATION (iv) CHLORINATION (v) Storage water TANK |
|
| 19. |
Arrange the following substance in the ascending order of the number of constituent element(s) present in them. (a) Sodium bicarbonate, (b) Calcium carbonate ( c) Water, (d) Copper |
|
Answer» d,C,b,a (i) Copper (ii) WATER (iii) Calcium CARBONATE (iv) Sodium bicarbonate |
|
| 20. |
Arrange the following sequence in orderto derive the formula of zine oxide.(a)The valencies of ions formed by zine and oxygen are written below without positive and negative sign.(b)The positive ion that isZn^(+2)followed by the negative ion , that is oxide(O^(-2)ion are written with ther charges.(c )The valencies of zinc and oxide ion have common factors and hence they aredivided by their highest common factor.(d)The magnitudeof valencies of the ions should be interchanged and written as subscripts on right sideof the respective ions. |
|
Answer» badc (ii)The VALENCIES of ions formed by zine and oxygen and are written below without positive and negative sign.(iii) The valencies of ZINC and oixde have common factors and hence they are divided by the HIGHEST common FACTOR. (iv)The magnitude of the valenciesof the ions should be interchanged and written as SUBSCRIPTS on right side of the respective ions. |
|
| 21. |
Arrange the following point in sequence for derivation of formulae of compounds.(a) The valencies of the ions or radicals are written below the radicals without positive and negative sing.(b)If the valencies have common factors , they are divided by the highest common factor.(c ) The magnitude of valencies should be interchanged and writtenassubscripts on the right side of the respective ions.(d) The positive radical followed by negative radical are written with their chatges.(e)If the radicals contain more than one atom , they are enclosed in brackets and the appropriate valency (more than one ) written assubscripts outside the bracket. |
|
Answer» bcdea (ii)The valencies of the ions or radicals are written below the radicals without positive and negative sign. (iii) The magnitude of valencies should be interchanged and written as subscripts on the right SIDE of the respective ions. (iv)If the valencies have common factors , they are dividedby the HIGHEST common factor. (v)If the radicals CONTAIN more than one atom , they are enclosed in brackets and the appropriate VALENCY (more than one) written as subscript outside the BRACKET. |
|
| 22. |
Arrange the following layers of the atmophere in the decreasing order of their altitudes. (a) Stratosphere (b) Troposphere ( c)Mesophere (d) Exosphere ( e) Ionophere |
|
Answer» cdeab (ii) Ionosphere (III) Mesophere (iv) Stratophere (v) Troposphere |
|
| 23. |
Arrange the following in a proper sequence for the separation of constituents of gun powder (a) Carbon disulphide is added to the mixture (b) Carbon powder is separated (c ) Residue is filtered (d) KNO_(3) is separated from its aqueous solution by heating it (e) Mixture is filtered and hot water is added to the residue |
|
Answer» aedbc (ii) Mixture is filtered and hot water is added to the RESIDUE (iii) Residue is filtered (IV) Carbon powder is SEPARATED (v) `KNO_(3)` is separated from its aqueous solution by heating it |
|
| 24. |
Arrange the following given reactions in a sequence for the process of nitrogen fixation. (A) Formation of metal nitrates (B) Formation of nitrogen doxide. ( C) Formation of itric oxide (D) Formation of nitric acid |
|
Answer» cbad (II) Formation of nitrogen dioxide (iii) Formation of nitric ACID (iv) Formation of metal NITRATES |
|
| 25. |
Arrange the following equations in the correct sequence to obtain a normal salt. (a) A1(OH)C1_(2) + HCI rarr A1C1_(3) + H_(2)O (b) A1(OH)_(3) + HC1 rarr A1(OH)_(2)C1 + H_(2)O (c) A1(OH)_(2)C1 + HC1 rarr A1(OH)C1_(2) + H_(2)O |
|
Answer» CBA (II) `AI(OH)_(2)CI + HCI rarr AI(OH)CI_(2) + H_(2)O` (iii) `AI(OH)CI_(2) + HCI rarr AICI_(3) + H_(2)O` |
|
| 26. |
Arrange the following examples in the oredr of matter, compound, element and mixture. (a) Dilute acid, (b) Argon (c ) Water, (d) Ball |
|
Answer» a,C,b,d (i) Ball (ii) Water (iii) Argon (IV) DILUTE acid |
|
| 27. |
Arrange the following equations in the correct sequence to obtain a normal salt. (a) NaOH + Na_(2)HPO_(4) rarr Na_(3)PO_(4) + H_(2)O (b) NaOH + H_(3)PO_(4) rarr NaH_(2)PO_(4) + H_(2)O (c) NaOH + NaH_(2)PO_(4) rarr Na_(2)HPO_(4) + H_(2)O |
|
Answer» bca (ii) `NaHO + NaH_(2)PO_(4) rarr Na_(2)HPO_(4) + H_(2)O` (iii) `NaOH + Na_(2)HPO_(4) rarr Na_(3)PO_(4) + H_(2)O` |
|
| 28. |
Arrange the compounds in sequence of increasing number of oxygen atoms. (a) Suger(b) Plaster of pairs (c) Starch(d) Glucose |
|
Answer» abdc (ii) Glucose (III) SUGAR (iv) Starch |
|
| 29. |
Arrange the components of air in the increasing order of their presentage by volume. (a) Nitrogen (b) Carbon dioxide ( c) Oxygen (d) Argon |
|
Answer» bdca (II) ARGON (III) Oxygen (iv) Nitogen |
|
| 30. |
Arrange statements in sequence of increasing order of heat content. (a) Ice of 10 g mass at 10^(@)C(b) water of 10 gmass at 10^(@)C(c )Ice of 10 g mass at 40^(@)C(d) Water of 10 g mass at 40^(@)C (e) Steam of 10 g mass at 10^(@)C |
|
Answer» acbde (III) WATER of 10 g mass at `10^(@)C`(iv)Water of 10 g mass at `40^(@)C` (v) Steam of 10 g mass at `10^(@)C` |
|
| 31. |
Arjun's family stays stays in an area which is in the heart of the city for sake of convenience. During the summer vacation, Arjun's family stays in their farm hous which has all amenities. As the farm house is located in the city outskirts, they could escape from the hot sum in summer. Why is the temperature in the peripheral area relatively lower than that in the heart of the city ? |
| Answer» Solution :In the interior of CITIES, the density of POPULATION is more, as a result a large volume of traffic exists which leads to and increase in the gaseous contents such as `CO_(2)`, METHANE and nitrous OXIDES which contribute to global warming. Hence the temperature in the interior of the cities is higher than in the OUTSKIRTS of the cities. | |
| 32. |
Aquatic animals are able to survive in water bodies like rivers and oceans during winter when the atmospheric temperature is -10^(@)C. This is because |
|
Answer» density of ice is less than WATER. |
|
| 33. |
Antacid pair among the following is |
|
Answer» `Ma(OH)_(2), NaOH` |
|
| 34. |
An invertad glass jar is placed over a burning candle. After a while, the candle is removed and the inverted glass jar is placed in a through in a trough of water. What do you obseve and justifyyour observetions. |
| Answer» Solution :BURNING of candle produces `CO_(2)` and water. When the CYLINDER fontaining `CO_(2)` is incerted into water through the `CO_(2)` dissolves in water as a result the water LEVEL slightly reises in the glass jarr. Thus it indicates that `CO_(2)` is soluble in water. | |
| 35. |
An element X on treatment with oxygen gives Y which on hydrolysis produces Z.Z on treatment with higher oxide of carbon 'A' gives milky white precipitate 'B'. Identify X, Y, Z, A and B? |
|
Answer» Solution :As Z on treatment with oxide "A" of carbon GIVES MILKY appearance to the solution by forming. B, Z is `Ca(OH)_(2), A "is" CO_(2), B "is" CaCO_(3)`. As Z is produved by hydrolysis of Y. Y is CAO. As Y is prepared by treating "X" with oxygen, X is Ca. `underset(("X"))(Ca + O_(2)) rarr underset(("Y"))(CaO)` `underset("Y")(CaO) + H_(2)O rarr underset("Z")(Ca(OH)_(2))` `underset("Z")(Ca(OH)_(2)) + underset("A")(CO_(2)) rarr underset("B")(CaCO_(3)) + H_(2)O` |
|
| 36. |
An element 'X' is non-malleable and non-ductile. X is extensively used to protect another element Y from corrosion.Identify X and Y and name the process thatis mentioned above. Also compare thereactivity of X and Y. |
| Answer» SOLUTION :Element X is zinc which is NON -malleable and non-ductile. Y is iron. The process of process of protecting rion by zinc is called galvanization. The element X(Zn) in more REACTIVE than Y (Fe). | |
| 37. |
An element X, in which the difference in electrons present valence shell (M shell) and penultimate shell is equal to the number of electrons present in first shenll, undergoes combustion to produce Y. Y turns lime water milky due to the formaion of Z. Identify X, Y and Z. |
|
Answer» `C, CO_(2),CaCO_(3)` Hence X is SULPHUR `underset((X))(S)+O_(2)tounderset((Y))(SO_(2))` `underset(Y)(SO_(2))+underset(("milky white"))(Ca(OH)_(2))tounderset(Z)(CaSO_(3))darr+H_(2)O` |
|
| 38. |
An element 'X', in which the difference in electrons present in valence shell and penultimate shell is equal to the number of electrons present in first shell, undergoes combustion to produce 'Y'. 'Y' turns lime water milky due to theformation of 'Z'.Identify 'X','Y' and'Z'. |
|
Answer» `C,CO_(2),CaCO_(3)` Hence X is sulphur `underset((X))(S+O_(2))tounderset((Y))(SO_(2))` `underset((Y))(SO_(2))+Ca(OH)_(2)tounderset(("MILKY white"))underset((Z))(CaSO_(3)darr+H_(2)O)` |
|
| 39. |
An element X burns with a brilliant blue flame in the presence of oxygen ot form Y. Y truns acidified potassium dichromate paper green. Identify X and Y. |
|
Answer» Nitrogen nitrogen dioxide |
|
| 40. |
An element X burns with a briliant blue flame in the presence of oxygen to form Y.Y turns acidified potassium dichromate paper green. Identigy X and Y. |
|
Answer» Nitrogen, nitrogen dioxide |
|
| 41. |
An element with electronic configuration 2, 8, 1 forms acompound with carbonate. How does this compound remove both temporary and permanent hardness of water? |
|
Answer» Solution :Element with ELECTRONIC configuration 2, 8, 1 is NA and its carbonate is SODIUM carbonate which is called washing soda. It is used for the removal of both temporary and PERMANENT hardness of water. `Ca(HCO_(3))_(2) + Na_(2)CO_(3) rarr CaCO_(3) darr + 2NaHCO_(3)` `CaSO_(4) + Na_(2)CO_(3) rarr CaCO_(3) darr + Na_(2)SO_(4)` `CaCO_(3)` is insoluble and hence settles down. In this way washing soda can be used to remove both temporary and permanent hardness of water. |
|
| 42. |
An atom of anelement 'X' has three completely filled shell . Theratio of electrons in 1 and 4 shells is 1:1. Find the atomic number of 'X' . Alsowrite the formulae of its bicarbonate , chromate and phosphide. |
Answer» Solution :The electronic arrangement of X must be 2 , 8 , 18 , 2 So the ATOMIC NUMBER of X ahould br 30 . The element X is Zn which can form`Zn^(+2)`ion by LOSING 2 electrons from N shell.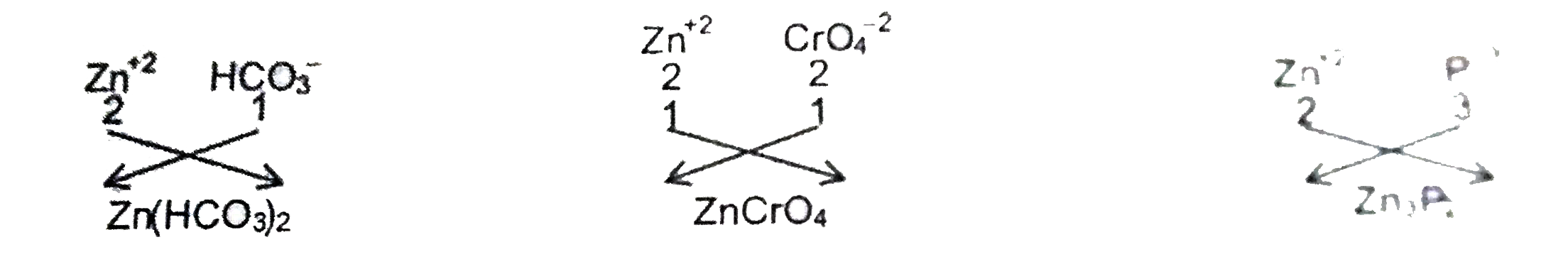
|
|
| 43. |
An atom of an element has two shells with electrons . Thevalence shell has 2n+1electrons where nis the number of electrons in the inner shell . Thiselement can form a radical 'X' with hydrogen and two radicals Y and Z with oxygen, Z has more number of oxygen than Y. identifythe element and the radicals. write the reaction taking place between solutions of two salts , AgZ and XCl . Identify the type of reaction. |
|
Answer» Solution :K shell can have only 2 electrons. That means n=2 and the number of electrons in L shell is 2n+1 which is equal to 5. The electronic arrangement that is 2,5 corresponds to nitrogen. Nitrogen can form `NH_(4)^(+)`with hydrogen. Y is `NO_(2)^(-)and Z" is " NO_(3)^(-)`. `AgNO_(3)+NH_(4)Cl toNH_(4)NO_(3)` This is a double DECOMPOSITION reaction where silver chloride being insoluble settles at the bottom. Therefore, this is a PRECIPITATION reaction. |
|
| 44. |
An atom of an element hasthe electrons distributedin four shells . If the electrons present in I and IV shells is in1:1ration and , the ratio of electrons in II and IIIshell is also1:1, predict its valency and justofy .Write the formulaeof (a) nitride (b) silicate (c ) chloride. |
|
Answer» Solution :`{:(K,L_(,),M,N),(2_(,),8,8,2):}` Hence the valency of an ATOM of an element (M) with above electronic CONFIGURATION is 2 andby losing twoelectrons it attains octetconfiguration. (a)Formula of its nitride is`M^(+2)N^(-3)toM_(3)N_(3)` (b)Formula of its silicate is`M^(+2)SiO3^(-2)toMSiO_(3)` ( C)Formula of its chloride is `M^(+2)Cl^(-)toMCl_(2)` |
|
| 45. |
An atom is electrically neutral . Justify. |
| Answer» Solution :In an atom , the number of electrons is EQUAL to the number of protons INSIDE the nucleus . SINCE the protons and electrons carry equal and oppositecharges , an atom is electriclly NEUTRAL. | |
| 46. |
An atom is electrically neutral . |
|
Answer» An atom is ELECTRICALLY NEUTRAL due to the PRESENCEOF EQUAL NUMBER of positively and negatively charged particles. |
|
| 47. |
An apartment owner called for a plumberto replace one of the leaking pipe lines. The plumber arrived andfound that the galvanized iron pipes are deteriorated andcorroded. He advised theapartment owner to replace the galvanized iron pipes with the PVCpipes. Why did the plumber give the above suggestion? |
| Answer» Solution :PVC pipes are polyvinyl chloride pipeswhich are thermoplastic in nature and more durable as theyare completely TUST free and less EXPENSIVE than galvanised iron pipes. Though galvanization prevents ruting , since the PIPELINES are in CONTINUOUS contact with water, the coating may GET worn off over a period of time and it may be corroded. | |
| 48. |
Among the noble gases which gas does not have octet configuration? |
|
Answer» Neon |
|
| 49. |
Among the following, which substance turns phenolphthalein pink ? |
|
Answer» SODA water |
|