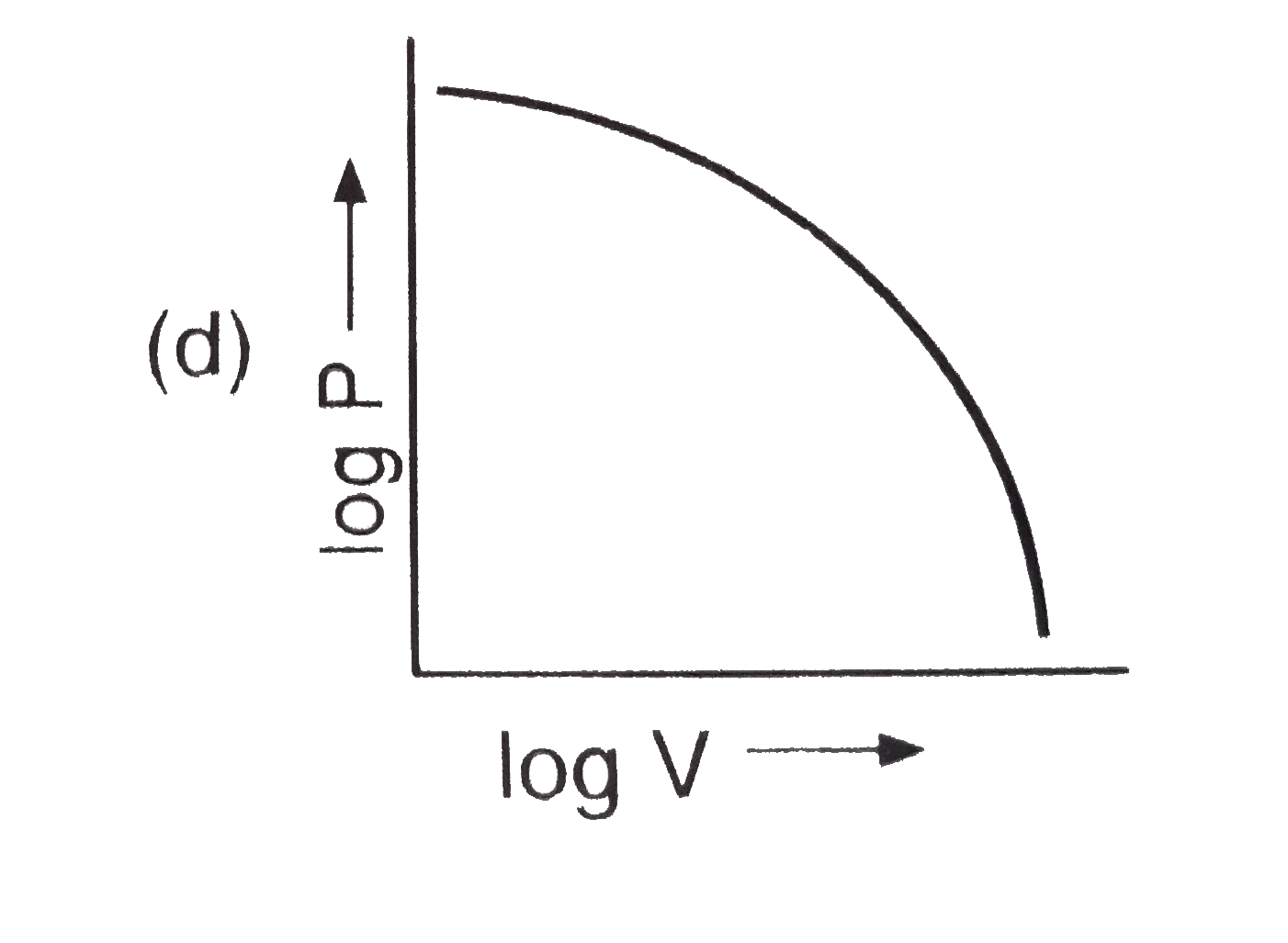Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The pollutant which deteriorates the plant cellulose |
|
Answer» BENZOPYRENE |
|
| 2. |
The pollutant released in Bhopal gas tragedy was………………. . |
|
Answer» Ammonia |
|
| 4. |
The pollutant released in Bhopal gas tragedy was......... |
| Answer» SOLUTION :METHYL ISOCYANATE | |
| 5. |
The pollutant released in Bhopal gas tragedy was : |
|
Answer» Ammonia |
|
| 6. |
The polarising power of which of the following pair is similar |
|
Answer» `LI, MG` |
|
| 7. |
The polarising power of Mg is almost same as |
|
Answer» Li |
|
| 8. |
The pOH of a solution is 5.725. Calculate the [H^(+)]. |
|
Answer» Solution :`pH+pOH=14""pH=14-pOH=14-5.725=8.275` `pH=-log_(10)[H^(+)]rArr8.275=log_(10)[H^(+)]RARR` `[H^(+)]="anti"LOG(-8.275)="anti"log(9-8.275-9)` `[H^(+)]="anti"log(0.275-9)=5.309xx10^(-9)"mol"dm^(-3)` |
|
| 9. |
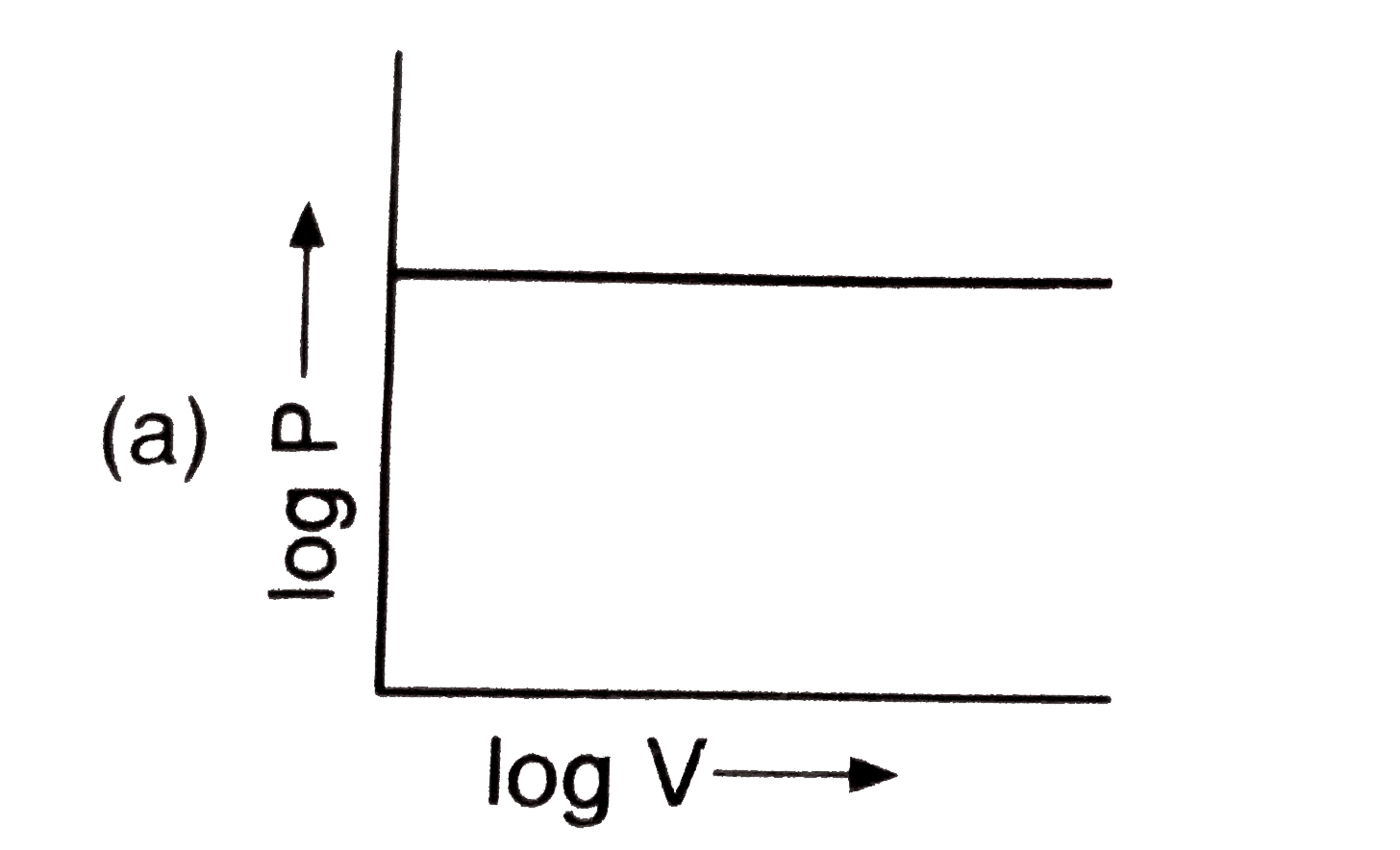
The plot of volume versus pressureat constant temperature is a……………………… |
|
Answer» |
|
| 10. |
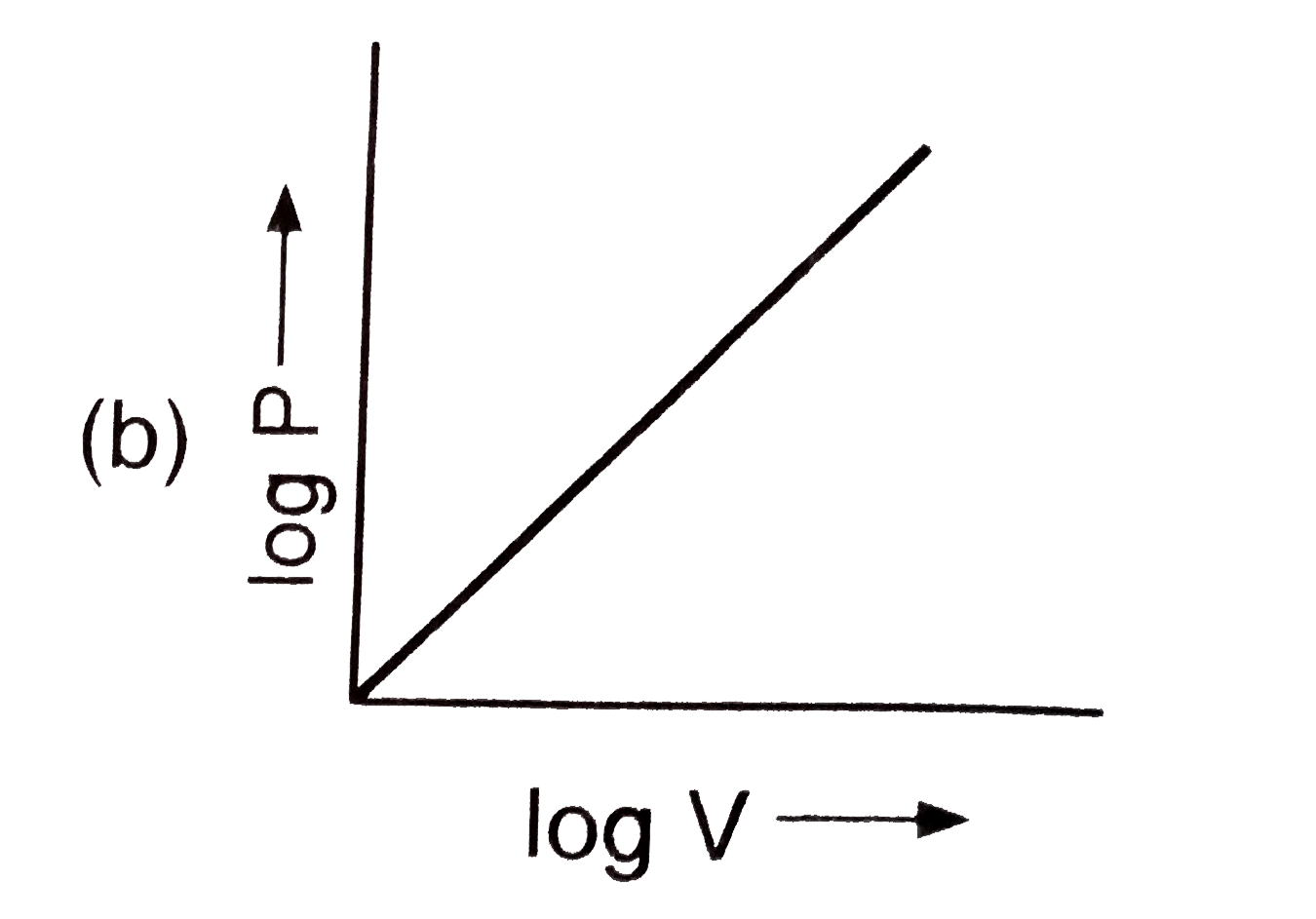
The plot of log V against log P at constant temperature for a fixed mass of gas is : |
|
Answer»
|
|
| 11. |
The platinum-chlorine distance has been found to be2.32Å in several crystalline compounds This value applies to both compounds A and B given here Based on the above structures, answer the following questions The C-C single-bond distnaance is 1.54 Å What is the distance between the terminal carbons is propane ? Assume that the four bonds of any carbon atoms arre pointed towards the corners of a reqular tetrahedron . |
|
Answer» `3.08Å` Distance between two terminal C-atoms `=2x` `:. X =1.54 SIN ((109^(@)28')/(2))` ` = 1.26 Å` 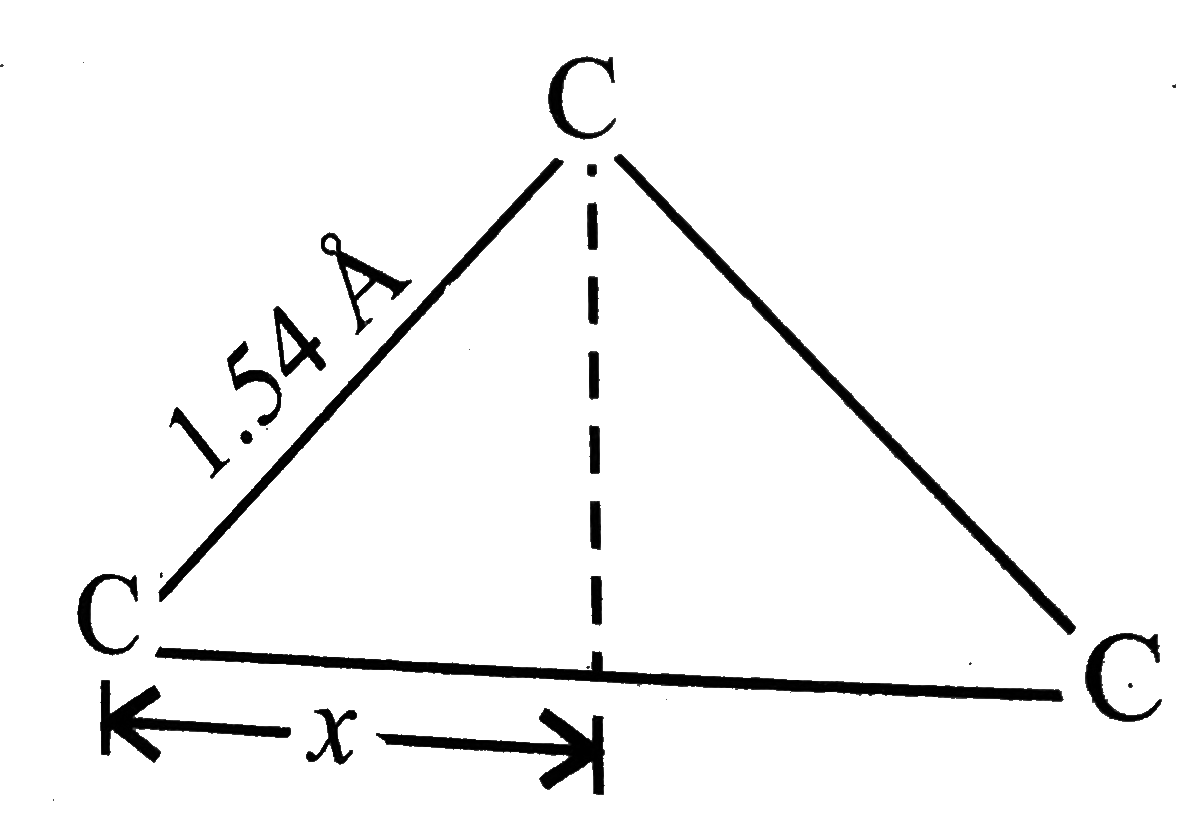 . .
|
|
| 12. |
The platinum-chlorine distance has been found to be2.32Å in several crystalline compounds This value applies to both compounds A and B given here Based on the above structures, answer the following questions Structure B is . |
|
Answer» cis-isomer |
|
| 13. |
The pK_(ln) of an indicator is 10.5. For which pH transition range is the indicator most suitable ? |
| Answer» SOLUTION :`pK_(LN)pm1, i.e., 9.5 ` to 11.5 . | |
| 14. |
The placement of element in the periodic table is closely related to its__________ |
|
Answer» OUTER SHELL ELECTRONIC configuration |
|
| 15. |
The pK_(al) and pK_(a_2)of an amino acid are 2.3 and 9.7 respectively. The isoelectric point of amino acid is |
|
Answer» `12` ` =(1)/(2)[2.3 +9.7 )=6` |
|
| 16. |
The pKa values of theacids A to D are found to be 4.19,3.41, 4.46 and 4.76. The acid having pKa of 3.41 is |
| Answer» ANSWER :C | |
| 17. |
The pK_(a) of weak acid (HA) is 4.5 . The pOH of an aqueous buffered solution of HA is which 50% of the acid is ionized is |
|
Answer» `7.0` `PH = pK_(a)+log .([A^(-)])/([HA])=pK_(a)=4.5 ` (Given) `pOH = 14- pH = 14 - 4.5 = 9.5. |
|
| 18. |
The pK_(a) value of acetic acid is 4.7447 at 25^(@)C. How would you obtain a buffer of acetic acid and sodium acetate with pH = 4? |
|
Answer» Solution :`pH=pK_(a)+"log"(["SALT"])/(["Acid"])rArr4=4.7447+"log"(["Salt"])/(["Acid"])` `"log"(["Salt"])/(["Acid"])=4.000-4.7447=-0.7447=1.2553` Taking antilog on both sides, `therefore(["Salt"])/(["Acid"])="anti"log(-1.2553)rArr(["Salt"])/(["Acid"])=0.18` So by MIXING sodium ACETATE and acetic acid in the ratio 0.18 : 1 mole per `dm^(3)`, the BUFFER of pH = 4 is obtainetd. |
|
| 19. |
The pk_a of phenol is 10. What is the concentration of phenolate anion in 0.05M aqueous phenol ? |
| Answer» SOLUTION :`2.23 XX 10^(-6)M` | |
| 20. |
The pK_(a) of an acid HA is 4.77 and pK_(b) of a base BOH is 4.75 . The pH of 0.1 M aqueoussolutionof the salt AB is |
|
Answer» 7.02 `pH = 7 + (1)/(2) (pK_(a)-pK_(b))` `=7 + (1)/(2) (4.77-4.75)= 7.01` |
|
| 21. |
The pK_(a) of acetylsalicylic acid (aspirin) is 3.5. The pH of gastric juice in human stomach is about 2-3 and pH in the small intestine is about 8. Aspirin will be |
|
Answer» Unionized in the small intestine and in the stomach |
|
| 22. |
The pK_(a) ofacetic acid is 4.74 . The concentration of CH_(3)C O O His 0.01 M. The pH ofCH_(3) C O O H is |
|
Answer» 3.37 `=(1)/(2)[4.74-log 10^(-2)]=(1)/(2)xx6.74=3.37` |
|
| 23. |
The pK_a of acetic acid and pK_b of ammonium hydroxide are 4.76 and 4.75 respectively . Calculate the pH of ammonium acetate solution. |
|
Answer» Solution :`pK_a` of `(CH_3COOH)`= 4.76 `pK_b` of `(NH_4OH)`= 4.75 Ammonium ACETATE is a SALT of this both. The PH of ammonium acetate solution is calculate by FOLLOWING EQUATION. `pH=7+1/2(pK_a + pK_b)` `=7+1/2(4.76-4.75)` `=7+1/2(0.01)=7+0.005 = 7.005` |
|
| 24. |
The pK_(a)ofacetic acid and pK_(b) of ammonium hydroxide are 4.76 and 4.75 respectively. Calculate the hydrolysis constant of ammonium acetate at 298K and also the degree of hydrolysis and pH of (a) 0.01 M and (b) 0.04 M solutions. |
|
Answer» |
|
| 25. |
The pK_a of a weak acid, HA, is 4.80. The pK_b of a weak base, BOH is 4.78. The pH of an aqueous solution of the corresponding salt BA, will be |
|
Answer» 9.22 `therefore` pH=7+1/2 `(pK_a-pK_b)` =7+1/2(4.80+4.78) =7.01 `K_2=([CO][H_2]^3)/([CO_4][H_2O])`…(2) `CH_(4(G)) + 2H_2O_((g)) hArr CO_(2(g)) + 4H_(2(g))` `K_3=([CO_2][H_2]^4)/([CO_4][H_2O]^2)` ....(3) |
|
| 26. |
The pK_a of a weak acid (HA) is 4.5. The pOH of an aqueous buffered solution of HA in which 50 % of the acid is ionized is |
|
Answer» `7.0` When the ACID is 50% ionised , `[A^-]=[HA]` or pH=`pK_a+log 1` or pH = `pK_a` Given `pK_a=4.5` `therefore` pH=9.5 `therefore` pOH=14-4.5=9.5 |
|
| 27. |
The pK_(a) fo acetic acid and pK_(b) of ammonium hydroxide are 4.76 and 4.75 respectively .Calculate the pH of ammonium acetate solution . |
|
Answer» SOLUTION :As `CH_(3)CO ONH_(4) ` is a SALT of weak ACID, weak base, `pH = 7 + (1)/(2) [pK_(a)-pK_(b)] = 7 + (1)/(2) (4.76-4.75) = 7 + 0.005 = 7. 005` |
|
| 28. |
The pK_a and pK_b of one weak acid (HA) and weak base (BOH) are 3.2 and 3.4 respectively. Find pH of its salt AB. |
|
Answer» 7.2 `pH=1/2(pK_w+pK_a-pK_b)` `=1/2(14+3.2-3.4)` `=1/2(13.8)=6.9` |
|
| 29. |
The piston shown in the figure is moved downwards such that the vapour volume is decreased by 246.3cc. How many grams of H_(2)Ois condensed? (Assume aqueous tension at 27°C is 360 mm Hg). |
|
Answer» 1.8 gram `W = (0.5 XX 0.2463 xx 18)/(0.0821 xx 300) = 0.09 GM`. |
|
| 30. |
The pine oil used in froth floatation process an example of |
|
Answer» FROTH stabilizer |
|
| 31. |
The physical state of dispersed phase and dispersion medium in colloid like pesticide spray respectively are |
|
Answer» solid,GAS |
|
| 32. |
Which of the following a larger value for D_(2)O than H_(2)O |
|
Answer» FREEZING and boiling POINTS |
|
| 33. |
The physical constants which are less for D_(2)O than H_(2)O are |
|
Answer» FREEZING POINT and BOILING point |
|
| 34. |
The physical constants which are less for D_(2)Othan H_(2)O are |
|
Answer» Freezing point and BOILING point |
|
| 35. |
The physical constants which are less for D_2O than H_2O are |
|
Answer» FREEZING and BOILING points |
|
| 36. |
The physical constant which is less for water than for heavy water is |
|
Answer» DIELECTRIC constant |
|
| 37. |
The photonhaving6000awavelengthhas energyE. Ifthe energyof photon2E findthewavelengthof photon. |
| Answer» SOLUTION :`3000A` | |
| 38. |
The photon has a momentum as well as wavelength Which property of matter is revealed in the above statement? |
| Answer» SOLUTION :DUAL NATURE of RADIATION and MATTER | |
| 39. |
The photochemical smog is essentially caused by presence by |
|
Answer» `O_(2) and O_(3)` |
|
| 40. |
The phenomenon of Synersis is |
|
Answer» SEPARATION of the DISPERSED PHASE FORM the gel |
|
| 41. |
The phenomenon of spontaneous nuclear disintegration of radioactive elemets. Isotopes by emitting alpha, beta or gamma -rays in order to give stable nucleus is known as radioactivity. It is totally a nuclear phenomenon i.e., only depends upon the nucleons . It is alos known as activity and is given by A=lambdaN where lambda =decay constant N=no. of atoms at time t Activity per gram of the sample is known as specific activity . The cause of radioactivtiy of a nucleide can be attributed to a certain degree to its neutron to proton ratio i.e., n/p ratio. Lighter nucleides with n/p =1 or slightly greater than one, are non-radioactive or stable nucleides. Nucleides with Z gt 20 , requires large number of neutrons than protons to moderate the effect of the increasing protonic repulsions . Nucleides with Z gt 83 , the protonic repulsions are too large to overcome by proton -neutron interactions . The stable nuclei lie within zone of stability , all the nuclei which fall outside of this zone are invariably radioactive and unstable in nature. Nuclei which are above the stability zone have excess protons. These nuclei attain stability by making adjustments in n/p ratio for moderate value. Choose the correct statement(s) |
|
Answer» The ACTIVITY of the same radioactive element for 5 CC and 10 cc volume will be same |
|
| 42. |
The phenomenon of spontaneous nuclear disintegration of radioactive elemets. Isotopes by emitting alpha, beta or gamma -rays in order to give stable nucleus is known as radioactivity. It is totally a nuclear phenomenon i.e., only depends upon the nucleons . It is alos known as activity and is given by A=lambdaN where lambda =decay constant N=no. of atoms at time t Activity per gram of the sample is known as specific activity . The cause of radioactivtiy of a nucleide can be attributed to a certain degree to its neutron to proton ratio i.e., n/p ratio. Lighter nucleides with n/p =1 or slightly greater than one, are non-radioactive or stable nucleides. Nucleides with Z gt 20 , requires large number of neutrons than protons to moderate the effect of the increasing protonic repulsions . Nucleides with Z gt 83 , the protonic repulsions are too large to overcome by proton -neutron interactions . The stable nuclei lie within zone of stability , all the nuclei which fall outside of this zone are invariably radioactive and unstable in nature. Nuclei which are above the stability zone have excess protons. These nuclei attain stability by making adjustments in n/p ratio for moderate value. Nucleide with low n/p ratio, which are below the zone of stability , the stability cannot be achieved by |
|
Answer» `BETA`-emission |
|
| 43. |
The phenomenon of spontaneous nuclear disintegration of radioactive elemets. Isotopes by emitting alpha, beta or gamma -rays in order to give stable nucleus is known as radioactivity. It is totally a nuclear phenomenon i.e., only depends upon the nucleons . It is alos known as activity and is given by A=lambdaN where lambda =decay constant N=no. of atoms at time t Activity per gram of the sample is known as specific activity . The cause of radioactivtiy of a nucleide can be attributed to a certain degree to its neutron to proton ratio i.e., n/p ratio. Lighter nucleides with n/p =1 or slightly greater than one, are non-radioactive or stable nucleides. Nucleides with Z gt 20 , requires large number of neutrons than protons to moderate the effect of the increasing protonic repulsions . Nucleides with Z gt 83 , the protonic repulsions are too large to overcome by proton -neutron interactions . The stable nuclei lie within zone of stability , all the nuclei which fall outside of this zone are invariably radioactive and unstable in nature. Nuclei which are above the stability zone have excess protons. These nuclei attain stability by making adjustments in n/p ratio for moderate value. Nucleide with high n/p ratio which are above the zone of stability , can stability cannot be achieved by |
|
Answer» `BETA`-EMISSION |
|
| 44. |
The phenomenon of spontaneous nuclear disintegration of radioactive elemets. Isotopes by emitting alpha, beta or gamma -rays in order to give stable nucleus is known as radioactivity. It is totally a nuclear phenomenon i.e., only depends upon the nucleons . It is alos known as activity and is given by A=lambdaN where lambda =decay constant N=no. of atoms at time t Activity per gram of the sample is known as specific activity . The cause of radioactivtiy of a nucleide can be attributed to a certain degree to its neutron to proton ratio i.e., n/p ratio. Lighter nucleides with n/p =1 or slightly greater than one, are non-radioactive or stable nucleides. Nucleides with Z gt 20 , requires large number of neutrons than protons to moderate the effect of the increasing protonic repulsions . Nucleides with Z gt 83 , the protonic repulsions are too large to overcome by proton -neutron interactions . The stable nuclei lie within zone of stability , all the nuclei which fall outside of this zone are invariably radioactive and unstable in nature. Nuclei which are above the stability zone have excess protons. These nuclei attain stability by making adjustments in n/p ratio for moderate value. Which of the following statement(s) is (are) correct ? |
|
Answer» Radioactivity only DEPENDS upon the nucleons |
|
| 45. |
The phenomenon of spontaneous nuclear disintegration of radioactive elemets. Isotopes by emitting alpha, beta or gamma -rays in order to give stable nucleus is known as radioactivity. It is totally a nuclear phenomenon i.e., only depends upon the nucleons . It is alos known as activity and is given by A=lambdaN where lambda =decay constant N=no. of atoms at time t Activity per gram of the sample is known as specific activity . The cause of radioactivtiy of a nucleide can be attributed to a certain degree to its neutron to proton ratio i.e., n/p ratio. Lighter nucleides with n/p =1 or slightly greater than one, are non-radioactive or stable nucleides. Nucleides with Z gt 20 , requires large number of neutrons than protons to moderate the effect of the increasing protonic repulsions . Nucleides with Z gt 83 , the protonic repulsions are too large to overcome by proton -neutron interactions . The stable nuclei lie within zone of stability , all the nuclei which fall outside of this zone are invariably radioactive and unstable in nature. Nuclei which are above the stability zone have excess protons. These nuclei attain stability by making adjustments in n/p ratio for moderate value. Which of the following statement is correct ? |
|
Answer» The radioactivity of `RaCl_(2)` and RA will be same |
|
| 46. |
Splitting of spectral lines in the presence of electric field is called _______ |
|
Answer» PHOTOELECTRIC EFFECT |
|
| 47. |
The phenomenon of negative catalysis is |
|
Answer» Autocatalysis |
|
| 48. |
The phenomenon in which atoms or groups in a compound can attract electrons, is called |
|
Answer» mesomeric EFFECT |
|
| 49. |
The phenomenon by which activated charcoal removes colouring matter from pure substances is |
|
Answer» ADSORPTION |
|