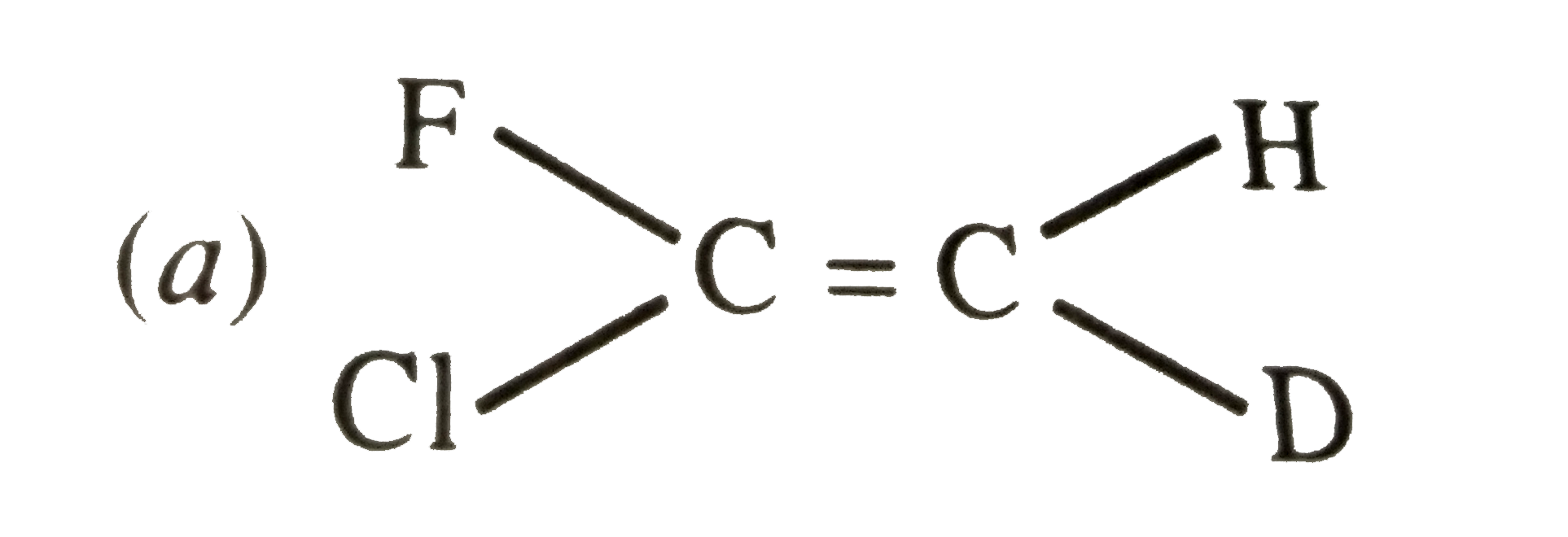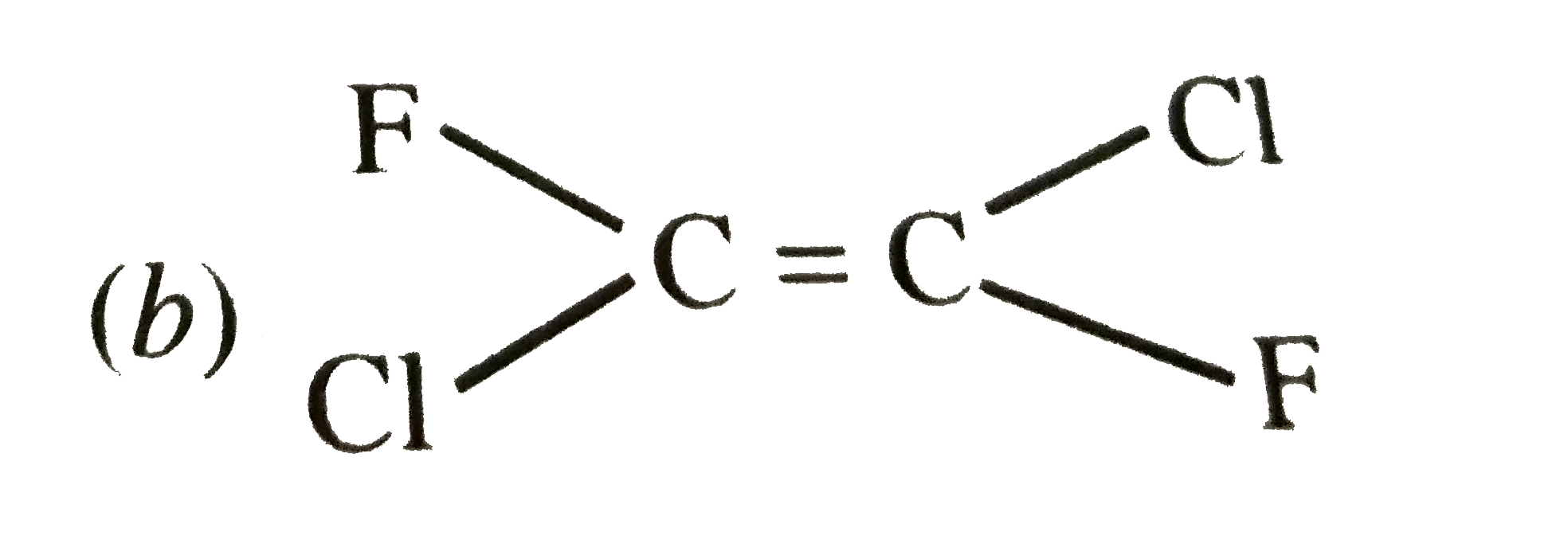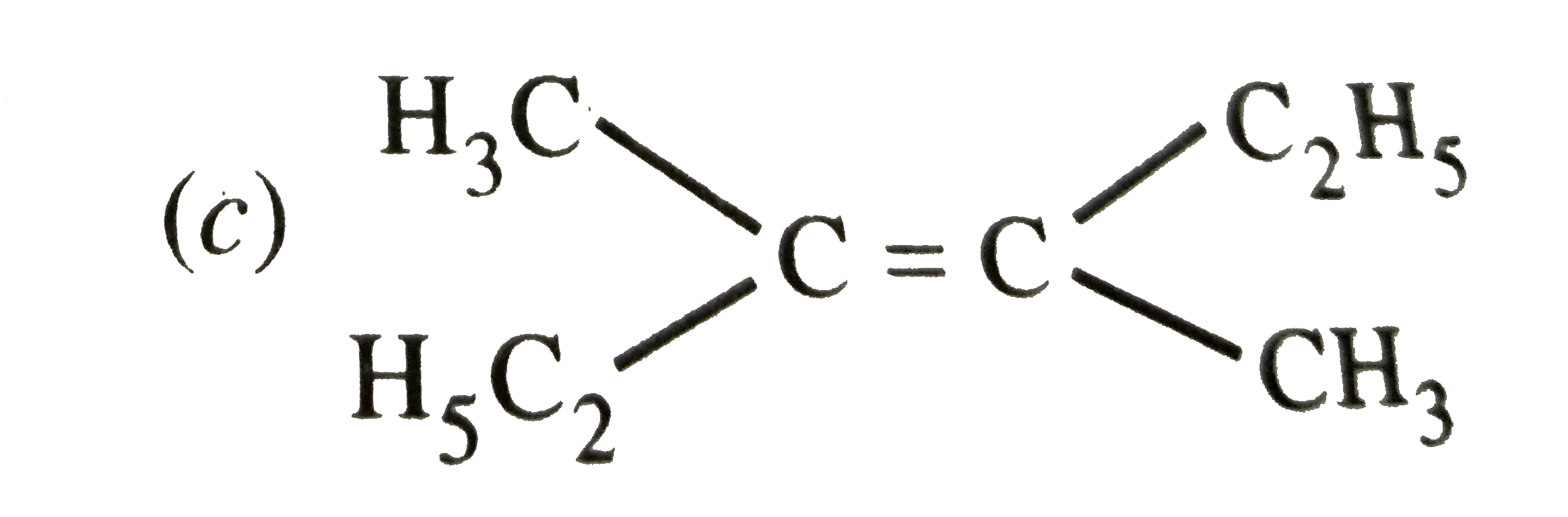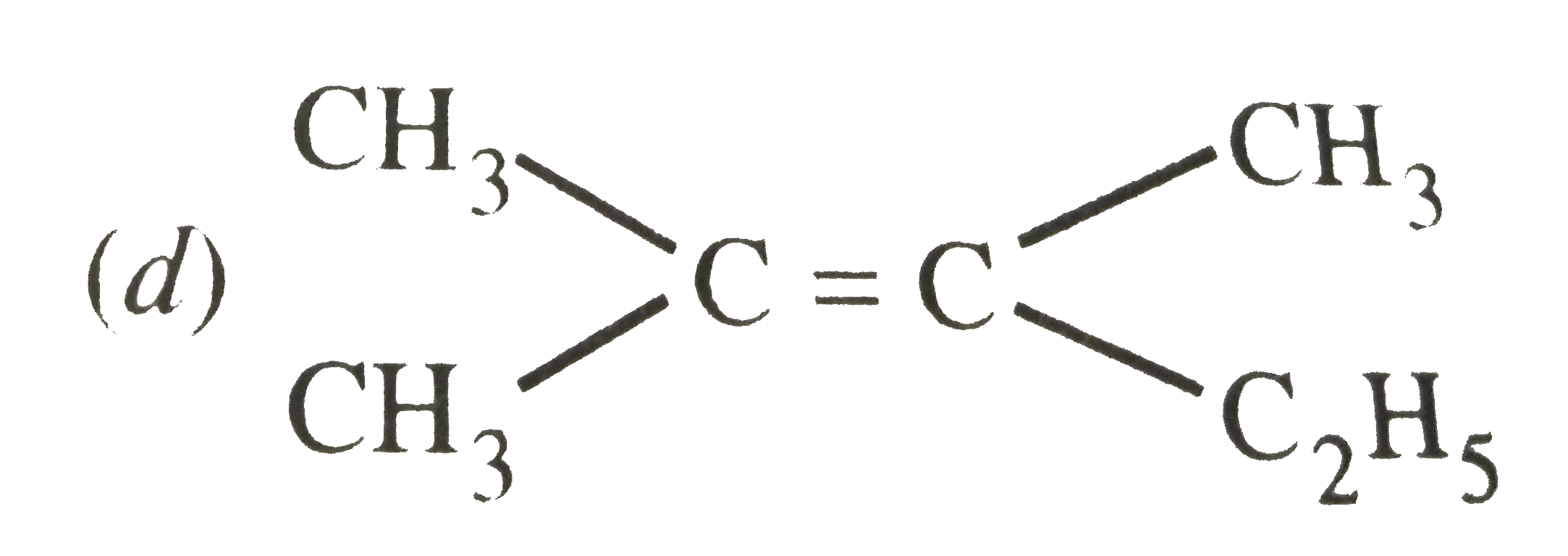Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Two members of a homologous series have different |
|
Answer» GENERAL formula |
|
| 2. |
Which of the following will not show geometrical isomerism ? |
|
Answer»
|
|
| 3. |
Which of the following molecule does not obey the octet rule and also has lone pair on the central atom. |
|
Answer» `C Cl_4` |
|
| 4. |
Which of the following cannot be prepared by wurtz reaction in good yield? |
|
Answer» `(CH_3)_2CHCH_2CH(CH_3)_2` 
|
|
| 5. |
Which combination is not give molecular orbital from 1s - 1s and 1 s - 2s ? Why ? |
| Answer» SOLUTION :Combination of 1S - 2s is not GIVE molecular orbital because the energy difference is more between them. | |
| 6. |
Which of the following orders regarding the bond order is correct? |
|
Answer» `O_(2)^(-)GT O_(2) gt O_(2)^(+)` |
|
| 7. |
The study of toxicity of organo metallic compounds is termed as |
|
Answer» CLASSIFICATION |
|
| 8. |
Which of the follwing has noreaction with water? |
|
Answer» Ba |
|
| 9. |
When zeolite which is hydrated sodium aluminium silicate is treated with hard water, the sodium ions are exchanged with: |
|
Answer» `Ca^(2+)` IONS `RARR CaAL_(2)Si_(3)O_(3).xH_(2)O+2Na^(+)` or `MgAI_(2)Si_(3)O_(8).xH_(2)O` |
|
| 10. |
What indicated by figure ? |
| Answer» SOLUTION :Here anhydrous `CaCl_2` in evaporating dish and pressure is same in manometer. The MOISTURE in BOX is ABSORB by `CaCl_2` so there is not EQUILIBRIUM. | |
| 11. |
Which of the following reaction are disproportionstion reaction and comproportionaton reactions ? Ag^(2+)Ag to2Ag^(+) |
|
Answer» Solution :`overset(+2)(AG^2)+overset(0)(Ag)tooverset(+1)(2Ag^+)` The OXIDATION numbers of the reaction `Ag^(2+)` are +2 and 0 respectively and the oxidation NUMBER is INTERMEDIATE between the oxidation numbers +2 and 0. So it is a comproportionation reaction. |
|
| 12. |
Which of the following is more reactive than diphenyl regarding electrophilic aromatic substitution? |
|
Answer»
|
|
| 13. |
Which of the following steps is not correct in the mechanism of electrophilic substitution of benzene? |
|
Answer» Generation of electrophile LIKE `X^(+),R^(+),RC^(+)O,NO_(2)^(+),`etc. 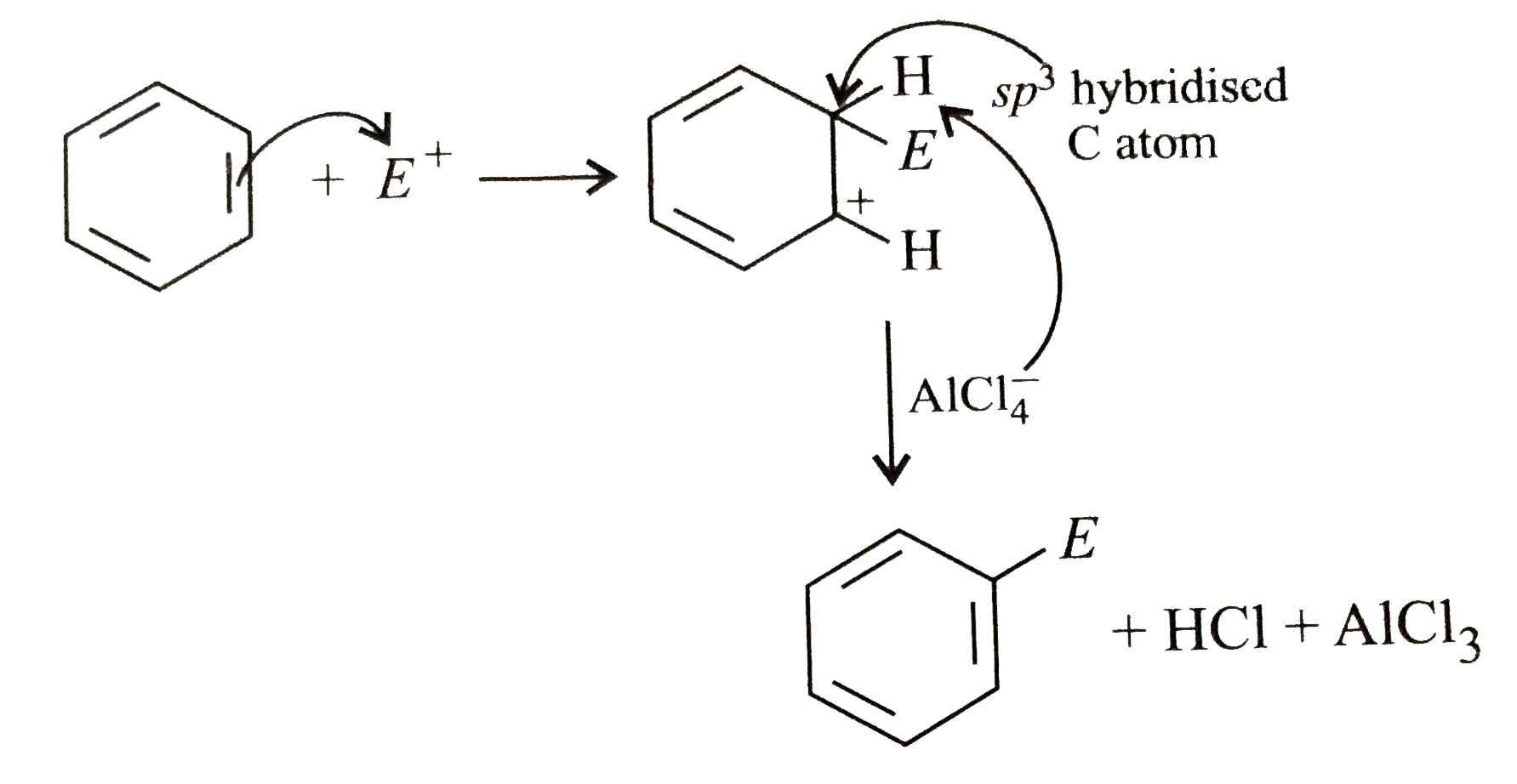
|
|
| 14. |
When 40 mL ofa 0.1M weak monoacid base is titrated with 0.16M HCI, the pH of solution at the end point is 5.23. Calculate K_(b). What will be the pH if 15 mL of 0.12M NaOH is added to the resulting solution ? |
|
Answer» |
|
| 15. |
The type of hybridizationof boron in diborane is |
|
Answer» SP-HYBRIDIZATION |
|
| 16. |
Which one of the following alkaline earth metal carbonates is thermally most stable? |
| Answer» SOLUTION :Thermal STABILITY increases with decreases in cationic SIZE. | |
| 17. |
Total number of moles of HCl reacts with one mole of borax to converts all borons to boric acid? |
|
Answer» |
|
| 18. |
X is pale yellow solid, It hydrolyess to POCl_(3) in moist air and finally gets converted into phosphoric acid. X exists as an ionic solid. The total number of atoms present in its cation is. |
|
Answer» `POCl_(3)+3H_(2)OtoH_(3)PO_(4)+3HCl` In the solid STATE, `PCl_(5)` (X) EXISTS as `[PCl_(4)]^(+)[PCl_(6)]^(-)` in which the CATION has 5 atmos (4Cl+1P). |
|
| 19. |
The two type of bonds present in B_(2)H_(6) are covalent and |
|
Answer» ionic 
|
|
| 20. |
Whichof thefollowingis anexampleofamorphousubstance ? |
|
Answer» GLUCOSE |
|
| 21. |
Which of the following is the most reactive towards electrophilic substitution reaction ? |
|
Answer»
|
|
| 23. |
What are total dissolved solids (TDS)? |
|
Answer» Solution :(i) Most of the salts are soluble in water. It include CATIONS like calcium, magnesium, sodium, POTASSIUM. iron and anions like carbonate, bicarbonate, chloride, sulphate, phosphate and nitrate. (ii) USE of drinking water having TDS (total dissolved solids) concentration higher than 500 ppm causes POSSIBILITIES of irritation in stomach and INTESTINE |
|
| 24. |
The volume of a gas measured at 27^@C and 1 atm pressure is 10 litres. To reduce the volume to 5 litres at 1 atm pressure, the temperature required is |
| Answer» ANSWER :B | |
| 25. |
What is the volume of 1.5 N H_2O_2 ? |
|
Answer» SOLUTION :Normality of `H_2O_2= (H_2O_2 "volume STRENGTH")/5.6` `therefore` Volume strength= 1.5 X 5.6 = 8.4 `H_2O_2` |
|
| 26. |
which of the following products is not formed in above reaction : products are |
|
Answer» `H-underset(O)underset(||)(C)-H` 
|
|
| 27. |
Which of the following reactions is not correct for preparation of alkanes ? |
|
Answer» `(CH_3)_2CHCl OVERSET(LiAlH_4)to (CH_3)_2CH_2` |
|
| 28. |
Which of the following has maximum density ? |
|
Answer» `O_2` at `25^@C` and 1 ATM |
|
| 29. |
Write name of Fe_(2)(SO_(4))_(3) according to stock notation method. |
| Answer» SOLUTION :IRON (III) SULPHATE | |
| 30. |
What is bulk properties of matter ? Give examples. |
|
Answer» Solution :Definition : The properties associated with a COLLECTION of a large number of atoms IONS and MOLECULES. Example : (i) An individual molecule of a liquid does not oil but the BULK boils. (ii) Collection of water molecules have wetting properties, individual molecules do not wet. |
|
| 31. |
Write formula and name about alkyl groups |
|
Answer» Solution :(A) In branched chain hydrocarbons the alkyl group present as a branch. Alkyl groups (-R): The removed of one hydrogen atom from the molecule of an alkane GIVES an alkyl group. `underset("alkane")(R- H) overset(-H)rarr underset("alkyl group")(-R)` Nomenclature (Ane + AYL = Alkyl) this SMALL alkyl groups are in branced chain hydrocarbons: In a branched chain compound small chains of carbon ATOMS are attached at one or more carbon atoms of hte parent chain. The small carbon chains (branches) are called alkyl groups: For example: (i) `CH_(3) - underset(underset(CH_(3))(|))(CH) - CH_(2) - CH_(3)` In four carbon containing parent chain, the `-CH_(3)` (Methyl) alkyl group is in branch. (ii) `CH_(3) - underset(underset(CH_(2)CH_(3))(|))(CH) - CH_(2) - overset(overset(CH_(3))(|))(CH) - CH_(3)` In five carbon containing parent chain the `-CH_(3) and -CH_(2)CH_(3)` groups in branch. (B) Nomenclature of alkyl group: In order to name such compounds,the name of alkyl groups are prefixed to the name of parent alkane. General formula alkyl group: `C_(n) H_(2n + 1)` Name of alkyl group : (Ane + yl) = Alkyl. One H is less in alkyl group than correspond in alkane.  The branch and unbranch structures of propyl, butyl and pentyl are as following. (a) Structure and name of propyl: `C_(3)H_(7)` (i) `underset("n- propyl")(CH_(3) CH_(2) CH_(2))`- (ii) `underset("isopropyl")(CH_(3) - underset(underset(CH_(3))(|))(CH)-)` (b) Structure and name of butyl: `C_(4)H_(9)` (i) `underset("n -butyl")(CH_(3) - CH_(2) - CH_(2) - CH_(2)-)` (ii) `underset("isobutyl")(CH_(3) - underset(underset(CH_(3))(|))(CH) - CH_(2) -)` (iii) Secondary butyl: `CH_(3) - CH_(2) - overset(|)(C )H - CH_(3) OR CH_(3) - CH_(2) - underset(underset(CH_(3))(|))(CH) -` (c ) Structure and names of pentyl: `C_(5)H_(11)` (i) `underset("n-pentyl")(CH_(3) - CH_(2) - CH_(2) - CH_(2) - CH_(2) -)` (ii) Second pentyl: `{:(CH_(3)- CH- CH_(2)- CH_(2) - CH_(3)),("|"),(CH_(3) - CH_(2)- CH_(2) - CH-),("|"),(""CH_(3)):}` OR `{:(CH_(3)- CH_(2) - CH- CH_(2) - CH_(3)),("|"),(CH_(3)CH_(2)- CH- ),("|"),(""CH_(2)CH_(3)):}` (iii) Isopentyl `CH_(3) - underset(underset(CH_(3))(|))(CH) - CH_(2) - CH_(2)- OR (CH_(3))_(2) CH CH_(2) CH_(2)-` (iv) Neopentyl `CH_(3) - underset(underset(CH_(3))(|))overset(overset(CH_(3))(|))(C ) - CH_(2) - OR (CH_(3))_(3) C- CH_(2) -` |
|
| 32. |
What reaction will take place if a silver coin is put in dillute HNO_(3)? |
|
Answer» |
|
| 33. |
What is the equivalent weight of SnCl_(2) in the reaction, SnCl_(2) + Cl_(2) to SnCl_(4)? (mol.wt. of SnCl_(2)=190) |
|
Answer» 95 EQ. wt. of `SnCl_(2)` = Eq. wt of `Cl_(2)` `therefore` (190)/(E_(1)= (71)/(35.5)` or `E_(1) =95` |
|
| 34. |
What is covalency ? |
| Answer» SOLUTION :NUMBER of electrouns SHARED between the atoms during covalent bond formation. | |
| 35. |
When zeolite which is hydrated sodium aluminium silicate is treated with hard water, the sodium ions are exchanged with |
|
Answer» `CA^(2+)` ions `rarr CaAL_(2)Si_(3)O_(3).xH_(2)O+2Na^(+)` or `MgAI_(2)Si_(3)O_(8).xH_(2)O` |
|
| 36. |
The values of electronegativity of atoms A and B are 1.2 and 4.0 respectivity . The % ionic character of the A-B bond is |
|
Answer» `50%` % ionic character = `16 (chi_(A) - chi_(B)) + 3.5(chi_(A) - chi_(B))^(2)` ` 16 (4-0 - 1.2) + 3.5 (4 .0 - 1.2)^(2)` `= 16 xx2.8 + 3.5 xx (2.8)^(2)` `= 44.8 + 27 .44 = 72.24` |
|
| 37. |
What is K_(c) for the following equilibrium when the equilibrium concentration of each substance is : [SO_(2)]= 0*60 M, [O_(2)]= 0*82 M and [SO_(3)] = 1*90 M ? 2 SO_(2) (g) + O_(2) (g) hArr 2 SO_(3) (g) K_(c) = [SO_(3)]^(2)/([SO_(2)]^(2) [O_(2)])= (1*90M)^(2)/((0*60 M)^(2) (0*82M)= 12*229 )=12*229 M^(-1) or 12* 229 L mol^(-1) |
| Answer» SOLUTION :` K_(c) = [SO_(3)]^(2)/([SO_(2)] [O_(2)]) = (1.90 M)^(2)/((0*60 M) ^(2) (0.82M))=12*229 M ^(-1) or 12*229 L " mol"^(-1)` | |
| 38. |
When the Normality and Molarity is same ? |
|
Answer»
|
|
| 39. |
which one of the following does not have intramolecular hydrogen bonding? |
|
Answer» water |
|
| 40. |
Which of the following is true about the charge acquired by p-type semiconductors ? |
|
Answer» POSITIVE |
|
| 41. |
Which of the following reaction is oxidation and which is reduction ? {:((i),2H_2O+2e^(-),rarr,2OH^(-)+H_2),((ii), Al,rarr,Al^(3+)+3e^(-)),((iii) , Fe^(3+),rarr,Fe^(2+)-e^(-)),((iv),2O^(2-)+C,rarr,CO_2+4e^(-)),((v) ,Br^(-),rarr,1//2Br_2+e^(-)),((vi), 2H_2O-4e^(-), rarr,4H^(+)+O_2):} |
|
Answer» |
|
| 42. |
Water gas is produced by |
|
Answer» PASSING steam through a RED hot coke bed. `underset"Steam"(H_2O) + underset"Red hot"C to underset"Water gas"(H_2+CO)`-28 kcal |
|
| 43. |
Which of the following statements is not true about classical smog? |
|
Answer» Its main components are produced by the action of sunlight on emissions of auto mobiles and factories. NOTE: Gases released by automobiles and factories are not responsible for classical smog. |
|
| 44. |
Which of the following is the strongest base |
|
Answer» `CH_(3)COO^(-)` |
|
| 45. |
Which of the following is not soluble in NaOH? |
|
Answer» `Fe(OH)_(3)` |
|
| 46. |
Write the IUPAC names of the following halides a. CH_(2)BrCHCl CH_(2)CH_(2)Cl b. CH_(3)C Cl_(2)CHBr.CH_(3) c. CH_(3)-underset(CH_(3))underset("|")"CB"r-underset(Cl)underset(|)CH-underset(CH_(3))underset("|")"CH"-CH_(3) |
|
Answer» Solution :a. Isobutyl BROMIDE b. Propylidene chloride c. Propylidene iodide d. Bromoform E. CARBON TETRA chloride. |
|
| 47. |
Which of the following are biodegradable pollutants |
|
Answer» Pesticides |
|
| 49. |
x mole HI is taken in 10 lit vessel and heated . If the equilibrium concentration of H_(2 (g)) is 0.1 mol - lit^(-1) , value of x and mass of I_(2) at equilibrium respectively are |
|
Answer» 2, 254 G |
|
| 50. |
What will be DeltaH for the reaction, CH_(2)Cl_(2)rarrC+2H+2Cl? (B.E. of C-H and C-Cl bonds are "416 kJ mol"^(-1) and "325 kJ mol"^(-1) respectively) |
|
Answer» Solution :`H-OVERSET(H)overset(|)UNDERSET(Cl)underset(|)C-ClrarrC=2H+2Cl` `DeltaH=SigmaB.E._(R)-SigmaB.E._(P)` `=2xxB.E.(C-H)+2xxB.E.(C-Cl)` `=2xx416+2xx325="1482 kJ"` |
|