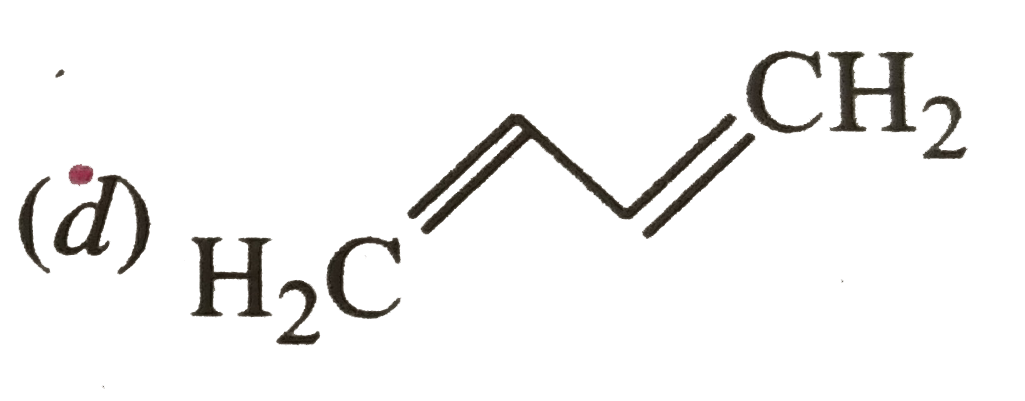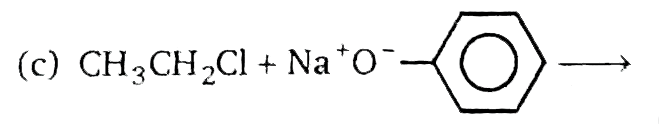Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What are Freons? Discuss their uses and environmental effects. |
|
Answer» Solution :Freons are the chlorofluoro derivatives of methane and ethane. Freon is represented as Freon - CBA Where, c = number of carbon atoms, b = number of hydrogen atoms, a = total number of fluorine atoms. `CF_2Cl_2`c = 1-1 = 0 `H = 0 + 1 = 1` `F = 2` So freon - 12 is `CF_2Cl_2` Uses of Freons (i) Freons are used as refrigerants in refrigerators and air conditioners. (ii) It is used as a propellant for aerosols and foams. (iii) Itis used as propellant for foams to spray out deodorants, shaving creams and insecticides. ENVIRONMENTAL effects of Freons (i) Freon gas is a very powerful greenhouse gas which means that it traps the heat normally radiated from the earth out into the space. This causes the earth.s temperature to increase, resulting in rising sea levels, droughts, stronger storms, flash floods and a host of other very unpleasant effect. (ii) As freon moves throughout the air, its chemical ingredients causes depletion of ozone layer. Depletion of ozone increases the amount of ultraviolet radiations that reaches the earths surface, resulting in SERIOUS risk to human HEALTH. High levels of ozone, in turn, causes respiratory problems and can also kill plants. |
|
| 2. |
Which of the following pairs show diagonal relationship ? |
| Answer» ANSWER :B | |
| 3. |
What is the oxidation number of gold in the complex [AuCl_4]^- ? |
| Answer» ANSWER : | |
| 6. |
What quantity of copper (II) oxide will 2.80 litres of hydrogen at N.T.P. ? |
|
Answer» `underset(underset("79.5 G 22.4 L at N.T.P")("1 mol1mol"))(CUO+H_(2))rarrCu+H_(2)O` 22.4 L of `H_(2)` REACT with CuO at N.T.P = 79.5 g 2.8 L of `H_(2)` react with CuO at N.T.P `= ((79.5g))/((22.4L))XX(2.8L)=9.94g`. |
|
| 7. |
Which of the following molecules involve intramolecular H-bond? |
|
Answer» Ortho-nitrophenol |
|
| 8. |
Which of the following statements is true ? |
|
Answer» `HNO_3` is a STRONGER acid than ` HNO_2` |
|
| 9. |
Write equation [H^+] and p^(Ka) of acid buffer. |
|
Answer» SOLUTION :`[H^+]=K_a "[weak ACID]"/"[CONJUGATE base]"` `pK_a=pH-log(["conjugate base" , A^-])/"[weak acid , HA]"` |
|
| 10. |
The strucutre of H_2O_2 is |
|
Answer» PLANAR |
|
| 11. |
Write the principle involved in the estimation of carbon and hydrogen.Give diagram and calculation. |
|
Answer» Solution :Principle: A known mass of an organic compound is burnt in presence of excess of oxygen and CUPRIC oxide. CARBON and Hydrogen. In the compound are converted into `CO_(2)` and `H_(2)O`. The masses of `CO_(2)` and `H_(2)O` PRODUCED is DETERMINED from which the percentage of carbon and Hydrogen are calculated. Calculation: % age of Carbon `=(12xxM_(2)xx100)/(44xxM)`, % age of Hydroen `=(2xxM_(1)xx100)/(18xxM)` 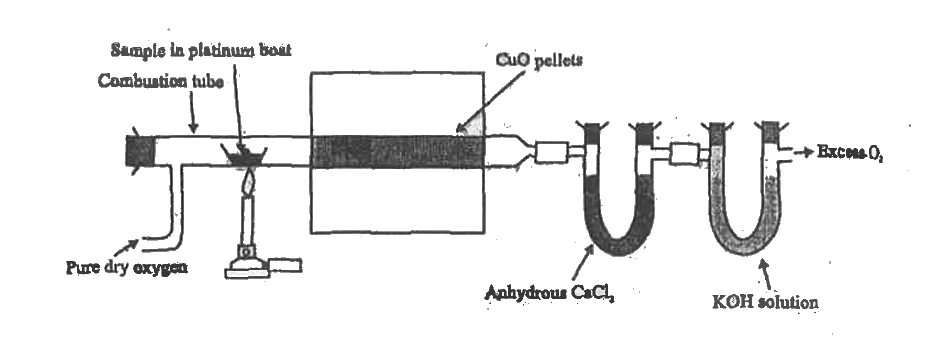
|
|
| 12. |
Why are potassium and caesium, rather than lithium used in photoelectric cells? |
| Answer» Solution :COMPARED to LI, K and cs have lower ionisation energy which makes the release of VALENCE ELECTRONS EASIER in K and Cs. | |
| 13. |
What is the correct strctural formula of borax (Na_(2)B_(4)O_(7).10H_(2)O) |
| Answer» SOLUTION :`Na_(2) [B_(4)O_(5)(OH)_(4)].8H_(2)O`. | |
| 14. |
Which of the following acids on heating loses a molecule of H_(2)O to form an alpha, beta- unsaturated acid ? |
|
Answer» `CH_(3)CHOHCOOH` |
|
| 15. |
Which is correct product ? |
|
Answer»

|
|
| 16. |
Which one of the following is an aromatic compound ? |
|
Answer» Cyclopentadienyl cation |
|
| 17. |
Which of the following observations was not correct during Rutherford's scattering experiment? |
|
Answer» Most of the `alpha`-PARTICLES passed through the gold foil undeflected. |
|
| 18. |
Two sample of air 1 cm^(3) each are take sample. A is kept at temperature T_(0) at sea level and another sample (B) at a height where pressure is 1//3 atm. The absoulte temperature of (B) at that height is |
|
Answer» `T_(0)//3` |
|
| 19. |
The vividhBharati stationof All indiaRadioDelhi. Broadcastson a frequency of 1.368kHz(kilohertz)Calculatethe wavelengthofthe elctromagneticradiationemittedbytransmitter .Whichpartof theelectromagneticspectrum does itbelong to ? |
|
Answer» Solution :`C= v lambda` `gamma= (c )/( v )` `= 2193m` WHEREC= speedof electron magneticradiation `=-3.0 xx 1 0^(8)ms^(-1)` v= FREQUENCY= 1368kH z =1368 `xx 10^(3) HZ` `1368 xx 10^(3)s^(-1)` this is acharacteristicradiowavewavelength. |
|
| 20. |
Using given information in question provided calculate equilibrium constant of requried reaction. Calculate equilibrium constant of required reaction. A(g)+2B(g) hArr4C(g)""K_(P_(1))=X C(g) hArrD(g)""K_(P_(2))=Y Value of K_(p) for reaction (1)/2A(g)+B(g)hArr2D(g) |
|
Answer» `sqrtYxxX^2` |
|
| 21. |
What is alumino silicate ?Which ions are used to balance its electric charge ? |
|
Answer» Solution :If aluminium ATOMS replace few silicon atoms in three-dimensional network of silicon dioxide , overall structure known as aluminosilicate ACQUIRES a negative charge. Cations such as `Na^(+) , K^(+) , Ca^(+2)` are USED to BALANCE the electric charge. |
|
| 22. |
Write short notes on Deuterium |
| Answer» Solution :`1H_2` or D. It occurs NATURALLY in very small traces. It's nucleus consists of a PROTON, a neutron and ONE electron revolving around the nucleus. Its chemical properties are similar to thoseof protium but their REACTION rates are different. | |
| 23. |
Which element do you think would have been named by (i) Lawrence Berkeley Laboratory (ii) Seaborg's group? |
|
Answer» Solution :LAWRENCIUM (LR) Z = 103 SEABORGIUM (Sg) Z = 106 |
|
| 24. |
What happens when: a. Whater is added to CaC_(2) and the resulting gas is passed through dil H_(2)SO_(4) containing HgSO_(4). B. Hydrated MgCl_(2) is heated in presence of NH_(4)Cl. C. FeCl_(3) solution is treated with Mg. D. NH_(4)Cl is heated with Mg.e. CO_(2) is passed through limewater. f. SO_(2) is passed through limewater. |
Answer» Solution :`CaC_(2)+H_(2)OrarrCa(OH)_(2)+underset("Acetylene gas")(C_(2)H_(2)uarr)` 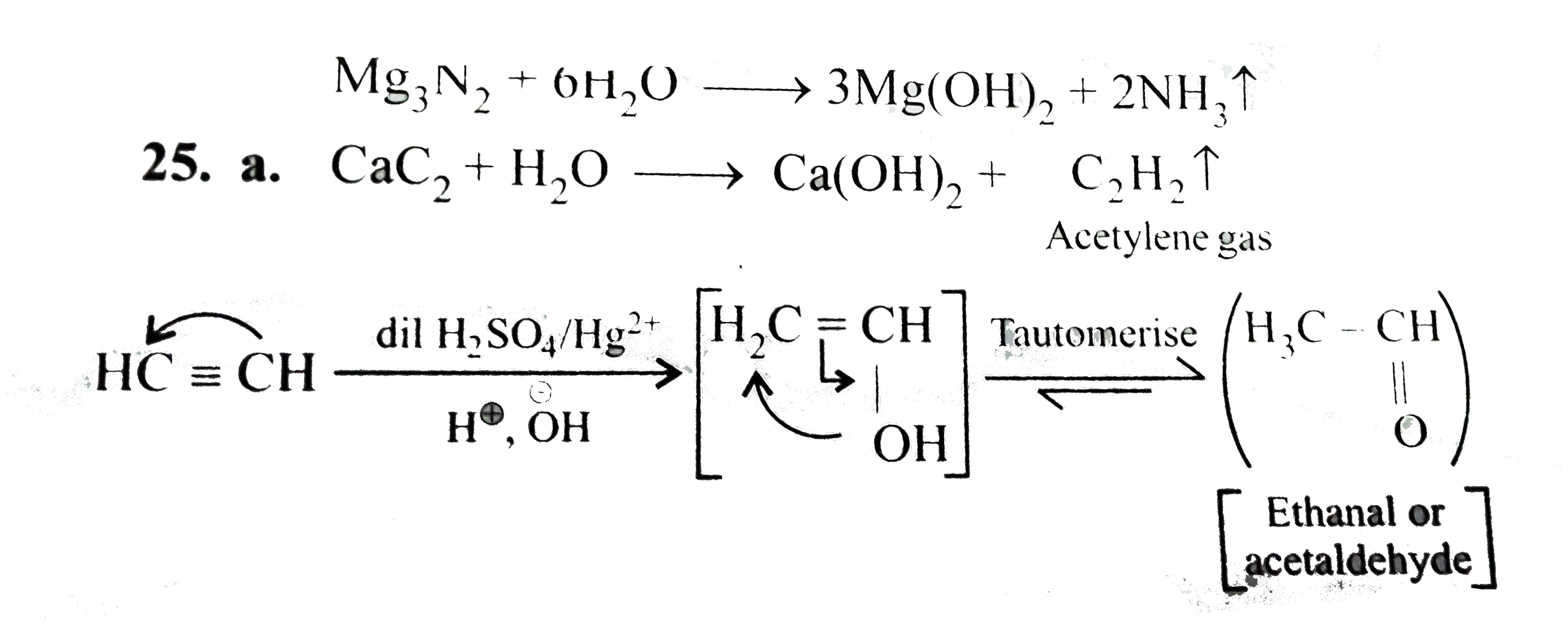 `CaC_(2)` reacts with `H_(2)O` to produce `CA(OH)_(2)` and acetylene, `C_(2)H_(2)` gas, when `C_(2)H_(2)` gas is further passed through `H_(2)SO_(4)` containing `HgSO_(4)`, acetaldehyde `(CH_(3)CHO)` is produced. (b). `MgCl_(2).6H_(2)O+NH_(4)ClrarrMgCl_(2).underset("Double salt")(NH_(4)Cl.6H_(2)O` `MgCl_(2).NH_(4)Cl.6H_(2)Ooverset(Delta)(rarr)MgCl_(2).NH_(4)Cl+6H_(2)O` `MgCl_(2).NH_(4)Cloverset(Delta)(rarr)MgCl_(2)+NH_(4)Cl` Hence, anhydrous `MgCl_(2)` is produced. (c). `FeCl_(3)+3H_(2)OrarrFe(OH)_(3)+3HCl` `Mg+2HClrarrMgCl_(2)+H_(2)` (d). `Mg+2NH_(4)ClrarrMgCl_(2)+2NH_(3)+H_(2)` (e). When `CO_(2)` is passed through limewater initially the solution becomes milky due to formation of `CaCO_(3)`. When excess of `CO_(2)` is passed, the solution becomes clear again due to the formation of `Ca(HCO_(3))_(2)` `underset("Limewater")(Ca(OH)_(2))+CO_(2)rarrunderset(("Milkiness"))(CaCO_(3))+H_(2)O` `CaCO_(3)+underset(("excess"))(CO_(2))+H_(2)Orarrunderset(("Soluble, hence the milkiness disappears and the solution becomes clear"))(Ca(HCO_(3))_(2))` (F). `Ca(OH)_(2)+SO_(2)rarrCaSO_(3)+H_(2)O` Solution becomes milky due to formation of `CaSO_(3)` |
|
| 25. |
Which of the following will not show deflection from the path on passing through an electric field ? Proton, C athode rays, E lectron, N eutron |
| Answer» SOLUTION :N eutron is neutral so, no deflection from the p ATH on passing through an electric FIELD. PROTON, CATHODE rays an d electron being the charged particle will show deflection from the p ath on passing through an electric field. | |
| 26. |
When a metal is to be extracted from its ore and if the gangue associated with the ore is silica, then |
|
Answer» An ACIDIC FLUX is NEEDED |
|
| 27. |
Write prefix and suffix in the following functional group? (i) -COCl (ii) -CONH_(2) (iii) -COOR (iv) -underset(underset(H)(|))(C )=O (v) -underset(underset(OH)(|))(C )= O (vi) -underset(underset(X)(|))(C )=O (vii) - underset(|)(C )= O (viii) -C -= N (ix) -underset(underset(O)(||))(C )- O^(-) |
Answer» SOLUTION :
|
|
| 28. |
Which of the following statements about inorganic naphthalene is false? |
|
Answer» INORGANIC naphthalene is `B_(5)N_(5)H_(8)` |
|
| 29. |
Which of the following group has the maximum hyperconjugation effect when attached to bezene ring ? |
|
Answer» |
|
| 30. |
What will be obtained by reaction of calcium carbide with heavy water ? Give its chemical reaction. |
|
Answer» Solution :Product : `Ca(OD)_2` and `C_2D_2` REACTION : `CaC_2+ 2D_2O to Ca(OD)_2 + C_2D_2` |
|
| 31. |
What happens when Sodium to from sodium peroxide. |
|
Answer» Solution :Sodium burns in AIR or OXYGEN to FORM sodium peroxide. `2Na+O_(2)toNa_(2)O_(2)` |
|
| 32. |
What is the physical significance of Psi and Psi^(2) ? |
| Answer» Solution :`PSI` as such has no PHYSICAL SIGNIFICANCE. `Psi^(2)` gives the probability of FINDING the electron at any POINT around the nucleus. | |
| 33. |
Which acid is formed by reaction of rain water with CO_2 ? |
|
Answer» `H_2 CO_3` |
|
| 34. |
Why alkaline earth metals are harder than alkali metals. |
|
Answer» Solution :(i) Atomic RADIUS of alkaline earth metals are small and their densities are larger than those of alkali metals. (ii) Alklaine earth metals have close packed crystal STRUCTURE (iii) GENERALLY alkaline earth metals are soft yet less than that of alkali metals. This is because the METALLIC bonding in alkaline earth metals are stronger than alkali metals. |
|
| 35. |
Which one of the following electrolysis is used in Down's process of extracting sodium mealt ? |
|
Answer» `NACI+KCI+KF` |
|
| 36. |
Which of the following represents the given mode of hybridization sp^(2)-sp^(2)-sp-sp from left to right ? |
|
Answer» `H_(2)C=CH-C-=N` |
|
| 37. |
What is position of element contain n = 3 and electronic configuration is ns^(2)np^(4)in periodic table ? |
|
Answer» Solution :`N =3`, third period , `3s^(2)3P^(4)` ` = 10 + ` VALENCE electron ` = 10 + 2 + 4 = 16^(th)` group. Last electron FILLED in p-orbital so it belong in p-block. |
|
| 38. |
The tendency to form p_(pi) - p_(pi) multiple bonds in group 14 is most prominent for |
|
Answer» C |
|
| 39. |
What volume of air containing 21% oxygen (by volume) is required to completely burn 1 kg of carbon containing 100% combustible substances? |
|
Answer» |
|
| 40. |
Write the expression for solubility product of calcium phosphate in terms of its molar solubility, S. |
| Answer» SOLUTION :`{:(Ca_(3)(PO_(4))_(2) ,hArr,3 CA^(2+),+,2PO_(4)^(3-),,),(S,,3S,,2S,,):} K_(sp)=(3S)^(2)(2S)^(3)=72S^(5)` | |
| 41. |
Three equivalents of aluminiumchloride on reacting withpredominantly gives |
|
Answer»
|
|
| 42. |
Which element in 3d, 4d, and 5d transition series has the highest paramagnetism in (a) elemental form, (b) +1 O.S. ( c) +2 O.S., (d) +3 O.S. [O.S. ="oxidation state"] |
Answer» Solution :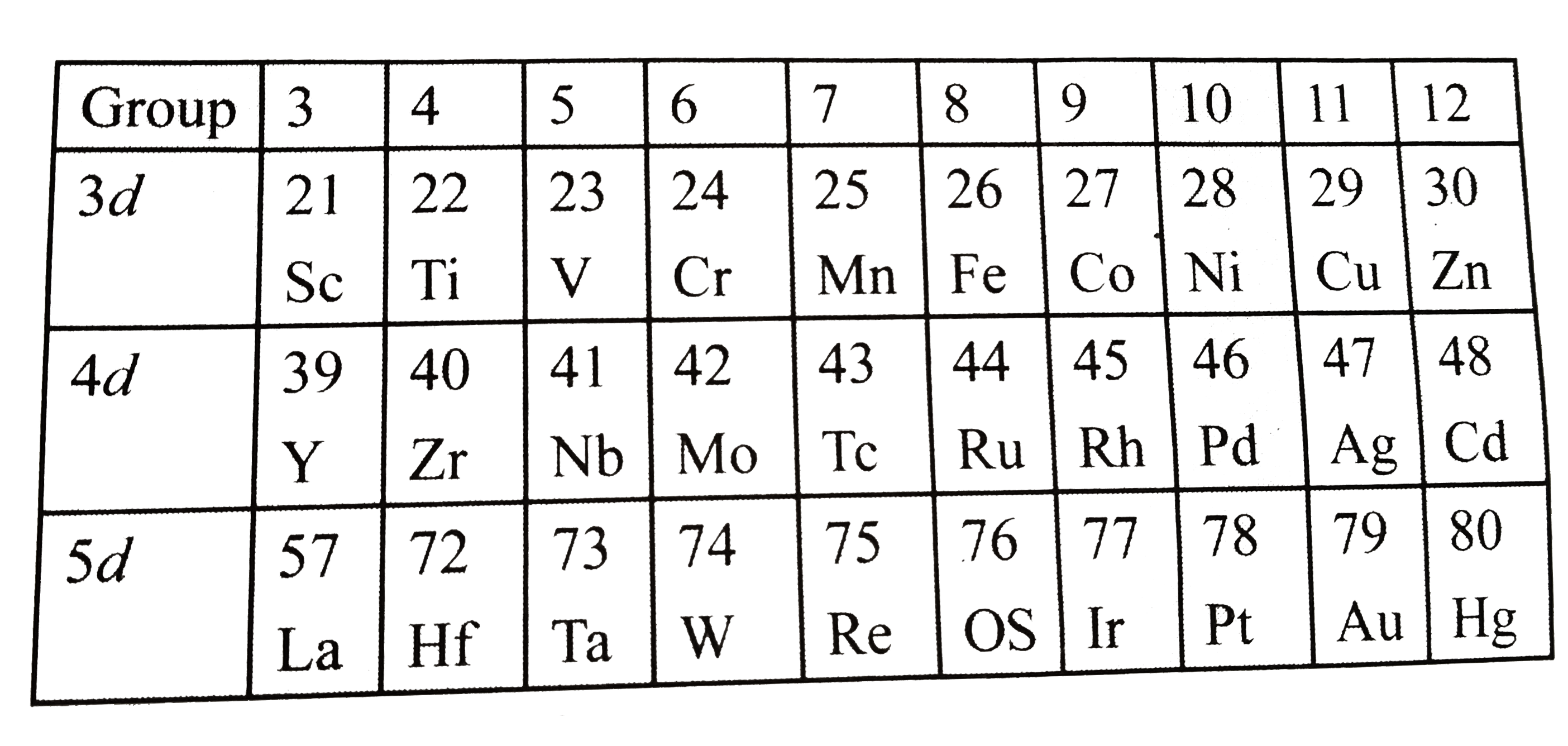 (a) In elemental form: `3d` series: `Cr (Z = 24) RARR 3d^(5) 4S^(1),n = 6, mu = sqrt(6+(6+2))` `= sqrt(48) BM` `4d` series: `Mo(Z=42) rArr 4d^(5) 5s^(1),n=6, mu = sqrt(48) BM` `5d` series: `W(Z=74) rArr 5d^(4) 4s^(2),n=4, mu = sqrt(24) BM` But `Re` has the highest number of unpaired `e^(-)`, as shown: `Re(Z=75) rArr 5d^(5) 6s^(2),n = 5, sqrt(35) BM` (b) `In + 1` oxidation state: `3d` series: `Mn(Z=25) rArr3d^(5) 4s^(2), Mn^(1+) = 3d^(5) 4s^(1), n=6, mu = sqrt(48) BM` `4d` series: `Te(Z = 43) rArr 4d^(5) 5s^(2), Te^(1+) = 4d^(5) 5s^(1), n=6, mu = sqrt(48) BM` `5d` series: `Re(Z=75) rArr 5d^(5) 6s^(2),Re^(1+)=5d^(5) 6s^(-),n=6, mu=sqrt(48) BM` (c ) `In+2` oxidation state: `3d` series: `Mn(Z=25) rArr 3d^(5) 4s^(2), Mn^(2+) = 3d^(5), 4s^(0),n=5, mu = sqrt(35) BM` `4d` series: `Te(Z=43) rArr 4d^(5) 6s^(2), Te^(2+)=4d^(5) 5s^(0),n=5, mu = sqrt(35) BM` `5d` series: `Re(z=75) rArr 5d^(5) 6s^(2), Re^(2+)=5d^(5) 6s^(0),n=5, mu = sqrt(35) BM` (d) `In+3` oxidation state: `3d` series: `Fe(Z=26) rArr 3d^(6) 4s^(2), Fe^(3+)=3d^(5) 4s^(0),n=5, mu = sqrt(35) BM` `4d` series: `Ru(Z=44) rArr 4d^(7) 5s^(1)` (exxeption electronic configuration). `5d` series: `Os (Z=76) rArr 5d^(6) 7s^(2), Os^(3+) = 5d^(5) 7s^(0), n=5, mu = sqrt(35) BM` Note: (i) From the abov calculation it is clear that in the elemental form (zero `OS`), group `6` has the highest `mu`, except `W`, in `5d` series, but it is `Re` of group `7`. (ii) `In+1 OPS`, group `7` has the highest `mu`. (iii) `In+2 OS`, again group `7` has the highest `mu`. (iv) `In+3 OS`, group `8` has the highest `mu`. |
|
| 43. |
The volume (in ml) of 0.10 M AgNO_(3) required for complete precipitation of chlorine ions present in 30 ml of 0.01 M solution of [Cr(H_(2)O)_(5)Cl]Cl_(2) as silver chlorine is close to __________ |
|
Answer» `[Cr(H_(2)O)_(5)Cl]Cl_(2)=(0.01xx30)/(1000)=3xx10^(-4)` moles `underset(?)underset(2)(AgNO_(3))+underset(3xx10^(-4))underset(1)([Cr(H_(2)O)_(5)Cl])Cl_(2)rarr2AgCl` `IMPLIES 6xx10^(-4)=0.1xxV` `V=6xx10^(-3)"lit "implies V=6ml` |
|
| 44. |
What is dipole moment in molecule ? Write about dipole moment of two atom containing molecule. |
|
Answer» Solution :DIPOLE MOMENT of molecule is equal to the resultant dipole moment of all the individual hond. The magnitude of this resultant dipole moment not only depends upon the values of the individual dipole moment of the bonds but ALSO on that arrangement in space. Dipole moment of dJatomic molecules : All diatomic molecules are linear. It has two types. (i) Homonuclear atom `A_(2)` (ii) Heteronuclear AB  (iv)As the difference of electronegative BENT two atoms increases ionic character increases and covalent prop decreases. Define of EN decrease polarity & `mu` also DECREES and covalent character increases. 
|
|
| 45. |
Which of the following reaction will not give ether as a major product ? |
|
Answer» `CH_(3)CH_(2)Cl+Ag_(2)O(dry)to` |
|
| 46. |
What are spontaneous reactions ? What are the conditions for the spontaneity of a process ? |
|
Answer» Solution :Charles law states that " At contant pressure, the VOLUME of a given mass of an ideal gas is directly proportional to its temperature". According to Chearles Law , if were to take a balloon filled with air and increase the temperature of the air INSIDE, the volume of air would increase causing the balloon to expand. This is caused by the heating of the molecular of air inside the balloon causing them to move repidly . In the same manner if we COOLED the balloon in a freezer , the volume of air decrease, MAKING the balloon look PARTIALLY deflated. |
|
| 47. |
When H_2 and I_2 are mixed and equilibrium is attained, then |
|
Answer» AMOUNT of HI formed is equal to the amount of `H_2` dissociated |
|
| 48. |
What is the co-ordination number of Cl^(-)in NaCl crystal ? |
|
Answer» 8 `therefore` Co-ordination NUMBEROF `Cl^(-)` `=Co-ordination number of `Na^(+)`=6 |
|
| 49. |
Which of the following molecule is optically active |
|
Answer»

|
|
| 50. |
Which of the following statements are true about metals ? |
|
Answer» VALENCE band overlaps with CONDUCTION band. |
|