Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What is Average rotational Kinetic energy ? |
|
Answer» Solution :`(1)/(2)m U^(-2)=` Average ROTATIONAL KINETIC energy. Where `u^(2)=(u_(1)^(2)+u_(2)^(2)+…..u_(n)^(2))/(n)`. |
|
| 2. |
Write the reactions of B_(2)H_(6) with (a)H_(2)O (b)O_(2) (c) NH_(3). |
| Answer» SOLUTION :For ANSWER, CONSULT SECTION 11.4. | |
| 3. |
The ultraviolet radiation has frequency 6xx10^(16)//s The wavelength would be |
|
Answer» `2XX10^(6)` cm |
|
| 4. |
Which of the following is true for London smog ? |
|
Answer» It occurs during WARM weather |
|
| 5. |
Write IUPAC names of the following compounds. |
Answer» SOLUTION :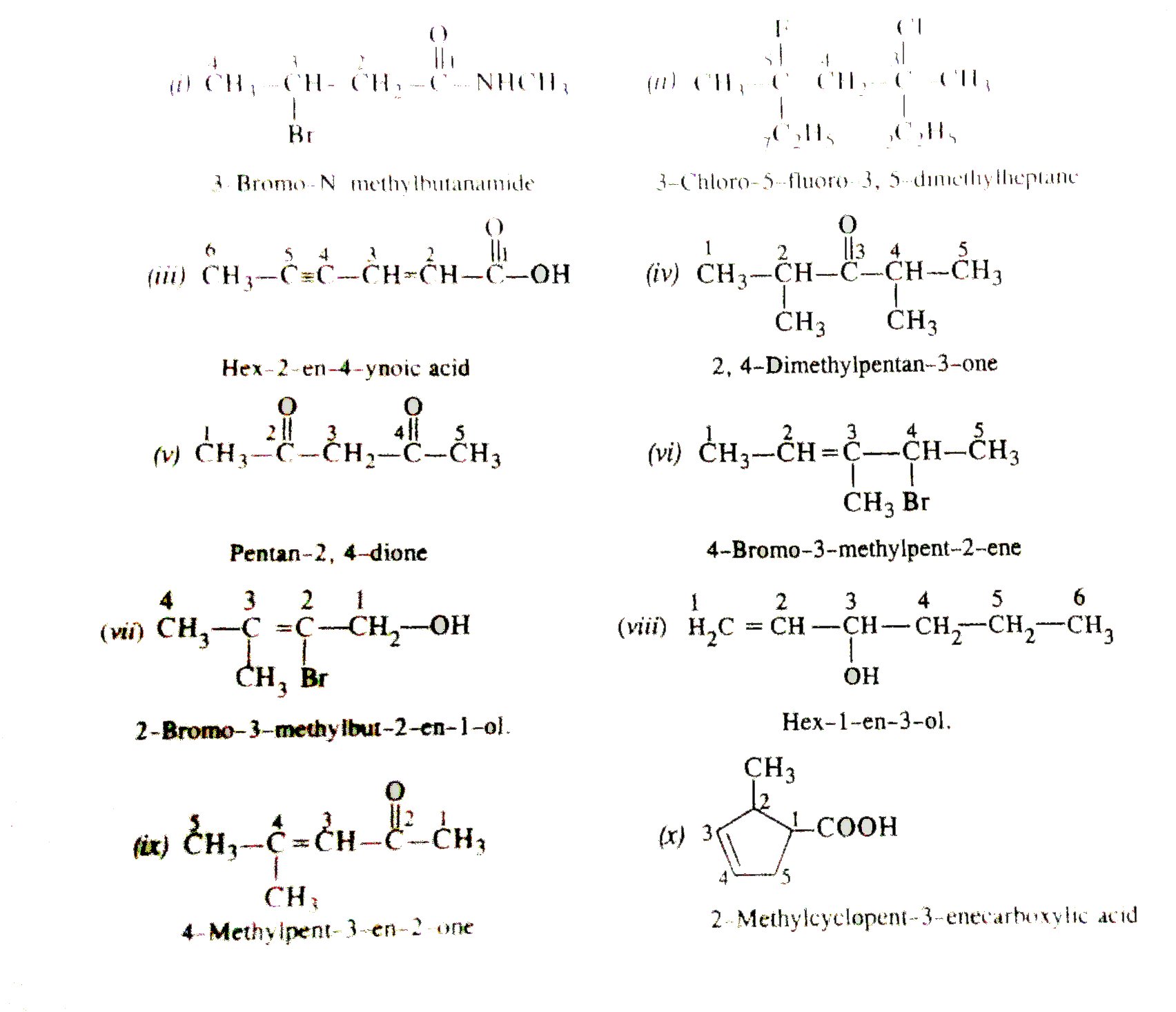
|
|
| 6. |
When 100ml of 0.06M Fe(NO_3)_3, 50ml of 0.2M FeCl_3 and 100ml of 0.26M Mg(NO_3)_2, are mixed, the concentration of NO_3^(-) ions in the final solution is |
|
Answer» 0.028M |
|
| 7. |
What is mean by activator groups ? Explain with suitable examples. |
|
Answer» Solution :Definition : After moon substitution of BENZENE it ENTERS into the benzene ring which promotes electrophilic reaction which give easy reaction in comparision to benzene ring, such group is known as activator groups. Examples : -OH, -OR, `-NH_(2), -NHR, -NHCOR`, etc., groups are ortho para directing activator groups. While `-Cl, -Br` are ortho directing in nature but they are not activator groups. Explanation : Activators groups donate their electrons towards RINGS, i.e., they donate electrons density benzene ring. Therefoe electron density in activator ring is more as compared to benzene. This is the reasons that electrophilic reactions are easily occurs in compounds with such activator groups. Such groups donates their electron pair into the ring negative resonance structure.  Such groups due to its own (+I) effect, donates electrons towards ring and increases negative density, increases ACTIVITY. e.g., R groups  Due to resonance and inductive effect of ortho and para groups they are activator in rings. While -Cl and -Br have medium activator effect. |
|
| 8. |
Whichof thefollowingcompoundis usedtoscanthe X - rayof thehumandigestivesystem? |
|
Answer» `BACL _(2)` |
|
| 9. |
What do you understand by the terms: (a) hydrogen economy reaction, (b) hydrogenation, ( c) 'syngas', (d) water gets shift reaction and (e) fuel cell? |
|
Answer» Solution :a. Hydrogen economy b. Hydrogenation c. SYNGAS d. Water-gas shift reaction e. Fuel CELL: A device which converts the energy produced during the combustion of a fuel directly into ELECTRICAL energy is known as fuel cell. Dihydrogen is used in hydrogen-oxygen fuel cells for generating electrical energy. Fuel cell has many advantages over the CONVENTIONAL fossil fuels. It does not cause any pollution and releases more amount of energy per unit MASS of fuel as compared to gasoline and other fuels. |
|
| 10. |
Using van der Waals equation calculate the constant 'a' when twomolesof a gasconfined in a four litre flask exerts a pressure of 11.0 atmosphere at a temperature of 300 K. The value of 'b' is 0.05 lit mol^(-1) |
|
Answer» Solution :`(P+(an^(2))/(V^(2)))(v-nb)=nRT` or`a=((nRT)/(V-nb)-P)(V^(2))/(n^(2))=((2molxx0.08205" L ATM K"^(-1)" mol"^(-1)xx300" K")/(4 L-2 mol xx0.05" L mol"^(-1))-11" atm")XX((4 L)^(2))/((2 mol)^(2))` |
|
| 11. |
Which one of the following statements is not true about the effect of an increase in temperature on the distribution of molecular speeds in a gas? |
|
Answer» The AREA under the distribution curve REMAINS the same as under the lower temperature. 
|
|
| 12. |
The stability of +1oxidation state among Al, Ga In and Tl increases in the sequence. |
|
Answer» `AL lt Ga lt In lt Tl` |
|
| 13. |
Which out of the following can be determined ? Absolute internal energy, absolute enthalpy, absolute entropy. |
| Answer» SOLUTION :The ENTROPY possessed by a SUBSTANCE at absolute ZERO is called residual entropy. | |
| 14. |
The rms velocity of hydrogen is sqrt(7) times the rms velocity of nitrogen. If T is the temperature of the gas, then: |
|
Answer» `T_((H_(2)))=T_((N_(2)))` |
|
| 15. |
Which of the following metal is present in 100 miligram / liter concentration in plasma ? |
| Answer» ANSWER :D | |
| 16. |
Two dimensional close packed structure can be generated by stacking the rows of close packed spheres. This can be done in two different ways. (I) The second row may be placed in contact with the first one, such that the spheres of the second row are exactly above those of first row. The spheres of the two rows are aligned vertically as well as horizontally. If we call the first row as 'A' type of row, the second row being exactly same as the first one is also of 'A' type. Similarly, we may place more rows to obtain AAAA.... type arrangement (II) In this type, the second row may be placed above the first one in a staggered manner such that its spheres. fit in depressions of first row. if the arrangement of spheres in the first row is called 'A' type the one in the second row is different and may be called 'B' type. When the third row is placed adjacent to the second in staggered manner, its spheres are aligned with those of first layer. Hence this layer is also 'A' type. The spheres of similarly placed fourth row will be aligned with those of the second row ('B' type). Hence this arrangement is of ABAB.... type Of type (I) & type (II), the packing efficiency |
|
Answer» HEXAGONAL CLOSE packing |
|
| 17. |
The total number of isomers for the compounds of the formula C_4H_10Oare |
|
Answer» 7 |
|
| 18. |
Two dimensional close packed structure can be generated by stacking the rows of close packed spheres. This can be done in two different ways. (I) The second row may be placed in contact with the first one, such that the spheres of the second row are exactly above those of first row. The spheres of the two rows are aligned vertically as well as horizontally. If we call the first row as 'A' type of row, the second row being exactly same as the first one is also of 'A' type. Similarly, we may place more rows to obtain AAAA.... type arrangement (II) In this type, the second row may be placed above the first one in a staggered manner such that its spheres. fit in depressions of first row. if the arrangement of spheres in the first row is called 'A' type the one in the second row is different and may be called 'B' type. When the third row is placed adjacent to the second in staggered manner, its spheres are aligned with those of first layer. Hence this layer is also 'A' type. The spheres of similarly placed fourth row will be aligned with those of the second row ('B' type). Hence this arrangement is of ABAB.... type |
|
Answer» is BETTER in TYPE I |
|
| 19. |
Write the difference between chemical oxygen demand (COD) and biochemical oxygen demand (BOD). |
Answer» SOLUTION :
|
|
| 20. |
The strength of H_2O_2sample available form market is 8.32% W/V. The volume and molar concentration of this sample are _____and _____respectively |
| Answer» SOLUTION :27.4 , 2.447 | |
| 22. |
What is the difference among complete combustion, incomplete combustion and combustion of alkane ? |
Answer» SOLUTION :
|
|
| 23. |
Which of the following species is expected have maximum ethalpy in an electrophilic aromatic substitution reaction. |
|
Answer» SPECIES (II) |
|
| 24. |
What is the value of change in entropy at equilibrium? |
| Answer» Solution :At EQUILIBRIUM, the CHANGE in entropy is zero. i.e., `DeltaS = 0`(at equilibrium) | |
| 25. |
The unit cell of a compound made up of the three elements X, Y and Z is given below. The formula of this compound is |
|
Answer» `X_(2)YZ_(3)` |
|
| 26. |
Which of the following will show optical isomerism? |
|
Answer»

|
|
| 27. |
What are the main chemical constituents of cement? |
| Answer» SOLUTION :`3CaO . SiO_2, 2CaO . SiO_2 and 3CaO . Al_2O_3`. | |
| 28. |
The set representing the correct order of ionic radii is ..... |
|
Answer» `LI^(+) gt Be^(2+) gt NA^(+) gt Mg^(2+)` Down the GROUP ionic radii increases with INCREASING atomic number because of the increase in the number of atomic shells but across the period the ionic radii DECREASE due to increase in effective nuclear charge as electrons are added in the same shell.`Li^(+) " and " Mg^(2+)`are diagonally related, so correct order is `Na^(+) gt Li gt Mg^(2+) gt Be^(2+)` ` Be^(2+) = 0.31Å, Mg^(2+) = 0.72 Å, Li^(+) = 0.76 Å, Na^(+) = 0.95 Å` |
|
| 29. |
Which of the following compounds exhibits geometrical isomerism ? |
|
Answer» `C_2H_5Br` |
|
| 30. |
What is common between d_(xy) and d_(x^2 - y^2) orbitals? |
| Answer» Solution :Both have IDENTICAL SHAPE, CONSISTING of FOUR lobes. | |
| 31. |
What are the different types of hydrides? |
|
Answer» Solution :The hydrides are CLASSIFIED as ionic , Covalent and METALLIC Hydrides. Ionic HYDRIDE - LiH • Covalent hydride -- CH • Metallic hydride - TIH |
|
| 32. |
Which of the following alkanes will give single product on methylene insertion? A) CH_3-CH_3 B) CH_3-CH-2-CH-3 C) (CH_3)_4C D)CH_3CH_2C (CH_3)_2CH_2CH_3 E)(CH_3)_2CHCH(CH_3)_2 |
| Answer» SOLUTION :C - Hinsertions | |
| 34. |
Write the mathematical formula for change in entropy. |
| Answer» SOLUTION :`DELTAS= (q_("REV") )/( T)` | |
| 35. |
Which of the following is least soluble in water |
|
Answer» `CaF_(2)` |
|
| 36. |
Which of the following sets of groups contains only electrophiles? |
|
Answer» `NH_(2)^(-),NO_(2)^(+),H_(2)O,NH_(3)` |
|
| 37. |
Which of the following compounds have delocalized electrons? (P) CH_(2)=Choverset(O)overset(||)"C"CH_(3) (Q) (R) (S) CH_(3)CH_(2)NHCH=CHCH_(3) |
|
Answer» <P> |
|
| 38. |
Which of the following is truely covalent? |
|
Answer» AgCI |
|
| 39. |
Which of the following is not used in the conversion of para hydrogen into ortho hydrogen? |
|
Answer» by heating more than `800^@C` |
|
| 40. |
Which is not an example of acidic refractory ? |
|
Answer» SILICA |
|
| 41. |
Which one of the following is a molecular crystal ? |
|
Answer» ROCK salt |
|
| 42. |
Write product of the following reaction :- CaCO_3 + HCl to |
| Answer» | |
| 43. |
Write the resonance structure for SO_(3), NO_(2) and NO_(3)^(-) . |
Answer» Solution :Resonance structure of `SO_(3)` : Structure (IV) is hybrid. Total valence electron = 24In `SO_(3)` total electron= 8 bonding + 16 nonbonding = 24  NITROGEN dioxide `(NO)_(2)` molecule : (I) (II) resonance structure and (III) is hybrid structure. Total valence electron= Two O (12) + N of (5) = 17 Total electron in `NO_(2)` = 6 bonding + 11 non-bonding = 17 Nitrate Ion `(NO_(3)^(-)) `:  Total valence electron= N (5) +`O_(3)`(18) + 1 = 24 Total electron in `NO_(3)^(-)` = 8 FOUR BOND + 16 non-bonding= 24 |
|
| 44. |
Which one of the following compounds cannot show tautomerism ? |
|
Answer» `CH_(3)-underset(O)underset(||)(C )-CH_(3)` |
|
| 45. |
When BCl_3 is treated with water, it hydrolyses and forms [B(OH)_4]^(-)only whereas AlCl_3 in acidifiedaqueous solution forms [Al(H_2O)_6]^(3+) ion. Explain what is the hybridizations of boron and aluminium in these species ? |
|
Answer» Solution :In trivalent state , most of COMPOUNDS being cavlent are hydrolyzed in water . For example ,the trichorides on hydrolysis in water form tetrahedral `[M(OH)_4]` species , the hybridizationstate of element M is `sp^3` . `BCl+ 3H_2O to B(OH)_3 + 3HCl` `B(OH)_3 + H_2O to [B(OH)_4]^(-) + H^(+)` Aluminum chloride in ACIDIFIED aqueous solution forms OCTAHEDRAL `[AL(H_2O)_6]^(+3)`ion. In this complex ion, the 3d-orbitals of Al are involved and the hybridizations state of Al is `sp^3d^2`. `AlCl_3 + H_2O overset(HCl)to [Al(H_2O)_6]^(3+) + 3Cl^(-)` 
|
|
| 46. |
The third virial coefficient of He gas is 4xx10^(-2)(L//"mol")^(2), then what will be volume of 2 mole He gas at 1 atm and 273 K? |
|
Answer» 22.0L |
|
| 48. |
When N_(2) molecule accept electron and form N_(2)^(-) then added electron enter in ...... orbital. |
|
Answer» Antibonding `(sigma^(**)) `MOLECULAR orbital Now, `N_(2)` accept electron and FORM`N_(2)^(-)` Then total electron are 15. `N_(2)^(-) = (pi_(2p_(x))^(**) )^(1)`because the next molecular orbital after `sigma_(2p_(z)) ` is `pi_(2p_(x))^(*) = pi_(2p_(y))^(**)`.Thus accepted electron `N_(2)`is filled in antiboncting molecular `pi_(2p_(x))^(**)` . |
|
| 49. |
Which of the following relationships is correct for a reaction involving both reactants and products both in either solid or liquid state? |
|
Answer» `Delta H GT Delta E` |
|
| 50. |
Write the complete chemical equation for the following . Nitration of benzene |
Answer» SOLUTION :
|
|









