Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
One mole of acetone requires less heat to vaporise than 1 mol of water. Which of the two liquids has higher enthalpy of vaporisation ? |
|
Answer» Solution :One mole of acetone REQUIRES less heat to VAPORISE than 1 mole of water. HENCE, acetone has less enthalpy of vaporisation and water has higher enthalpy of vaporisation. It can be represented as `( DELTA H_V) "water" gt (Delta H_V)` acetone. |
|
| 2. |
One mole of acetone requries less heat to vaporise than 1 mole of water. Which of the two liquids has higher enthalpy of vaporisation ? |
| Answer» Solution :LESS the heat required to VAPORISE 1 MOLE of a liquid , less is its ENTHALPY of vaporisation . Hence,water has higher enthalpy of vaporisation. | |
| 3. |
One mole of a symmetrical alkene on ozonolysis gives two of an aldehyde having a molecular mass of 44u. The alkene is : |
|
Answer» propane |
|
| 4. |
One mole of a symmetrical alkene on ozonolysis gives two moles of an aldehyde having a molecular mass of 44 u. The alkene is |
|
Answer» 2-butene |
|
| 5. |
One mole of a non-idealgas undergoes a change of state (1.0 atm,3.0 L,200 K) to (4.0 atm,5.0 L,250 K) with a change in internal energy (DeltaU)=40 L-atm. The change in enthalpy of the process in L-atm : |
|
Answer» 43 |
|
| 6. |
One mole of a non-ideal gas undergoes a change of state (2.0 atm, 3.0 L, 95 K)to(4.0 atm,5.0 L, 245 K) with a change in internal energy , DeltaU =30.0 L atm. The change in enthalapy (DeltaH) of the process in L atm is : |
|
Answer» 40 |
|
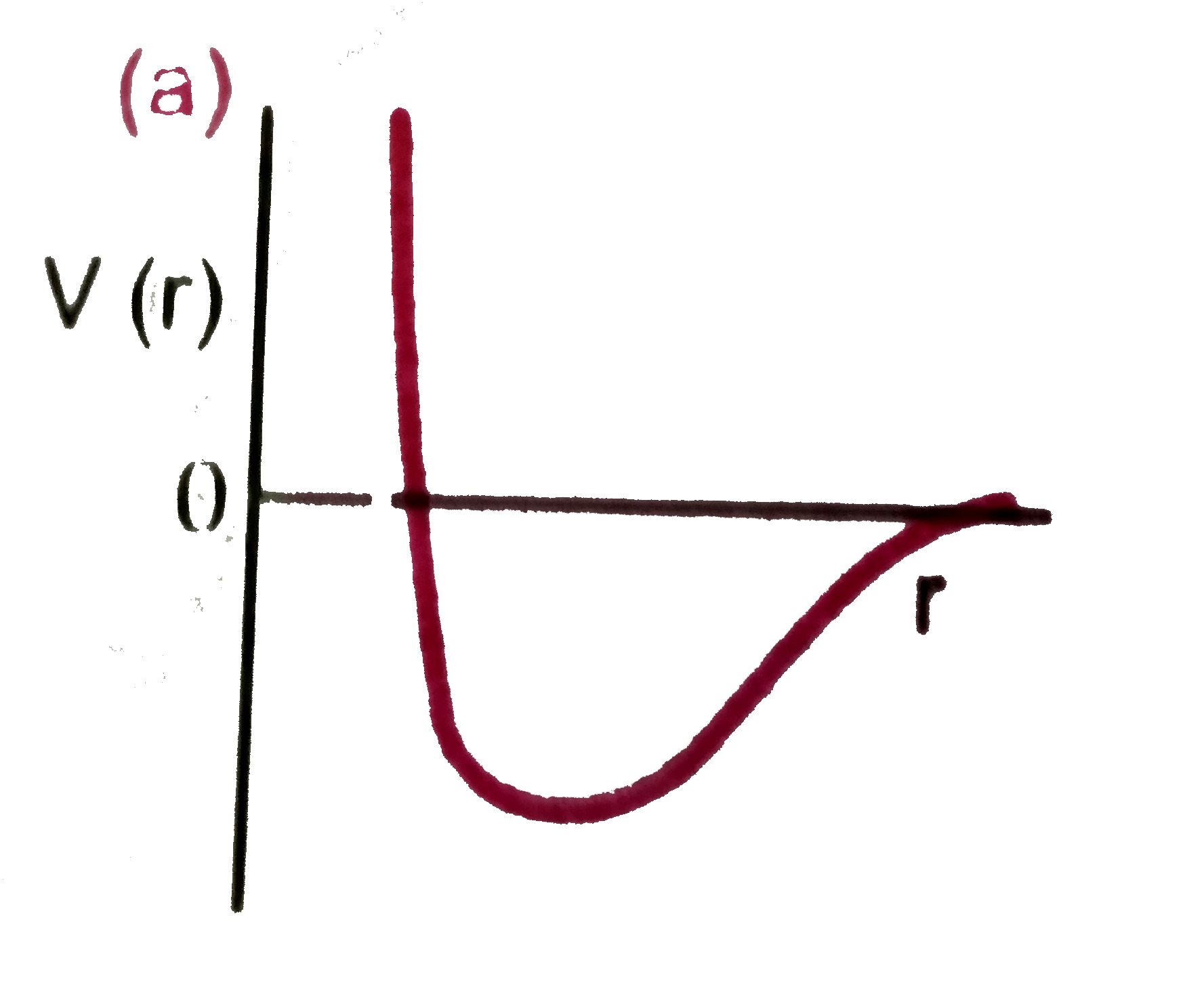
| 7. |
One mole of a monoatomic gas satisfies the equation P(V-b)=RT Where b is a constant. The relationship of interatomic potential V(r ) and interatomic distance r for the gas is given by |
|
Answer»
|
|
| 8. |
One mole of a mixture of CO and CO_(2) requires exactly 20g of NaOH is solution for complete conversion of alll the CO_(2) into Na_(2)CO_(3) . How many more grams of caustic soda would be required for the conversion of one mole of the totally oxidised form of given mixture. |
|
Answer» |
|
| 9. |
One mole of a mixture of CO and CO_(2) requires exactly 20 g of NaOH to convert all the CO_(2) " into " Na_(2)CO_(3). How many more grams of NaOH would it require for conversion into Na_(2)CO_(3) if the mixture (one mole) is completely oxidised to CO_(2) ? |
|
Answer» 60 g |
|
| 10. |
One mole of a liquid( 1 bar, 100 ml) is taken in an adiabatic container and the pressure increases steeply to 100 bar. Then at constant pressure of 100 bar, volume decrases by 1 ml.Find DeltaU andDeltaH. |
|
Answer» Solution :As the process is carriedout under adiabatic CONDITION, `q=0` . But `DeltaU = q + w`. Hence `Delta U =w`.Further, at constant pressure of100 bar, volume has decreased by1 ml, therefore, work of contraction . `= P Delta V = 100 ` bar ` xx 1 `ml. `= ( 100 xx10^(5) Nm^(-2)) ( 10^(-6)m^(3)) = 10J` Hence, `Delta U = 10 J` `Delta H =Delta U + Delta( PV)` `Delta ( PV) = (P_92)V_(2) - P_(1)V_(1))` But `P_(1)=1 "bar", V+(1) = 100 ml` `P_(2) = 100 "bar", V_(2) = 99 ml` `:. Delta ( PV) = ( 100 "bar" xx 99 ml) - ( 1 "bar" xx 100 ml) `= ( 9900- 100 ) "bar" ml = 9800 "bar ml ) = 980 J ` `:. Delta H = 10J + 980 J = 990 J` |
|
| 11. |
One mole of a hydrocarbon on ozonolysis yields one mole of glyoxal and two moles of formaldehyde. The hydrocarbon is: |
|
Answer» `CH_2 = underset(CH_3)underset(|)C-underset(CH_3)underset(|)C=CH_2` |
|
| 12. |
One mole of a gas changed from its initial state (15L,2 atm) to final state (4L,10 atm) reversibly. If this change can be represented by a straight line in P - V curve maximum temperature (approximate), the gas attained is x xx10^(2) K. Then find the value of x. |
|
Answer» `P-2=(10-2)/(4-15)(V-15)` `P-2=(8)/(11)(V-15)` `P=2-(8V)/(11)+(15xx8)/(11)` `P=((142)/(11)-(8V)/(11))` 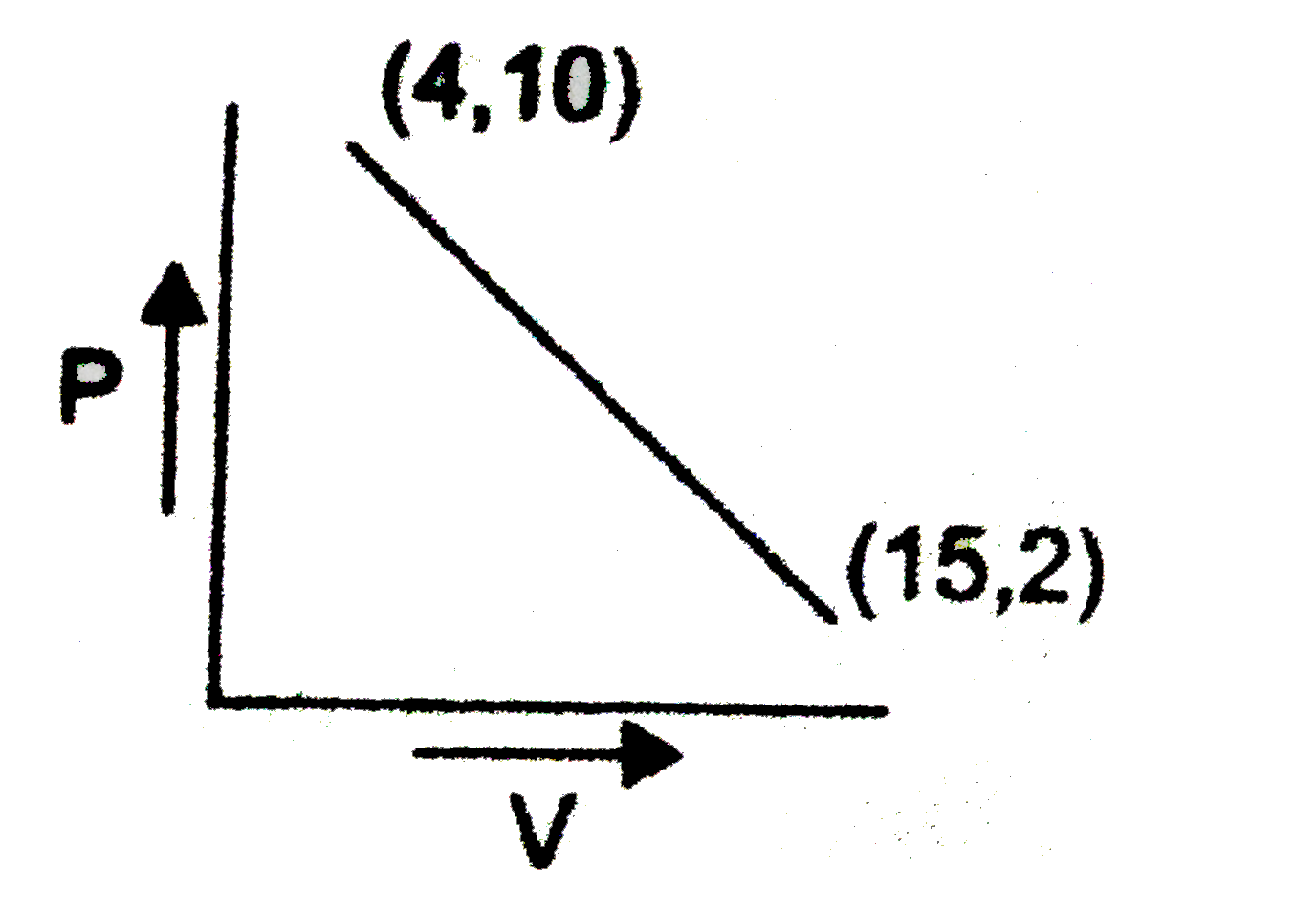 `f(T)=(1)/(nR)((142V)/(11)-(8V^(2))/(11))` `(d(F(T)))/(dV)=(1)/(nR)((142)/(11)-(8V^(2))/(11))=0` `V=(142)/(11xx16)=8.875` `P=(142)/(11)-(8)/(11)xx(8.875)/(16)=(71)/(11)` `T_("Max")=(PV)/(nR)=(71)/(11)xx(142)/(16xx0.0821)=700` `=7XX10^(2)K` |
|
| 13. |
One mole of 1,2-dibromopropane on treatment with X moles off NaNH_(2) followed by treatment with ethyl bromide gave a 2-pentyne. The value of X is |
|
Answer» one 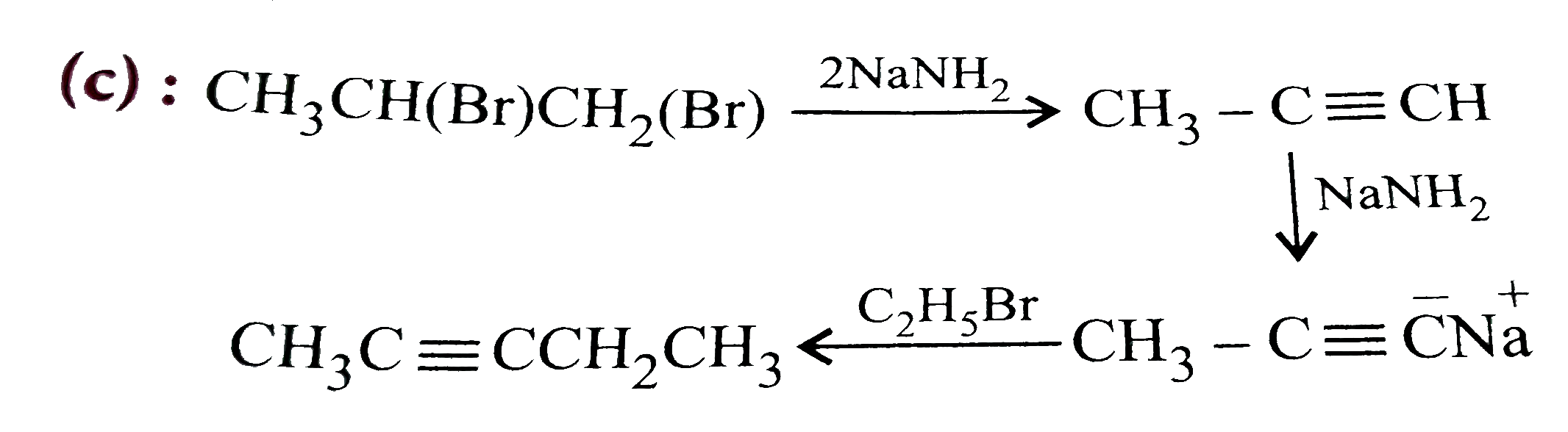 TOTAL NUMBER of MOLES of `NaNH_(2)=2+1=3` |
|
| 14. |
One mole is the amount of substance that contains as many elementary particles as 12 g of _^12C isotope of carbon. Calculate the number of moles in 1 litre of water ("Density of water is "lg//mL). Also calculate the number of water molecules in 1 litre of water |
| Answer» Solution :NUMBER of moles = `"MASS of the substance"/"GRAM molecular mass"`Number of moles in LITRE of water =`(1000g)/(18g)=55.55` Number of water molecules in litre =`55.55xx6.022xx10^23=334.5xx10^23=3.345xx10^25` | |
| 15. |
One mole is the amount of substance that contains as many elementary particles as 12 g of _^12C isotope of carbon. What do you mean by molar mass of a compound? |
| Answer» Solution :The MASS of one mole of any substance is CALLED the MOLAR mass. | |
| 16. |
One mole fo carbon dioxide was found to occupy a volume of 1.32 litre at 48^(@)C under a pressure of 16.4 atm. Calculate the pressure that would have been expected from (i) the ideal gas equation, (ii) van der Waal's equation (a = 3.59 atm "litre"^(2) mol^(-1), b = 4.27 xx 10^(-3) " litre " mol^(-1) and R = 0.0821 "litre" - atom K^(-1) mol^(-1)) |
|
Answer» |
|
| 17. |
One mole CO_(2)=........... |
|
Answer» `3 G CO_(2)` molecule |
|
| 18. |
One mole each of a monoatomic, diatomic and triatomic gases are mixed C_(p)//C_(v) for the mixture is |
|
Answer» `1.40` `C_(p) = (((5)/(2) R + (7)/(2) R + (8)/(2) R))/(3) = (10R)/(3)` `(C_(p))/(C_(v)) = (10)/(7) = 1.428` |
|
| 19. |
One mole Barium chloride and one mole Sodium phosphate are mixed in aqueous medium. |
|
Answer» `BaCl_(2)` acts as limiting reagent `therefore BaCl_(2)` is the limiting reagent `3BaCl_(2)rarr1 Ba_(3)(PO_(4))_(2)` `1 RARR? =1//3` |
|
| 20. |
One mol of a non-ideal gas undergoes a changeofstate ( 2.0 atm, 3.0 L, 95K)rarr (4.0 atm, 5.0 L, 245 K) with a change in internal energy,DeltaU=30.0L atm. The changein enthalpy ( DeltaH ) of the process in L atm is |
|
Answer» <P>`40.0` `= 30 + 4 xx5 - 2 xx 3 = 44 L atm` |
|
| 21. |
One molal solution contains 1 mole of a solute in : |
|
Answer» 1000 G of the solvent |
|
| 22. |
One ml. of H_(2)O_(2) solution gives 50 ml. of O_(2) at NTP, so it is |
|
Answer» 10 V |
|
| 23. |
One million of silver atoms weigh 1.79 xx 10^(-16)g. Calculate the atomic mass of silver. |
|
Answer» `6.022 xx 10^(23)` atoms of silver occupy mass `= (1.79xx10^(-16)g)/(10^(6))xx6.02xx10^(23)=107.8g` Atomic mass of silver `= 107.8 g = 107.8 U`. |
|
| 24. |
One millimole of C_aSO_4 weighs _______ . (Ca = 40, S = 32, O = 16) |
|
Answer» 136 G = gram molecular weight of `CaSO_4` = 40 + 32 + 64 = 136 g One millimole of `CaSO_4` `= 136xx10^(-3) = 0.136 g` |
|
| 25. |
One metal oxide consist 60% metal what is the proportion of oxygen ? |
|
Answer» 0.6 |
|
| 26. |
One metal hydrogen phosphate has molecular formula MHPO_(4)then what is formula for metal chloride ? |
|
Answer» MCl |
|
| 27. |
One litre water has 81 mg of calcium bicarbonate and 95 mg of magnesium chloride dissolved. Find out the temporary, permanent and total hardness of water sample. |
| Answer» SOLUTION : 50ppm, 100ppm and 150ppm | |
| 28. |
One litre sample of sea water is found to contain5.8 xx 10^(-3) g of dissolved oxygen. Calculate the concentration of dissolved oxygen in sea water in ppm if the density of sea water is 1.03 g cm^(-3). |
|
Answer» SOLUTION :`THEREFORE` Density `=("mass")/("Volume")` `therefore` Mass of 1 litre `(1000 cm^(3))` of sea water = Volume `xx` Density `=1000 xx 1.03 = 1030 g` Thus, 1030 g of sea water contain `5.8 xx 10^(-3)` g of dissolved oxygen. Therefore, mass of oxygen PRESENT in 106 (ONE million) g of sea water `=(5.8 xx 10^(-3))/1030xx 10^(6) = 5.63 g` HENCE, the concentration of dissolved `O_(2)` in sea water = 5.63 ppm. |
|
| 29. |
One litre of water contains 10^(-7)"moles of " H^(+)ions . Degree of ionisation of water (in percentage ) is |
|
Answer» `1.8 xx 10^(-7) ` ` ALPHA % =( 10^(-7))/( 55.5 )xx 100 = 18 xx 10^(-8)= 1.8 xx 10 ^(-7)` |
|
| 30. |
One litre of oxygen at NTP weighs 1.46 g. How many litres of oxygen are needed for the combustion of 21.0 g of Mg whose equivalent weight is 1/2 mole? |
|
Answer» SOLUTION :The volume occupied by 1 equivalent at NTP is equivalent volume. `therefore` equivalent volume of oxygen `=` volume of 8.0 g of oxygen at NTP (eq. WT. of O = 8 ) `= ( 8)/( 1.46) = 5.48` litres. (1.46) g oxygen contains 1 L at NTP) Given that the equivalent weight of `MG = (1)/(2)xx 24 = 12 ""`(wt. of 1 mole of Mg = 24 g) Now, in the COMBUSTION, equivalent of Mg = equivalent of oxygen. `therefore ("weight of Mg")/("eq. weight of Mg")=("volume of oxygen at NTP")/("eq. volume of oxygen")` `(21)/(12) = ("volume of oxygen")/(5.48)` `therefore` volume of oxygen `=` 9.59 litres. |
|
| 31. |
One litre of milk weighs. 1.035 kg. The butter fat is 4%(v//v) of milk has density of 875 kg//m^(3). Find the density of fat free skimed milk. |
|
Answer» `1L` milk `rarr 40mL` fat DENSITY of fat `=875kg//m^(3)=0.875g//mL` mass of fat `=40xx0.875=35g` fat free milk mass `=1035-35=1000g` `vol. = 1000-40=960mL` `p=(1000)/(960)=1.0416g//mL` |
|
| 32. |
One litre of gaseous mixture of CH_(4) and H_(2) effuses in 200 seconds while one litre of gas 'X' takes 10 minutes to effuse in identical condition s. If molar ratio of CH_(4):H_(2) in mixture is 1:2. find molar |
|
Answer» 20 |
|
| 33. |
One litre of hydrogen effused in 8min through a 8 fine aperture. What is the time required for the same volume of ozone to effuse under similar conditions? |
|
Answer» Solution :The ratio of times taken for same VOLUME of TWO gases, under similar conditions is given by Graham.s equation `(t_2)/(t_1) = SQRT((M_1)/(M_1)) " or " t_2 = t_1 sqrt((M_2)/(M_1))` Time required for OZONE to effuse `= 8 xx sqrt(48//2) = 39.2 `min. |
|
| 34. |
One litre of CO_(2) is passed over hot coke. The volume becomes 1.4 lit. The resultant mixture contains. |
|
Answer» 0.6 LIT `CO_(2)` `1-0.4=0.6L 2xx0.4=0.8L` VOLUME`0.6+0.8=1.4L` |
|
| 35. |
One litre of a mixture of O_(2) " and " O_(3) at NTP was allowed to react with an excess of acidified solution of KI. The iodine liberated required 40 mL of M//10 sodium thiosulphate solution for titration. What is the weight per cent of ozone in the decompose ozone. Assuming that one photon can decompose one ozone molecule, how many photon can decompose required for the complete decomposition of ozone in the original mixture ? |
|
Answer» Applying PV=nRT `1xx1=nxx0.0821xx273` n=0.044 mole Moles of `O_(3)`= moles of `I_(2)=1//2` moles of `Na_(2)S_(2)O_(3)` `=(1)/(2)xx(1)/(10)xx(40)/(1000)=0.002` moles Moles of `O_(2)` in the mixture `=0.044-0.002=0.042` moles MASS of `O_(2)=0.042xx32=1.344 g` Mass of `O_(3)=0.002xx48=0.096 g` `%O_(3)=(0.096)/(1.44)xx100=6.67` No of photons required to decompose 0.002 moles of ozone `=0.002xx6.02xx10^(23)` `=1.204xx10^(21)` |
|
| 36. |
One litre of a mxiture of CO and CO_(2) is passed over red hot coke when the volume increased to 1.6 L under the same conditions of temperature and pressure . The volume of CO in the original mixture is |
|
Answer» 400ml |
|
| 37. |
One litreof a mixture of carbonmonoxide and carbondioxide is reqeatedly passed over red hot coke. The mixture is cooled and collected to occupy 1.25L. What is the percentage volume of carbon monooxide in the given mixture? |
|
Answer» |
|
| 38. |
One litre of a mixture containing BaF_(2) and H_(2)SO_(4) was taken for analysis. 25 mL of this mixture was added to 100 mL of 0.05 N K_(2)CO_(3) solution and precipitate was filtered off. The filtrate required 12 mL fo 0.025 M oxalic acid solution using phenolphthalein as indicator. Find the strength of BaF_(2) and H_(2)SO_(4) in mixture. |
|
Answer» |
|
| 39. |
One litre of a gas weighs 4g at 300 K and 1 atm. If the pressure is reduced to 0.75 atm, the temperature at which one litre of the same gas weights 2g is |
|
Answer» 600 K |
|
| 40. |
One litre of a gas weighs 3.57 xx 10^-3 kg at N.T. P. The gas is ________ . ( C = 12, O = 16, S = 32) |
|
Answer» Carbon monoxide (b) `(64)/(22.4) = 2.857g = 2.857 xx10^(-3) kg` ( C) `(80)/(22.4) = 3.57 g = 3.57 xx10^(-3) kg` (d) `(44)/(22.4) = 1.96 g = 1.96xx10^(-3) kg` The correct answer is ( c). |
|
| 41. |
One litre of a gas weighs 2g at 300 K and 1 tm pressure. If the pressure is changed to 0.75 atm at which of the following temperatures, will one litre of the same gas weight 1 g? |
|
Answer» Solution :According to ideal gas EQUATION, `PV = nRT = w//M RT` For a particular gas : `(P_(1)V_(1))/(P_(2)V_(2)) = (n_(1)RT_(1))/(n_(2)RT) = (w_(1)RT_(1))/(w_(2)RT_(2))` `(1 xx 1)/(0.75 xx 1) = (2 xx R xx 300)/(1 xx R xx T_(2))` or `T_(2) = 2 xx 300 xx 0.75 = 450 K` |
|
| 42. |
One litre of a gas collected at NTP will occupy at 2 atmospheric pressure and 27^(@)C |
|
Answer» `(300)/(2 XX 273)` LITRE |
|
| 43. |
One litre of a gas weighs 2 g at 300 K and 1 atm pressure. If the pressure is made 0.75 atm, at which of the following temperature will one litre of the same gas weigh 1 gram :1) 450K2) 600K3) 800K4)900K |
|
Answer» 450 K |
|
| 44. |
One litre of a gas weights 28 at 300 K and latm pressure. If the pressure is made 0.75 atm, at what temperature will one litre of the same gas weigh lg |
| Answer» ANSWER :D | |
| 45. |
One litre of a buffer solution containing 0.02 mol of propanoic acid and some sodium propanate has pH = 4.75. What will be the pH if 0.01 mol of hydrogen chloride is dissolved in the above buffer solution ? [Dissociation constant of propanoic acid at 25^(@)C is 1.34 xx 10^(-5)]. |
|
Answer» `4.11` `PH = pKa + "log" (["salt"])/(["acid"])` We get `4.75 = - log(1.34 xx 10^(-5)) + "log" (["salt"])/(["acid"])` which gives `4.75 = 4.87 + "log" (["salt"])/(0.02M)` or `(["Salt"])/(0.02M) = 0.76` or `["salt"] = 1.52 xx 10^(-2)M` Hence, AMOUNT of sodium propanoate to be added `= 1.52 xx 10^(-2) "mol"` The addition of `0.01` hydrogen chloride convert the equivalent amount of sodium propanoate into PROPANOIC acid. |
|
| 46. |
One litre glass bulb contains 2 xx10^(21) molecules of nitrogen at a pessure of 7.57 xx 10^(21) Newton m^(-2). Find the RMS velocity and temperature of the gas. If the ratio of most probable velocity and RMS velocity is 0.82, find the most probable velocity of nitrogen gas at the same temperature. |
|
Answer» Solution :From the ideal equation PV = nRT `T = (PV)/(nR) =(7.57 xx 10^3 xx 1 xx 6.-23 xx 10^(23))/(1.013 xx 10^5 xx 2 xx 10^(21) xx 0.0821) = 274.6K` `T = (PV)/(Nr)= (7.57 xx 10^3 xx 1 xx 6.023 xx 10^23)/(1.013 xx 10^5 xx 2 xx 10^(21) xx 0.0821) = 274.6 K` R.M.S . Velocity (C )= `sqrt((3RT)/(M)) = sqrt((3 xx 8.314 xx 274.6)/(28 xx 10^(-3))) = 495.5 ms^(-1)` Most probable velocity `(C_P) = 0.82 xx ` RMS velocity `= 0.82 xx 494.5 = 405 ms^(-1)`. |
|
| 47. |
One litre glass bubl contains2 xx 10^(21) molecules of nitrogen at a pressure of7.57 xx 10^(-3)N.m^(-2). Find out the RMS velocity of nitrogen. |
|
Answer» Solution :`C = sqrt((3PV)/(M))` Where V is molar VOLUME Volume OCCUPIED by one mole of `N_(2)` gas ` = (6 xx 10^(23))/(2 xx 10^(21)) = 30 L = 30 xx 10^(-3)m^(3) = 3 xx 10^(-2) m^(3)` `P = 7.57 xx 10^(3) N.m^(-2), M = 28g = 28 xx 10^(-3)` Kg RMS VELOCITY `C = sqrt((3PV)/(M)) = sqrt((3xx 7.57 xx 10^(3) xx 3 xx 10^(-2))/(28 xx 10^(-3))) = 494.5ms^(-1)` |
|
| 48. |
One litre gas at 400K and 300atm pressure is compressed to a pressure of 600 atm and 100K. The compressibility factor is changed from 1.2 to 1.6 respectively. Calculate the final volume of the gas. |
|
Answer» 2 LITRE |
|
| 49. |
One litre flask contains air, water vapour and a small amount of liquid water at 50^(@)C and 200 torr. If this flask is connected to another one litre evacuated flask, what will be the final pressure in each flask ? Given that the aqueous tension at50^(@)C si 92 torr |
| Answer» SOLUTION :146 TORR | |