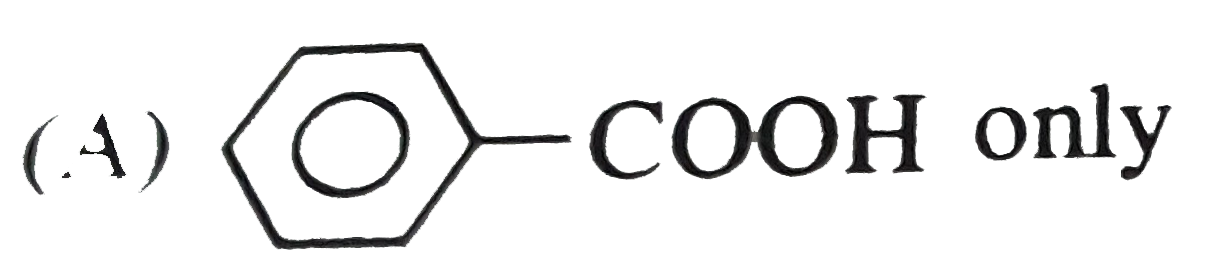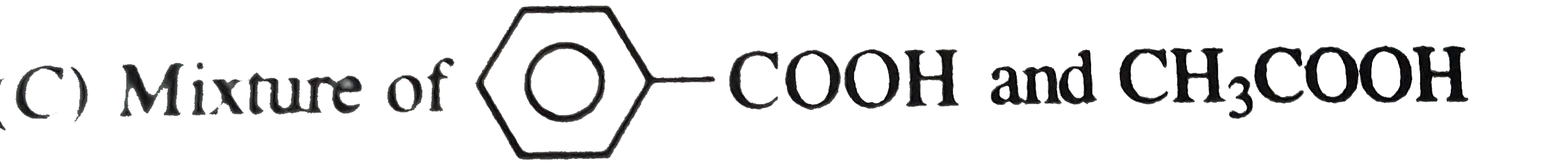Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Oxides of sulphur (SO_(2),SO_(3)) are due to |
|
Answer» buring of sulphur containing fossil ORE (b) `2CuFeS_(2)+5.5O_(2)underset("Roasting")overset("Smelting")(rarr) Cu_(2)O+2FeO+4SO_(2)` (c ) `SO_(2)+O_(3)rarr SO_(3)+O_(2)` `SO_(3)+O_(3) rarr SO_(3)+O_(2)`. |
|
| 2. |
Oxides of nitrogen |
|
Answer» are TOXIC to living TISSUES |
|
| 4. |
Oxides of beryllium and magnesium are ...... in water. |
|
Answer» Insoluble |
|
| 5. |
Oxides of alkaline earth metals are stable due to |
|
Answer» HIGH lattice ENERGY |
|
| 6. |
Oxidesand hydroxidesof alkaloineearthmetalsEXCEPT berylliumare ______in nature. |
|
Answer» ACIDIC |
|
| 7. |
Oxide that is most acidic |
|
Answer» `Cl_2O_7` |
|
| 8. |
oxidation states of P in H_(4)P_(2)O_(5),H_(4)P_(2)O_(6),H_(4)P_(2)O_(7) are respectively |
|
Answer» <P>`+3,+5,+4` or X=+3 O.N of p in `h_(4)P_(2)O_(6)=4(+1)+2x+6(-2)=0` or x =+4 O.N of p in `H_(4)P_(2)O_(7)=4(+1)+2x+7(-2)=0` or x=+5 |
|
| 9. |
Oxidation states of the metal in the mineral haematite and magnetite ,respectively are |
|
Answer» II, III in haematite and III in magnetite O.S of Fe is II and III . |
|
| 10. |
Oxidation states of P in H_(4)P_(2)O_(5),H_(4)P_(2)O_(6),H_(4)P_(2)O_(7) respectively are |
|
Answer» `+3, +5, +4` |
|
| 11. |
Oxidation states of P in H_(4)P_(2)O_(5),H_(4)P_(2)O_(6),H_(4)P_(2)O_(7) are: |
|
Answer» `+3,+5,+4` `H_(4)P_(2)O_(6),4+2X-12=0,X=+4` `H_(4)P_(2)O_(7),4+2x-14=0,X=+4` |
|
| 12. |
Oxidation states of P in H_4P_2O_5, H_4P_2O_6 and H_4P_2O_7 are respectively : |
|
Answer» `+3, +5 and +4` |
|
| 13. |
Oxidation state of terminal sulphur atoms in S_(4)O_(6)^(2-)02 (tetra thionate ion) are |
|
Answer» |
|
| 14. |
Oxidation state of .S. in S_8molecule is |
| Answer» Solution :`S_8`molecule is a ELEMENTAL from of sulphur so, oxidation state is ZERO. | |
| 15. |
Oxidation state of phosphorus in pyrophosphosphate ion (P_(2)O_(7)^(-4)) is |
|
Answer» SOLUTION :`P_(2)O_(7)^(-4)` `2x +7 (-2) = 4` `2x = +10 rArr X = +5` |
|
| 16. |
Oxidation state of oxygen is -1 in the compound : |
|
Answer» `NO_2` |
|
| 18. |
Oxidation state of oxygen in H_2O_2is |
|
Answer» `-2` |
|
| 22. |
Oxidation state of nitrogen is incorrectly given for M {:("Compound ","Oxidation State"),("a)" [Co(NH_3)_5Cl]Cl_2 ,-3),("b)" NH_2OH,-1),("c)" (N_2H_5)_2SO_4,+2),("d)" (Mg_(3))N_2,-3) :} |
|
Answer» `4x=-8,x=-2` |
|
| 23. |
Oxidation state of Ni in Ni(CO)_(4) is |
|
Answer» 0 |
|
| 25. |
Oxidation state of K in KO_(2) is same as that in : |
|
Answer» `KO_(3)` |
|
| 28. |
The oxidation state of Fe in Fe_(3)O_(8) is |
|
Answer» `(3)/(2)` |
|
| 29. |
Oxidation state of each CI in CaOCI_(2) is / are |
|
Answer» 0 |
|
| 32. |
Oxidation state +4 is less common in |
|
Answer» C |
|
| 33. |
Oxidation product of acetylene with chromic acid is |
|
Answer» Acetic acid |
|
| 34. |
Oxidation potential of Zn, Cu, Ag are 0.76V, -0.34V, -0.80V, respectively then write down order of tendency of losing e^(-). |
| Answer» Solution :`ZngtCugtAg` (Greater the VALUE of oxidation POTENTIAL, greater the `e^(-)` releasing CAPACITY.) | |
| 35. |
Oxidation of toluene withCrO_(3) in presence of(CH_(3)CO)_(2)Ogives a product (A) which onhydrolysis forms Benzaldehyde. A is ___ |
|
Answer» Chromoum complex |
|
| 36. |
Oxidation of toluene by acidic KMnO_(4) gives poor yield of benzoic acid while oxidation of p-nitrotoluene gives good yield of p-nitrobenzoic acid. Why? |
| Answer» Solution :Oxidant is an electrophile, it can attack and destroy the ring in acse of TOLUENE. But in p-nitrotoluene, the -`NO_(2)` GROUP deactivates the ELECTROPHILIC attack on benzene NUCLEUS and thus INCREASES yield of p-nitrobenzoic acid. | |
| 37. |
Oxidation of sulphur dioxide into sulphur trioxide in the absence of a catalyst is a slow process but this oxidation occurs easily in the atmosphere. Explain how does this happen. Give chemical reactions for the conversion of SO_2 into SO_3 |
| Answer» SOLUTION :The presence of particulates in the air CATALYSES OXIDATION of `SO_2` to `SO_3` (`2 SO_2+ O_2 overset"Particulates " to 2SO_3`) | |
| 38. |
Oxidation of sulphur dioxide into sulphur trioxide in the absence of a catalyst is a slow process but this oxidation occurs easily in the atmosphere. Explain how does this happen. Give chemical reactions for the conversion of SO_2 into SO_3. |
|
Answer» Solution :The oxidation of SULPHUR DIOXIDE into sulphur trioxide can occur both photochemically or non-photochemically. In the NEAR ULTRAVIOLET region, the `SO_2` molecules react `O_3` photochemically. `SO_2 + O_3 overset(hv)to SO_3 +O_2` `2SO_2 + O_2 overset(hv)to 2SO_3` Non-photochemically, `SO_2` may be oxidised by molecular oxygen in presence of dust and soot particles. `2SO_2 + O_2 overset("Particulates")to 2SO_3` |
|
| 39. |
Oxidation of nitrogen monoxide was strudied at 200^@C with initial pressure of 1 atm NO and 1 atm of O_2. At equilibrium partial pressure of oxygen is found to be 0.52 atm. Calculate K_P value. |
Answer» SOLUTION :`2NO(g) + O_2(g) hArr 2NO_2(g)` `[O_2]` at equilibrium `RARR 1 - x = 0.52` ` x = 0.48` So, `P_(NO) = 1-2x = 1 - 2(0.48) = 0.04` `P_(O_2) = 0.52` `P_(NO_2) = 2x= 2(0.48) = 0.96` `K_P ((P_(NO_2)))/((P_(NO)^2)(P_(O_2)))=(0.96 XX 0.96)/(0.04 xx 0.04 xx 0.52)` `K_P = 1.107 xx 10^3` |
|
| 40. |
Oxidation of nitrogen monoxide was studied at 200^(@)C with initial pressures of 1 atm NO and 1 atm of O_(2). At equilibrium partial pressure of oxygen is found to be 0.52 atm calculate K_(P) value. |
|
Answer» <P> Solution :`2NO(G)+O_(2)(g)hArr2NO_(2)(g)` `K_(P)=((P_(NO_(2))^(2)))/((P_(NO))^(2)(P_(O)))=(0.96xx0.96)/(0.04xx.0.04xx0.52)` `K_(P)=1.017xx10^(3)` |
|
| 41. |
Oxidation of methyl phenyl ketone with conc. HNO_(3) gives |
Answer» Solution :Oxidation of methyl phenyl KETONE with conc. `HNO_(3)` gives a mixture of benzoic acid and acetic acid having less number of CARBON atoms as compared to PARENT ketone. 
|
|
| 42. |
Oxidation of cyclopentanol to cyclopentanone can be accomplished by using |
|
Answer» TOLLEN's reagent |
|
| 43. |
Oxidation of C_(6)H_(5)CH_(2)Cl with alk, KMnO_(4) gives_____ |
|
Answer» `C_(6)H_(3)CHO` |
|
| 44. |
Oxidation of base at anode is given by the chemical equation :4OH^(-)toO_(2)+2H_(2)O+4e^(-)What is the equivalent weight of water? |
|
Answer» Solution :4moles of electrons =2 moles of WATER 2Faradays = 1mole of water Equivalent weight `=("molecular weight")/("number of FARADAYS")=(18)/(2)=9` |
|
| 45. |
Oxidation of an alkene X gives a diol , further oxidation gives a diketone. Which one of the following could be X ? |
|
Answer» `(CH_3)_2C=C(CH_3)_2` `UNDERSET"(X)"(C_6H_5CH=CHC_6H_5)underset"(BAEYER's reagent)"overset("Cold. aq. ALK." KMnO_4) to underset(("Diol contains two " 2^@ "alcoholic groups"))(C_6H_5-underset(OH)underset|CH-underset(OH)underset|CH-C_6H_5)overset"[O]"to underset"Diketone"(C_6H_5-undersetundersetO(||)C-undersetundersetO(||)C-C_6H_5)` |
|
| 47. |
Oxidation of alkenes by cleavage with acidic or alkaline KMnO_4 or acidic K_2Cr_2O_7 at higher temperature yields products depending upon the nature of alkene. A hot solution of is a strong oxidizing agent which gives only ketones and carboxylic acids and not aldehydes (as they cannot be isolated) Which of the these compounds on treatment with OsO_4 followed by Na_2SO_3 will give Cis-2-methylbutane-2,3-diol? |
|
Answer» 2-Methyl-2-butene |
|
| 48. |
Oxidation of alkenes by cleavage with acidic or alkaline KMnO_4 or acidic K_2Cr_2O_7 at higher temperature yields products depending upon the nature of alkene. A hot solution of is a strong oxidizing agent which gives only ketones and carboxylic acids and not aldehydes (as they cannot be isolated) Which of these compounds on oxidation with hot KMnOą gives only butanoic acid? |
|
Answer» Oct-3-ene |
|
| 49. |
Oxidation of acetaldehyde with selenium dioxide produces |
|
Answer» Ethanoic acid |
|
| 50. |
Oxidation numbers of the metal in the minerals haematite and magneetite respectively are: |
|
Answer» II,III in haematite and III in magnetite Fe:2x+3(-2)=0,x=+3 In magnetitte `(Fe_(3)O_(4))` is a mixed oxide of FEO and `Fe_(2)O_(3)` In FeO,the oxidation number of Fe: x-2=0 orx=+2 In `Fe_(2)O_(3)`O,the oxidation number of Fe: 2x+3(-2) =0 or x=+3 |
|