Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Silica is added to roasted copper pyrites during smelting in order to remove |
|
Answer» CUPROUS oxide |
|
| 2. |
Silica is a covalent solid, but dry is a molecular solid. Why ? |
|
Answer» Solution :In silica, a chain of `SiO_(4)` tetrahedra is PRESENT in a NETWORK 3D-structure. In dry ice, DISCRETE molecular units of `CO_(2)` are present. Between molecules, only weak dispersion forces exist. |
|
| 3. |
Silanes (Si_(H)H_(2n+2)) compounds are less stable (highly reactivcs) than the corresponding hydrocarbons (C_(n) H_(2n+2)). Why? |
|
Answer» The Si-Si bond is slightly WEAKER than the C - C bond |
|
| 4. |
Signa of Delta G for the melting of ice is negative at |
|
Answer» 265K |
|
| 5. |
sigma bond is stronger compare to p bond. |
| Answer» Solution :In a `SIGMA` BOND the overlapping is along the axis and is maximum EXTENT. Where as `pi` bond overlapping is sidewise and overlappingis MINIMUM. | |
| 6. |
Sigma bonds are : |
|
Answer» CYLINDRICALLY symmetrical about the BOND axis |
|
| 7. |
Sigma bond is not f orme in which overlapping of the following ? (i) 2p_(y) - 2p_(y) (ii) 2p_(z)- 2p_(z)(iii) 2s - 2p_(x)(iv) 2p_(x) - 2p_(x) |
| Answer» SOLUTION :(i) and (IV) | |
| 8. |
[SiF_6]^(2-) is known whereas [SiCl_6]^(2-) not. Give possible reasons. |
| Answer» Solution :The MAIN reasons are : (i) Six large chloride ions cannot be ACCOMMODATED around `Si^(4+)` due to LIMITATION of its size. (II) Interaction between LONE pair of chloride ion and `Si^(4+)` is not very strong. | |
| 9. |
SiF_6^(2-)is known while SiCl_6^(-2)is not - explain. |
| Answer» SOLUTION :Six LARGE CHLORIDE IONS cannot be accommodated AROUND `Si^(4+)` due to limitation of its size. Interaction between lone pair of chloride ion and `Si^(4+)` is not very strong. | |
| 10. |
SiF_(4) on hydropysis gives X and Y. SiCl_(4) on hydrolysis gives X and Z. Covalency of central atom in Y is C_(1) and that of halogenin Z is C_(2). Then C_(1) + C_(2) is: |
|
Answer» |
|
| 11. |
[SiF_(6)]^(2-)is knownwheares [SiCl_(6)]^(2-)not. Givepossiblereasons. |
|
Answer» Solution :The mainreasons are : (i) Due to smallersize ofF as comparedto Cl, six SMALL Fatoms can be easily accomodatedaround Si atom but six largeCl atoms cannot. (II) The lone PAIR of electrons on F are present in a smaller 2p-orbitalbut in Cl they are present on a LARGER 3p-orbital.Therefore, interaction of F lone pair electrons with d-orbitalsof siliconare stronger than that of Cl lone pairs. |
|
| 12. |
SiF_4 has octet, but it has residual bonding ability. Comment. |
| Answer» Solution :SILICON has four valance electons and forms four bonds with fluorine atoms. `SiF_(4)` has octet configuration. It is an ELECTRON PAIR acceptor, due to the AVAILABILITY of empty VALENCE d-orbitals. | |
| 13. |
SiCl_(4) overset(H_(2)O)(to) X overset("Heat")(to) Y overset(NaOH)(to) Z find X,Y,Z. |
|
Answer» `{:(SiO_(2), Si,NASI):}` 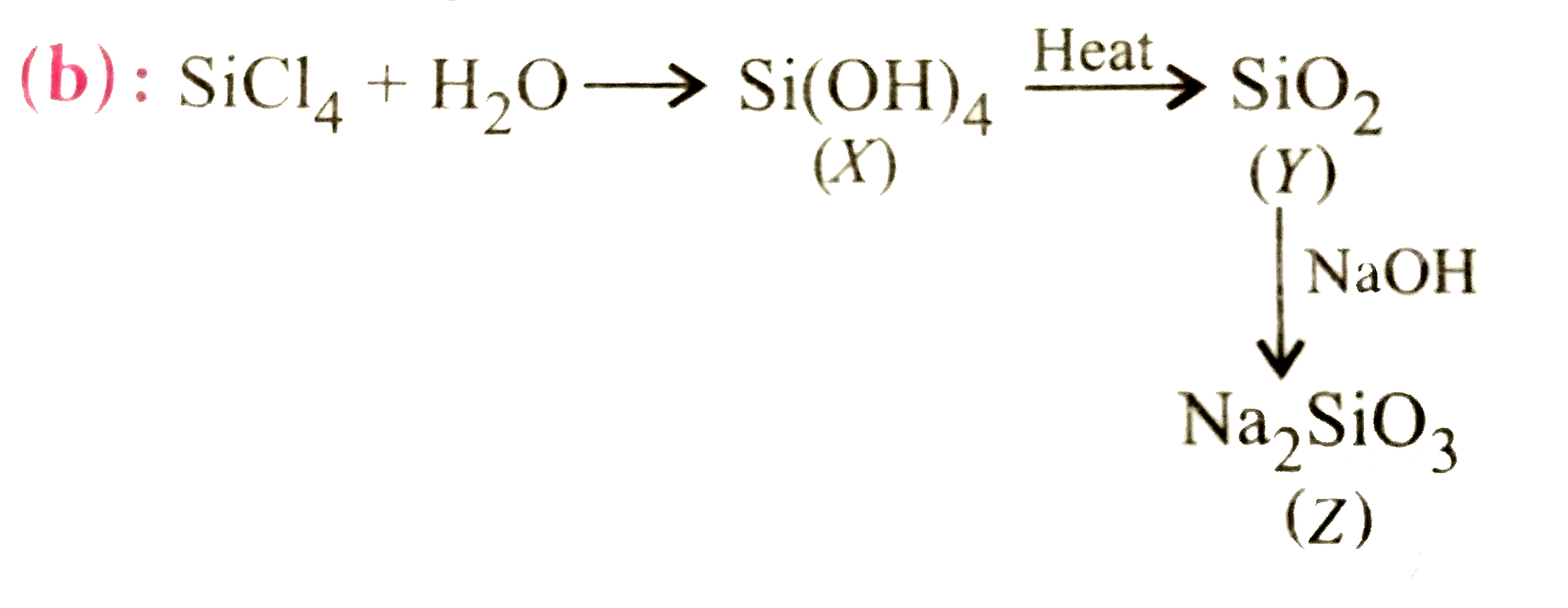
|
|
| 14. |
S.I. unit for dipole moment is |
|
Answer» e.s.u -cm |
|
| 15. |
Silicon has diagonal relationship with |
|
Answer» Sulphur 
|
|
| 16. |
Si has digonal relationship with |
|
Answer» Sulphur |
|
| 17. |
Shows which type of isomerism |
|
Answer» FUNCTIONAL GROUP of isomerism |
|
| 18. |
Shows which type of isomerism |
|
Answer» Functional GROUP isomerism |
|
| 19. |
Shows which type of isomerism |
|
Answer» functional group of isomerism `-overset(O)overset(||)(C)-O-` is bivalent group which has different alkyl groups in both compounds. As we know that ACCORDING to definitions of matamerism "when alkyl group is different around to bivalent to TRIVALENT atom 1 group compounds termed as metamers.  Although in second compound TRANS from is present but strucutre is ALSO present so it will be metamers not geometrical isomer. |
|
| 20. |
Show the polarisation of carbon-magnesium bond in the following structure. CH_(3)-CH_(2)-CH_(2)-CH_(2)-Mg-X |
Answer» Solution :SINCE electronegativity of Mg (1.2) is MUCH LOWER than that of C (2.5), therefore , Mg carries a PARTIAL positive charge while the carbon attached to it carries a partial negative charge, i.e., 
|
|
| 21. |
Show the polarisation of carbon -magnesium bond in the following structure. CH_(3)-CH_(2)-CH_(2)-CH_(2)-Mg-X |
Answer» Solution :Since electronegativity of MG (1, 2) is much lower than that of C(2.5), therefore, Mg carries a partical POSITIVE charge while the carbon attached to it carries a partical negative CHORE. 
|
|
| 22. |
Show the heterolysis of covalent bond by using curved arrow notation and ocmplete the following equation . Identify the nucleophile is each case . CH_(3) - OCH_(3) + HI to |
Answer» SOLUTION : 1 from HI ACTS as a NUCLEOPHILE |
|
| 23. |
Show the heterolysis of covalent bond by using curved arrow notation and ocmplete the following equation . Identify the nucleophile is each case .CH_(3) - Br + KOH to |
Answer» SOLUTION : `OH^(-)` from KOH is the nuclephilc |
|
| 24. |
Show the oxidising property of H_(2)O_(2) with PbS. |
|
Answer» Solution :Hydrogen peroxide oxidises LEAD sulphide to lead SULPHATE in a NEUTRAL medium. `PbS+3H_(2)O_(2)toPbSO_(4)4H_(2)O` |
|
| 25. |
Show the heterolysis of covalent bond by using curved arrow notation and complete thefollowing equations.Identify the nucleophile in each case. (i) CH_(3) -Br + KOH to (ii) CH_(3) - OCH_(3) + HI to |
Answer» SOLUTION :
|
|
| 26. |
Show the heterolysis of covalent bond by using curved arrow notation and complete the following equations.Identify the nucleophile is each case. CH_(3)-Br+KOHrarr |
Answer» SOLUTION : `OH^(-)` from KOH is the NUCLEOPHILE. |
|
| 27. |
Show the heterolysis of covalent bond by using curved arrow notation and complete the following equations.Identify the nucleophile is each case. CH_(3)-OCH_(3)+HIrarr |
Answer» SOLUTION : I from HI ACTS as a NUCLEOPHILE. |
|
| 28. |
Show the electron distribution in the molecular orbitals of the cycloheptatrienyl cation. Strategy: Use the polygon-andj-crcle method for deriving the relative energies of the Mos. |
Answer» Solution :The polygon-and-circle medthod shows that the three lowest ENERGY `MOs` are below the DIAMETER and are bonding The FOUR HIGHEST energy ones are above the diameter and are antibonding `MO s`
|
|
| 29. |
Show the distribution of electrons in oxygen atom (atomic number 8) using orbital diagram |
Answer» SOLUTION :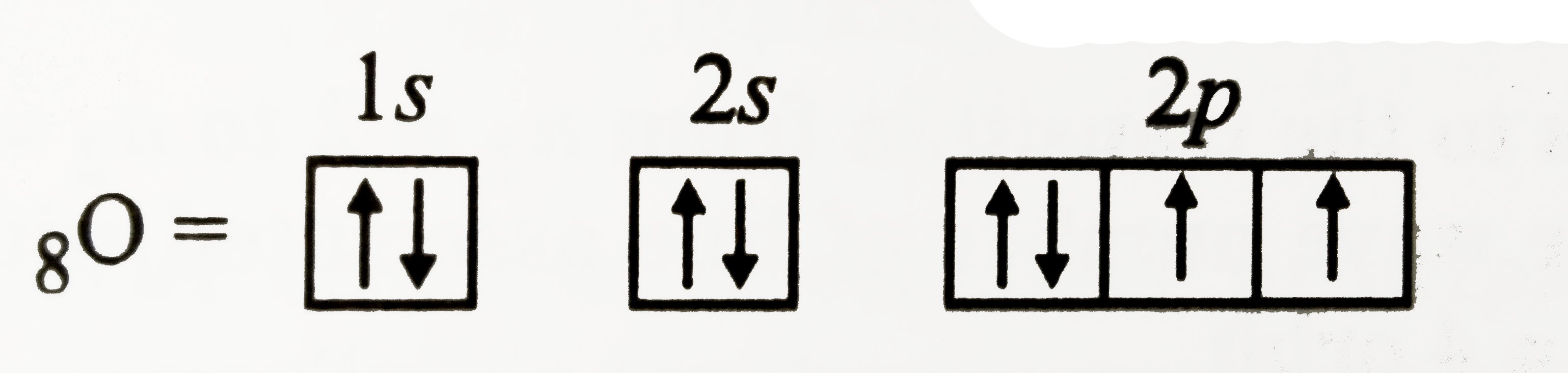
|
|
| 30. |
Show the distribution of electrons in oxygen atom (atom ic num ber 8) using orbital diagram . |
Answer» SOLUTION :
|
|
| 31. |
Show the direction of inductive effect in following compounds |
|
Answer» |
|
| 32. |
Show that the sum of mole fraction of a solution is equal to one. |
|
Answer» Solution :Consider a solution CONTAINING two COMPONENTS A and B whose mole fractions are `x _(A) and x _(B)` respectively. Let the number of moles of two components A and B are `n_(A) and n_(B)` respectively. `(n _(A))/(n _(A) + n _(B)), x _(B) = (n_(B))/(n _(A) + n _(B))` ` x _(A) + x _(B) = ( n _(A))/(n _(A) + n _(B)) + (n_(B))/( n _(A) + n _(B)) =1` |
|
| 33. |
Show that the reaction : CO(g) + 1/2O_(2)(g) to CO_(2)(g) at 300 K is spontaneous and exothermic, when standard entropy change is -0.094 kJ"mol"^(-1).The standard Gibbs energies of formation of CO_(2) and CO are -394.4 and -137.2 kJ"mol"^(-1) respectively. |
|
Answer» Solution :`Delta_(R)G^(@) = Delta_(f)G^(@)("products") - Delta_(f)G^(@)("REACTANTS")` ` = [Delta_(f)G^(@(CO_(2))] - [Delta_(f)G^(@)(CO) + 1/2Delta_(f)G^(@)(O_(2))]` `= -394.4 - [-137.2 + 0]` ` = -257.2 kJ` Since `Delta_(r)G^(@)` is negative, the REACTION is spontaneous. Now `DELTAG^(@) = DeltaH^(@) -TDeltaS` ` -257.2 = DeltaH^(@) -[300 xx(-0.094)]` `therefore DeltaH^(@) = -257.2 -28.2 = -285.4 kJ` Since `DeltaH^(@)` is negative, the reaction is exothermic. |
|
| 34. |
Show that the role of cone, sulphuric acid is not a dehydrating agent, but used for production of nitronium cation. |
|
Answer» Solution :Nitration mixture is a mixture of conc. nitric ACID and conc. sulphuric acid. Sulphuric acid acts as an acid and donates a proton to nitric acid. The protonated nitric acid, gives NITRONIUM cation by the loss of water molecule. `HHSO_(4)+HNO_(3) to HSO_(4)^(-)+H_(2)O^(+) -NO_(2) , H_(2) OVERSET(+)(O) NO_(2) to NO_(2)^(+)+ H_(2)O` Benzene gives nitorbenzene by electrophilic substitution. `C_(6)H_(6)+NO_(2)^(+) to C_(6)H_(5)NO_(2)+H^(+) , H^(+)+HSO_(4)^(-) to H_(2)SO_(4)` |
|
| 35. |
Show that the reaction CO+1//2O_2 to CO_2 at 300 K is spontaneous. The standard Gibbs free energies of formation of CO_2 and COare -394.4 and -137.2 "kJ mole"^(-1)respectively. |
|
Answer» Solution :`CO+1//2O_2 to CO_2` `DeltaG_"(REACTION)"^0=sum G_(f"(products)")^0-sum G_(f"(reactants)")^0` `DeltaG_"(reaction)"^0=[G_(CO_2)^0]-[G_(CO)^0+1/2G_(O_2)^0]` `DeltaG_"(reaction)"^0`=-394.4+[137.2+0] `DeltaG_"(reaction)"^0=-257.2 "kJ MOL"^(-1)` `DeltaG_"(reaction)"` of a reaction at a given temperature is NEGATIVE hence the reaction is spontaneous |
|
| 36. |
Show that the formation of sodium choride from gaseous sodium and gaseous chlorine is a redox reaction |
|
Answer» SOLUTION :`Na(G) RARR Na^(+)(g)+e^(-)]xx2` `CI_(2)(g)+2E^(-)rarr2cI^(-)(g)` `2Na(g)+cI_(2)rarr2Na^(+)CI^(-)(s)` |
|
| 37. |
Show that the cirumference of the Bohr orbit for hydrogen atom is an integral multiple of the de Broglie wavelength associated with the electron moving around the orbit. |
|
Answer» SOLUTION :According to Bohr POSTULATE of angular momentum, `mvr= n h/(2PI) or 2 pi r=n h/(mv)` ...(i) According to de Broglie equation, `lambda=h/(mv)` ...(ii) Substituting this VALUE in eqn. (i) we get `2 pi r= n lambda` Thus, the cirumference `(2pi r)` of the bohr orbit for hydrogen atom is an integral mutiple of de Broglie wavelength. |
|
| 38. |
Show that the data given below is in agreement with the law of constant composition {:("Exp. No.","Mass of iron","Mass of iron oxide"),("(i)","2.7812 g","3.9756 g"),("(ii)","3.0499 g","4.3596 g"),("(iii)","4.4913 g","6.4202 g"):} |
|
Answer» Solution :Experiement No. (i) MASS of iron = 2.7812 g , Mass of iron oxide = 3.9756 g PERCENTAGE of iron `= ((2.7812g))/((3.9756g))xx100=70%` Percentage of oxygen `= 100 - 70 = 30 %` Experiment No. (ii) Mass of iron = 3.0499 , Mass of iron oxide = 4.3596 g Percentage of iron `= ((3.0499))/((4.3596g))xx100=70%` Percentage of oxygen `= 100-70=30%` Experiment No. (iii) Mass of iron = 4.4913 g , Mass iron oxide = 6.4202 g Percentage of iron `= ((4.4913g))/((6.42402g))xx100=70%` Percentage of oxygen `= 100 - 70 = 30 %` Since the RATIO by weights of iron and oxygen in the THREE oxides is the same, the DATA is according to Law of constant Composition. |
|
| 39. |
Show that the circumference of the Bohr orbital for the hydrogen atom is an integral multiple of the de Broglie wave lenght associated with the electron revolving arround the nucleus. |
|
Answer» Solution :In order for the electron wave to EXIST in phase, the circumference of the ORBIT should be an integral MULTIPLE of the wavelength of the electron wave. Their electron wave is out phase. `mvr = nh//2pi` Where `mvr` = ANGULAR momentum Where `2 pi r` = circumference of the orbit Circumference of the orbit = `n LAMBDA`  `2 pir = n lambda` `n = 3, n = 4`. |
|
| 40. |
Show that the circumference of the Bohr orbit for the hydrogen atom is an intergral multiple of the de Broglie wavelength associated with the electron revolving around the orbit. |
|
Answer» Solution :According to Bohr.s THEORY, `mvr=(nh)/(2pi)`(n=1,2,3,…….so on) ` or 2pir=(nh)/(mv) or mv=(nh)/(2pir)`……..(i) According to de Broglie equation, `GAMMA=h/(mv) or mv=(h)/(gamma)`…………..(ii) COMPARING (i)and (ii), `(nh)/(2pir)=h/(pi)or 2pir=ngamma` THUS,the circumference`(2pir)`of the Bohr orbit for hydrogen ATOM is an into the de Broglie wavelength. |
|
| 41. |
Show that the circumference of the Bohr orbit for the hydrogen atom is an integral multiple of the de Broglie wave length associated with the electron revolving around the nucleus. |
|
Answer» SOLUTION :In ORDER for the electron WAVE to exist in phase,the circumference of the orbital should be an intergral multiple of the wavelength of the electron wave.otherwise ,the electron wave is out of phase. mvr=ng/2pi `2pir = N lambda` where mvr=angular momentum  where `2pir`=circumference of the orbit. where `2pir`=circumference of the orbit.n= 3, n=4 |
|
| 42. |
Showthat thecircumferencefo the Bohrorbitfor the hydrogenatom isan integralmultipleof thede-Brogliewavelengthassociatedwiththe electronrevolvingaroundthe orbit. |
|
Answer» Solution :ForBohr.smodelangularmomentumofelementis `m_(e)vr = (nh)/(2pi)` circumference `=2pi R = (nh)/(m_(e ) v)` and `m_( e)v= (nh)/(2pi r)` Accordingot de - BROGLIE `lambda= (h)/(mv) ` andmv `=(h)/(lambda)` as (i) and(II) mv = `(nh)/( 2pi r)= (h)/( lambda ) :., (N)/(2pi r)= (1)/(lambda):., 2pi r= n lambda` Bohr.sorbitcircumference `(2pi r ) = n ` de- Brogliewavelength In this wayde broylicwave lengthis associatedwthBohr.sorbitcircumference. |
|
| 43. |
Show that solubility of a sparingly soluble salt M^(2+)A^(2-) in which A^(2-) ion undergoes hydrolysis is given by: S=sqrt(K_(SP)(1+([H^(+)])/(K_(2))+([H^(+)]^(2))/(K_(1)K_(2)))) where K_(1) and K_(2) are the dissociation constant of acid HA_(2).K_(SP) is solubility product of MA. |
| Answer» | |
| 44. |
Show that six hydrogen atoms in benzene are identical. |
|
Answer» Solution :When benzene ADDS THREE molecules of chlorine in the presense of SUNLIGHT gives benzen hexachloride. `C_(6)underset("Benzene")(H_(6)) + 3Cl_(2) overset("sunlight")(rarr) underset("Benzene HEXS Chlonde(BHC)")(C_(6)H_(6)Cl_(6))` |
|
| 45. |
Show that presence of three double bonds in benzene. |
|
Answer» Solution :When benzene adds THREE molecules of hydrogen in presence of nickel catalyst of formcyclohexane `C_(6)H_(6) + underset("Benzene")(3H_(2)) underset(180^(@)C)overset(" "Ni" ")(rarr) underset("CYCLOHEXANE")(C_(6)H_(12)` |
|
| 46. |
Show that K_p = K_c with two examples |
|
Answer» Solution :`K_p +K_c (RT)^(Delta)g`. If `Delta n_g = 0. K_p = K_c` EXAMPLES `(i)H_(g) + l_(2)(g) hArr 2HI(g)` `Delta n_g =2-2=0` `(ii) N_(2)(g) + O_(2)(g) hArr 2NO(g)` `Delta n_(g) = 2-2 =0` |
|
| 47. |
Show that in the isothermal expansion of an ideal gas , DeltaU = 0 and DeltaH = 0 |
|
Answer» Solution :(a) For one moole of an ideal GAS, `C_(V) = ( dU)/( DT) ` or ` dU =C_(v) dT` For a finite change, `DELTAU = C_(v) DeltaT` For ISOTHERMAL process, `T=` constant so that`DeltaT =0`. Hence, `DeltaU = 0` (b)`DeltaH = DletaU + Delta(PV) =DeltaU+ Delta(RT) =DeltaU+ R DeltaT` But `DeltaU =0` ( proved above) and `DeltaT = 0` ( for isothermal process) , `:. DeltaH =0` |
|
| 48. |
Show that if the measurement of the uncertainty in the location of the particle is equal to its de Broglie wavelength,the minimum uncertinty in its velocity (/_\v) is equal to 1/4pi of its velocity(V) |
|
Answer» Solution :If, uncertainty in position`=/_\x=gamma`,the vale of uncertainty in velocity=`v/(4PI)` Heisenberg.s PRINCIPLE states that `/_\x./_\v.m=h/(4pi)`………….(i) de Broglie equation states that `gamma=h/(mv)`………..(2) `:.h=gamma.m.v`……..(3) `/_\x=h/(/_\v.4pi)`.............(4) `/_\X=(gammaxxm.v)/(/_\v.4pi.m)` `if/_\x=gamma` `/_\v=(CANCEL(x.m).v)/(cancel(x).4pi.cancel(m))=v/(4pi)`. |
|
| 49. |
Show that ground state energy of electron in hydrogen atom is equal to the first excited state energy of electron in He^(+) ion, assuming that their Rydberg's constants are equal |
|
Answer» SOLUTION :`E_(n) = - R_(H) (Z^(2))/(n^(2)) = - 13.60 (Z^(2))/(n^(2)) eV` For H-atom in the ground state, `Z = 1, n =1, :. E_(1) = -13.60eV` For `He^(+)` ion in the first EXCITED state, `Z = 2, n = 2, E_(2) = - 13.60 xx (2^(2))/(2^(2)) = - 13.6 eV` HENCE, the desired result is proved. |
|
| 50. |
Show that free energy (Delta G) as a criterion for spontaneous and non-spontaneous processes. |
|
Answer» Solution :Effect of temperature on the spontaneity of chemical reaction using Gibb's-Helmholtz equation : `Delta G = Delta H - T.Delta S` Case I : For an exothermic process, `Delta H` is negative. (a) If `Delta S` is positive, `Delta G` is always negative. The process is always spontaneous. (b) If `Delta S` is negative, (i) If the temperature is LOW such that `T Delta S lt Delta H`, then `Delta G` is negative. The process is spontaneous. (ii) If the temperature is high such that `T Delta S gt Delta H`, then `Delta G` is positive. The process is not spontaneous Thus, an exothermic process which is not spontaneous at a GIVEN temperature may become spontaneous at low TEMPERATURES. Case II : For an endothermic process, `Delta H` is positive. (a) If `Delta S` is negative, `Delta G` is always positive. `:.` The process is not spontaneous (b) If `Delta S` is positive, (i) If the temperature is low such that `TDelta S lt Delta H`, then `Delta G` is positive. The process is not spontaneous. (ii) If the temperature is high such that `T Delta S gt Delta H`, the `Delta G` is negative. The process is spontaenous. Thus, an endothermic process which is not spontaneous at a given temperature may become spontaneous at high temperatures. Case III :When any process attains equilibrium, `T Delta S gt Delta H, Delta G = 0` i.e., `Delta G = Delta H - T Delta S = 0`or `Delta H = T Delta S` at equilibrium For a chemical reaction, `A + B rarr C + D`, Gibb's ENERGY change is given by, `Delta G = SigmaG_(2) - Sigma G_(1)`. It is the difference between the total free energies of all the PRODUCTS and the total free energies of all the reactants. |
|