Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Standard entropies of X_(2), Y_(2) and XY_(3) are 60,40 and 50 kJ"mol"^(-1) respectively. For the reaction: 1/2 X_(2) + 3/2Y_(2)iff XY_(3), Delta H = -30 kJ to be at equilibrium, the temperature should be |
|
Answer» 500 K ` = 50 -1/2(60)-3/2(40) = -40J K^(-1) "mol"^(-1)` `T = (DeltaH^(@))/(DeltaS^(@)) = (-30 xx 10^(3) J"mol"^(-1))/(-40 J K^(-1)"mol"^(-1))` = 750 K |
|
| 2. |
Standard entropies of X_(2), Y_(2) andXY_(3) are 60,40 and 50JK^(-1)mol^(-1) respectively. For the reaction(1)/(2)X_(2) + (3)/(2)Y_(2) hArr XY_(3) , DeltaH= - 30 kJto be at equilibrium, the temperature should be |
|
Answer» 500K `DeltaS^(@) = SigmaS_(P)^(@) - Sigma _(R)^(@) = 50-( 30+60) = - 40 JK^(-1) MOL^(-1)` `DeltaG^(@) =DeltaH^(@) - T DeltaS^(@) ` . At equilibrium , `DeltaG^(@) = 0`. HENCE,`TDELTAS^(@) = DeltaH^(@)` or `T= (DeltaH^(@))/( DeltaS^(@)) = ( -30 xx 10^(3) J mol^(-1))/( -40JK^(-1) mol^(-1))= 750 K` |
|
| 3. |
Standar entropies of x_(2), y_(2) and xy_(3) are 60,40 and 50JK^(-1) mol^(-1) respectively for the reaction to be at equilibrium, the temperature should be (1)/(2) x_(2) +(3)/(2) y_(2) hArr xy_(3) Delta H= - 30kJ |
|
Answer» 750K `Delta S = Delta S_(xy) - ((1)/(2) sy_(2) + (3)/(2) sy_(2))` `50 - ((1)/(2) xx 60 + (3)/(2) xx 40) = -40JK^(-1) mol^(-1)` `Delta G = Delta H - T Delta S, Delta G = 0 = T = (Delta H)/(Delta S) = 750k` |
|
| 4. |
Standard entropies of x_(2) , y_(2) and xy_(3) are 60,40 and 50 "J/K mol"^(-1) respectively for the reaction (1)/(2) x_(2) + (3)/(2) y_(2) to xy_(3) , Delta H = - 30 "kJ" to be at equilibrium the temperature should be. |
|
Answer» 500 K `= 50 (30+ 60) = - 40 "J/K mol"` `Delta G = 0` `therefore Delta H = T Delta S therefore T = (-30 xx 10^(3) )/( - 40) = 750 K` |
|
| 5. |
Standard entropies of X_(2) , Y_(2) and XY_(3) are 60,40 and 50 "J/K mol"^(-1) respectively for the reaction (1)/(2) X_(2) + (3)/(2) Y_(2) to XY_(3) , Delta H = - 30 "kJ" to be at equilibrium the temperature should be. |
|
Answer» 750 K `Delta S = 50- (30 + 60)` `= - 40 J` For equilibrium `Delta G = 0 = Delta H - T Delta S` `T= (Delta H )/( Delta S ) = (- 30000)/( - 40)` `= 750 K` |
|
| 6. |
The standard molar engthalpy of vaporisation of benzene Delta_(vap)H^(@) at 353 K is 30.8 kJ mol^(-1). If the benzene vapours behave as an ideal gas, the change in internal energy of vaporisation of 78 g of benzene at 353 K in kJ mol^(-1) is |
|
Answer» `+37.56` |
|
| 7. |
Standard enthalpy of vaporisation Delta_("vap") H^(@) for water at100^(@)C is40.66 kJ mol^(-1) . The internal energy change of vaporisation of water at100^(@)C ( in kJmol^(-1)) is: |
|
Answer» `+37.56` `DeltaU =DeltaH - Deltan_(g)RT` `= 40.66xx 1000 J mol^(-1)-1 XX 8.314 JK^(-1) xx 373 K` `= 40660 - 3101 J mol^(-1)` `=37559 J mol^(-1)` `=37. 56 kJ mol^(-1)` |
|
| 8. |
Standard enthalpy of formation of H_(2(g)) , CI_(2(g)) and HCI_((g)) are 218, 121.68 and -92.31 KJ/mole respectively. Calculate the change in standard enthalpy for the reaction (1)/(2) H_(2(g)) + (1)/(2) CI_((g)) to HCI_((g)) |
|
Answer» `+431.99` KJ |
|
| 9. |
Standard enthalpy of ........... is not zero. |
| Answer» Answer :C | |
| 10. |
Standard enthalpy of formation is zero for ........... from following |
|
Answer» `F_(2(g)) ` |
|
| 11. |
Standard enthalpies of C_(6)H_(6)(l),H_(2)O(g) " and "CO_(2)(g) are respectively 11.7, -68.3 and -94 kCal. Calculate the calorific value of benzene. |
|
Answer» |
|
| 12. |
Standard electrode potential values Fe^(2+)//Fe,E^(@)=-0.44 V , Fe^(3+)//Fe^(2),E^(@)=0.77 v If Fe^(2+),Fe^(3+) and Fe blocks are kept together it will lead to |
|
Answer» increase in `Fe^(3+)` `Fe^(3+)+e^(-)rarrFe^(2+)` Thus the amount of `Fe^(3+)` will decrease |
|
| 13. |
Standard electrode potential values, (E^ɵ) for Al^(3+)//Alis -1.66 V and that of Tl^(3+) //Tlis +1.26 V. Predict about the formation of M^(3+) ion in solution and compare the electropositive character of the two metals. |
| Answer» Solution :Standard electrode potential values for two half cell reactions suggest that aluminium has high tendency to make `Al_((AQ))^(3+)`ions, whereas `Tl^(3+)`isnot only unstable in solution but is a powerful oxidising agent also. Thus `Tl^+`is more stable in solution than `Tl^(3+)` . Aluminium being able to FORM +3 ions easily, is more electropositive than thallium. | |
| 14. |
Standard electrodepotential values, E^(@) at Al^(3+)//Al is -1.66 V andthat of Tl^(3+)//Tl is +1.26 V. Predict about theformation of M^(3+)ions in solution and compare the electropositive characterof the twometals. |
| Answer» Solution :The negative valueof `E^(@)` for `Al^(3+)//Al` SUGGESTS that Al has a strongtendency to form`Al^(3+)`(aq) IONS. On the other hand,the positivevalue of`E^(@)`for `Tl^(3+)//Tl` suggests that Tl does not have HIGH tendency to form `Tl^(3+)` ions. Since Al can form `Al^(3+)` ions more easily than `Tl` does to form `Tl^(3+)` ions, THEREFORE Al ismore ELECTRONEGATIVITY than Tl. | |
| 15. |
Standard electrode potential data are used for understanding the stability of an oxidant in a redox titration . Some half reactions and their standard potentials are given below: MnO_4^(-) (aq) +8H^(+)(aq)+5e^(-)rarrMn^(2+)(aq)+4H_2O(l) ""E^@=1.51V Cr_2O_7^(2-) (aq)+14H^(+)(aq)+6e^(-) rarr 2Cr^(3+)(aq)+7H_2O(l) ""E^@=1.38V Fe^(3+)(aq)+e^(-) rarr Fe^(2+)(aq) ""E^@=0.77V Cl_2(g)+2e^(-) rarr2Cl^(-) (aq)""E^@=1.40V Identify the only incorrect statement regarding the quantitative estirnation of aqueous Fe(NO_3)_2. |
|
Answer» `MnO_4^(-)` can be used inaqueous HCL `2KMnO_4+16H^(+)+16Cl^(-)rarr 2Mn^(2+)+8H_2O+5Cl_2` The corresponding CELL is `Pt|Cl_2(g)|Cl^(-)(aq)||MnO_(4)^(-)(aq), Mn^(2+),H^(+)|Pt` `E_("cell")^@=E^@` (cathode) `-E^@` (anode) = 1.51 - 1.40 = 0.11 V Since , `E_("cell")^@` is +ve , the above reaction is feasible . Therefore , `MnO_4^(-)` will oxidise both `Fe^(2+) and Cl^(-)` IONS in aqueous medium . THUS, quantitative estimation of `Fe(NO_3)_2` is not suitable. |
|
| 16. |
Standar enthalpy of formation of threecombustible isomers compound A,B,C are -20Kcal/mol ,30kcal /mol and 40kcal /mol respectively then what willbe order of their enthalpy of combustion ? |
|
Answer» `DeltaH_(C)^(@)[A]ltDeltaH_(c)^(@)[B]lt DeltaH_(c)^(@)[C]` |
|
| 17. |
Stand electrode potential of three metals X,Y and Z are -1.2 V + 0.5 V and -3.0 V respecitvelyThe reducing power of these metals will be |
|
Answer» `XgtYgtZ` `Z(-30 V)gtX(-1.2 V)GTY(+0.54V)` |
|
| 18. |
Stacking of hexagonal close packed layers give rise to |
|
Answer» HCP structure |
|
| 19. |
Stacking of square close packed layers give rise to |
|
Answer» BCC STRUCTURE |
|
| 20. |
Stable from of C may be represented by the formula : |
| Answer» SOLUTION :Is the stable FORM of element C. It is actually `Cl_(2).` | |
| 22. |
Stable form of C may be represented by the formula : |
|
Answer» C In the ELECTRONIC configuration of C INDICATE chlorine. Its stable FORM is dichlorine`(Cl_(2))`i.e. `C_(2)` |
|
| 23. |
Stable form of A may be represented by the formula : |
|
Answer» A In the given ELECTRONIC configuration A represents NOBLE gas As the octet is COMPLETE. A is neon which has 10 ATOMIC number. |
|
| 24. |
Stability order of the following species ? |
|
Answer» `IgtIIgtIII` |
|
| 25. |
Stability of the species Li_(2), Li_(2)^(-) and Li_(2)^(+) increases in the order of : |
|
Answer» `Li_(2) lt Li_(2) lt Li_(2)^(+)` BOND ORDER ` = (1)/(2) (4-2) = 1 ` `Li_(2) ^(+) sigma (1s)^(2) sigma^(**) (1s)^(2) sigma (2s)^(1)` , Bond order`= (1)/(2) (3-2) = (1)/(2)` `Li_(2)^(-) sigma (1s)^(2) sigma^(**) (1s)^(2) sigma (2s)^(2)(2s)^(1) `, Bond order = `(1)/(2) (4-3) = (1)/(2)` `LI_(2)^(-)` is less STABLE than ` Li_(2)^(+)` because ` Li_(2)^(-)` has more number of electrons in the antibonding molecular orbitals . Otherwisegreater the bond order , greater is the stability . HENCE , the correct order is `Li_(2)^(-) lt Li_(2)^(+) lt Li_(2) ` . |
|
| 26. |
Stability of monovalent and trivalent cations of Ga. In lie the following sequence |
|
Answer» `Ga^(3+) LT In^(3+) gt TL^(3+)` |
|
| 27. |
Stability of iso-butylene can be best explaned by |
|
Answer» INDUCTIVE effect |
|
| 28. |
"Stability of carbocation depends upon the electron releasing inductive effect of groups adjacent to positively charged carbon atom, involvement of neighbouring groups in hyperconjugation and resonance". Write structures of various carbocations that can be obtained from 2-methylbutane. Arrange the carbocations in order of increasing stability. |
|
Answer» SOLUTION :2-Methylbutane has four DIFFERENT sets of equivalent hydrogen atoms marked as a, b, c and d. `{:(overset(a)(C)H_(3)-overset(b)(C)H-overset(c)(C)H_(2)-overset(d)(C)H_(3)),("|"),(""underset(a)(C)H_(3)):}` Removal of one hydrogen form any of these equivalent sets of hydrogens gives four different carbocations I, II, III and IV as shown below : 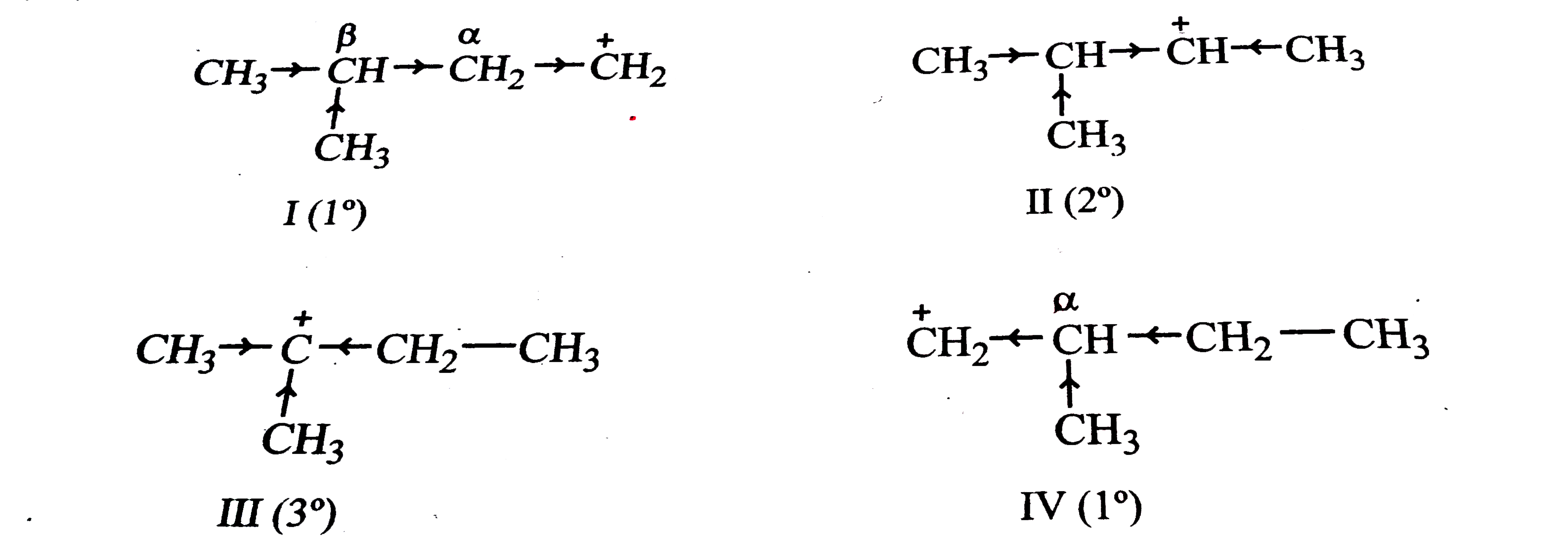 Now stability of carbocations decreases in the order : `3^(@) gt 2^(@) gt 1^(@)`. Since III being the `3^(@)` carbocation, it is the most STABLE. This is followed by carbocation (II) which is `2^(@)`. Out of the other two `1^(@)` carbocations, carbocation (IV) has an electron-donating `CH_(3)` group at `alpha`-carbon while carbocation (I) has a `CH_(3)` group at `beta`-carbon. Since +I effect decreases with distance, therefore, carbocation (IV) is more stable than carbocation (I). Thus, the overall stability of these four carbocations increases in the order : `I lt IV lt II lt III`. |
|
| 29. |
"Stability of carbocation depends upon the electron releasing inductive effect of groups adjacent to positively charged carbon atom, involvement of neighbouring groups in hyperconjugation and resonance". Draw the possible resonance structures for CH_(3)-underset(..)overset(..)(O)-overset(+)(C)H_(2) and predict which of the structures is more stable. Give reason for your answer. |
Answer» Solution :The given CARBOCATION has two RESONANCE structures, i.e., I and II. 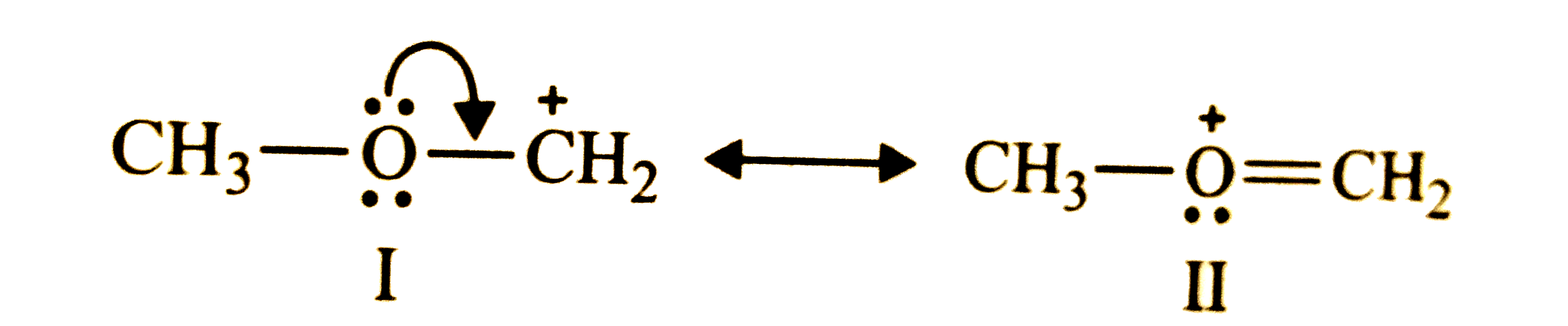 STRUCTURE (II) is more stable since both the carbon ATOMS and the oxygen atom have an octet of electrons. |
|
| 30. |
Stability of alkyl carbocations can be explained by |
|
Answer» INDUCTIVE EFFECT only |
|
| 31. |
Stability is more for the intermediate |
|
Answer» `C_6H_5overset(BAR"..")"C"H_2` |
|
| 32. |
SRP values of lanthanides lies between |
|
Answer» `-2.2` to `-2.4 V` |
|
| 33. |
sqrtV of two particles A and B are plotted against de-Broglie wavelengths. where V is the potential onthe particles. Which of the following relation is correct about the mass of particles? |
|
Answer» `m_A = m_B` |
|
| 34. |
Spodoptol, a sex attractant, produced by a female fall armyworm moth, can be prepared as follows. The structure of Spodoptol is (pK_a : terminal alkynes ~ 25, alcohols ~ 17) |
|
Answer» `n-C_4H_9-O-CH_2-(CH_2)_7-HC=CH_2` |
|
| 35. |
Spodumene is the silicate mineral of ___________ |
|
Answer» Beryllium |
|
| 36. |
Splitting of spectral lines in an electric field is called: |
|
Answer» COMPTON effect |
|
| 37. |
Splitting of spectral lines in an electric field is called |
|
Answer» Zeeman effect |
|
| 38. |
Splitting of spectral lines in an electric field is called……………. |
|
Answer» ZEEMAN EFFECT |
|
| 39. |
Splitting of signals is caused by |
|
Answer» PROTON |
|
| 40. |
Split the following redox reaction in to the oxidation and reduction half rections (a) 2K(s) +CI_(2)(g)rarrKCI(s) |
| Answer» Solution :`{:((a)K(s) toK^(+)(g)+e^(-)"["xx2),(Cl_(2)(g)+2E^(-)to2Cl^(-)(g)),(UL(K^(+)(g)+Cl^(-)(g)toKCl(s)"]"xx2)),(2K(s)+Cl_(2)(g)to2KCl(s)),((b) Al(s) to Al^(3+)(aq)+3e^(-)"["xx2),(ul(Cu^(2+)(aq)+2e^(-) to Cu(s)(s)"]"xx3)),(2Al(s)+3Cu^(2+)(aq) to 2Al^(3+)(g)+3Cu(s)):}` | |
| 41. |
Spinel is the mineral with the formula _________ |
|
Answer» |
|
| 43. |
Spin isomerism is shown by |
|
Answer» dichlorobenzene |
|
| 44. |
Spin m ultiplicity value of oxygen element is |
|
Answer» 2 |
|
| 45. |
Spin angular momentum of an electron has no analog in classical mechanics. However, it turns out that the treatment of spin angular momentum is closely analogous to the treatment of orbital angular momentum. Spin angular momentum = sqrt(s(s+1)h) Orbital angular momentum = sqrt(l(l+1)h) Total spin of an atom or ion is a multiple of (1)/(2). Spin multiplicity is a factor to confirm the electronic configuration of an atom or ion. Spin multiplicity (2 sum s +1) The orbital angular momentum for a 2p-electron is : |
|
Answer»
|
|
| 46. |
Spin angular momentum of an electron has no analog in classical mechanics. However, it turns out that the treatment of spin angular momentum is closely analogous to the treatment of orbital angular momentum. Spin angular momentum = sqrt(s(s+1)h) Orbital angular momentum = sqrt(l(l+1)h) Total spin of an atom or ion is a multiple of (1)/(2). Spin multiplicity is a factor to confirm the electronic configuration of an atom or ion. Spin multiplicity (2 sum s +1) Total spin of Mn^(2+) = (Z = 25) ion will be : |
| Answer» Answer :C | |
| 47. |
Specify the coordinate geometry around and hyridilization present about the central atom in BF_(3) is: |
|
Answer» `N`: Tetrahedral, `sp^(3)`, `B`: Tetrahedral, `sp^(3)` |
|
| 48. |
Specific test for the detection of H_(2)O_(2) is |
|
Answer» Reaction with acidified `TiO_2` `TiO_2 + 2H_2SO_4 to TI(SO_4)_2` ` + 2H_2O overset(H_2O_2) to underset("PE ti tan ic acid")(H_2TiO_4 + 2H_2SO_4)` |
|
| 49. |
Specific reagent for dehydrohalogenation of alkyl halides to get alkenes is |
|
Answer» AQUEOUS KOH |
|
| 50. |
Specific heat ofan elementary gas is found to be 0.313 J at constant volume. If the molar mass of the gasis 40 g mol^(-1) , what is the atomicity of the gas ? |
|
Answer» `:. C_(p) //C_(v) = 20.83 // 12.52 = 1.66` |
|