Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The hetero elements in organic compound are nitrogen, halogen, sulphur and phosphorousd, Nitrogen is estimated by Duma's method (or) Kjeldahl's method. The other hetero elements are estimated using Carius method.0.25 g of an organic compound gave 22.4 ml. of N_(2) at STP by Duma's method. The percentage of Nitrogen in the compound is |
|
Answer» `15%` `=(22.4)/(0.25).(28)/(22400).100=11.2%` |
|
| 2. |
The hetero elements in organic compound are nitrogen, halogen, sulphur and phosphorousd, Nitrogen is estimated by Duma's method (or) Kjeldahl's method. The other hetero elements are estimated using Carius method.In the estimation of halogen by Carius method, which of the following is carried out |
|
Answer» Oxidation of compound by CONC, `H_(2)SO_(4)` |
|
| 3. |
The Henry's law constant for two gases A and B are x and y respectively.The ratio of mole fractions of A to B is 0.2 The ratio of mole fraction of B and A dissolved in water will be |
|
Answer» <P>`(2x )/(y)` `P _(A) =x (x _(A)) _("in solution")""...(1)` `P _(B) =y (x _(B)) _("in solution")""...(2)` `((x_(B))/(x_(A))) _(" in solution") = (P_(B))/(P _(A))XX x/y = (x_(B))/(x _(A))xx x/y= (1)/(0.2) xx x/y = (5x)/(y)` |
|
| 4. |
The Henry's law constants for two gases A and B are x and y respectively. The ratio of mole fractions of A to B is 0.2. The ratio of mole fraction of B and A dissolved in water will be |
|
Answer» `(2x)/y` `((x_B)/(x_A))_("in solution")= ?` `P_A = x(x_A)_("in solution")""…(i)` `P_B = y(x_(B))_("in solution")""…(2)` `((x_B)/(x_A))_("in solution") =(P_B)/(P_A) XX x/y` `= (x_B)/(x_A) xx x/y =1/(0.2)xx x/y = (5x)/y` |
|
| 5. |
The Henry's law constant for the solubility of Nitrogen gas iin water at 350 Kis 8xx10^4 atm. The mole fraction of nitrogen in air is 0.5 . The number of moles of Nitrogen from air dissolved in 10 moles of water at 350 K and 4 atm pressure is …………….... . |
|
Answer» `4xx10^(-4)` `(x_(N_(2)))_("in air")= 0.5` TOTAL pressure = 4 atm Partial pressure of nitrogen = Mole fraction`xx` Total pressure =`0.5xx4 = 2` `(P_(N_(2))) = K_(H)xx "Mole fraction of" N_(2) "in solution"` `2=8xx10^(4)xx("NUMBER of moles of nitrogen")/("Total number of moles")` `(10+"No. of moles of" N_(2))/("No. of moles of" N_(2)) = (8xx10^(4))/(2)` `(10)/("No. of moles of" N_(2))+1 = 4xx10^(4)` `(10)/("No. of moles of" N_(2)) = 40000-1` `therefore` No. of moles of `N_(2) = (10)/(39999) = 2.5xx10^(-4)` |
|
| 6. |
The heavier members of 13 and 14 groups besides the group oxidation state also show another oxidation state which is two unit less than the group OS. Down the group decreasing, the stability state of higher OS increasing , and that of lower OS increasing. The concept which is commonly called inert pair effect has been used to explain many physical and chemical properties of the element of these groups.Which among the following is the strongest reducing agent ? |
|
Answer» GACL |
|
| 7. |
The heavier members of 13 and 14 groups besides the group oxidation state also show another oxidation state which is two unit less than the group OS. Down the group decreasing, the stability state of higher OS increasing , and that of lower OS increasing. The concept which is commonly called inert pair effect has been used to explain many physical and chemical properties of the element of these groups.Heavier members of group 13 exhibit oxidation state. |
|
Answer» `+3` only |
|
| 8. |
The heavier elements of groups 13 and 14 besides the group oxidation state exhibit another oxidationstate which is two units lower than the group oxidationstate and the stability of this lower oxidation state increases down the group. This conceptwhich is commonly called inert pair effect has been used to explainmany physical and chemical properties of the elementof these groups. The oxideswhich is the strongest acid is |
|
Answer» `Tl_(2)O_(3)` |
|
| 9. |
The heavier members of 13 and 14 groups besides the group oxidation state also show another oxidation state which is two unit less than the group OS. Down the group decreasing, the stability state of higher OS increasing , and that of lower OS increasing. The concept which is commonly called inert pair effect has been used to explain many physical and chemical properties of the element of these groups.Which among the following is the strongest oxidising agent ? |
|
Answer» `SiO_(2)` |
|
| 10. |
The heavier elements of groups 13 and 14 besides the group oxidation state exhibit another oxidationstate which is two units lower than the group oxidationstate and the stability of this lower oxidation state increases down the group. This conceptwhich is commonly called inert pair effect has been used to explainmany physical and chemical properties of the elementof these groups. Which of the following statements is incorrect ? |
|
Answer» CO is used as a reducing agent |
|
| 11. |
The heavier member of lanthanoid series metal react with following non-metal. ltBrgt The summation of atomicity of compound X, Y and Z is: |
Answer» 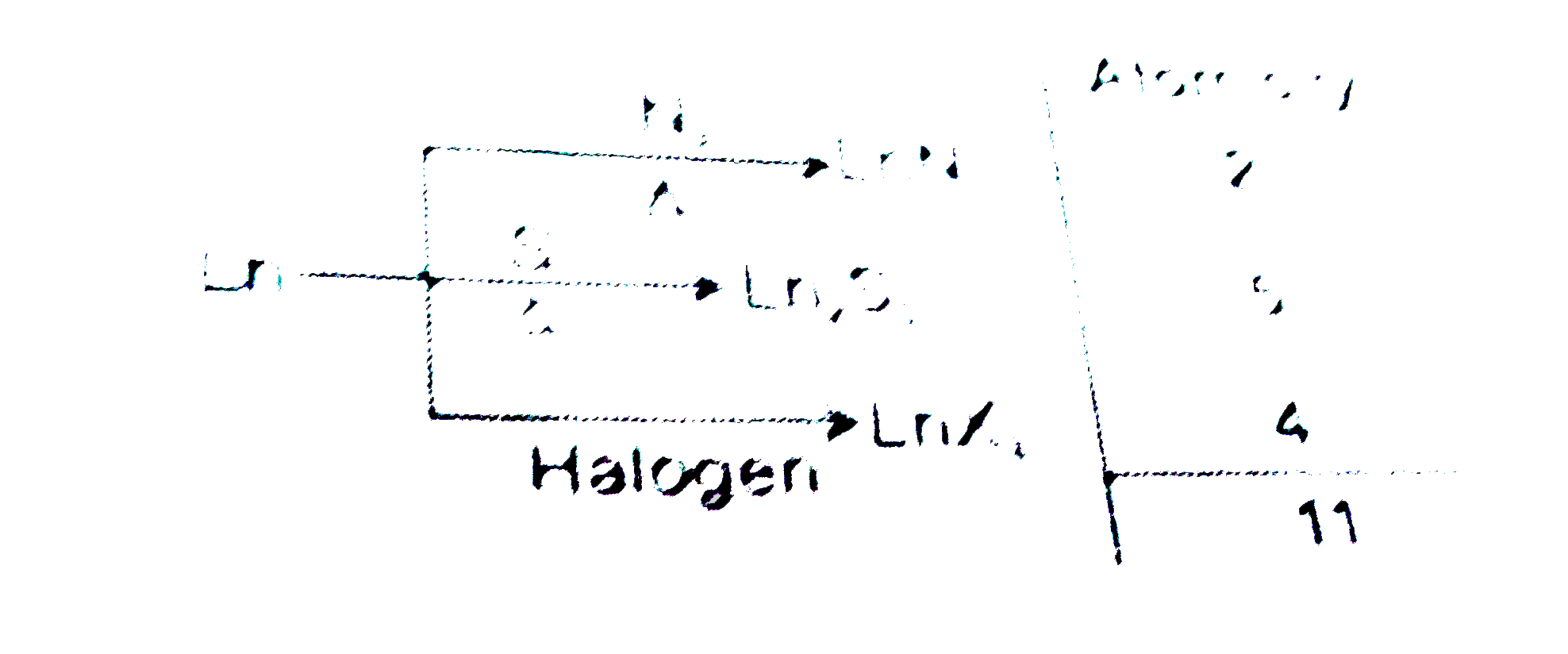
|
|
| 12. |
The heavier elements of groups 13 and 14 besides the group oxidation state exhibit another oxidationstate which is two units lower than the group oxidationstate and the stability of this lower oxidation state increases down the group. This conceptwhich is commonly called inert pair effect has been used to explainmany physical and chemical properties of the elementof these groups. Element of group 13 |
|
Answer» exhibit oxidationstate of `+3` only |
|
| 13. |
The heavier elements of groups 13 and 14 besides the group oxidation state exhibit another oxidationstate which is two units lower than the group oxidationstate and the stability of this lower oxidation state increases down the group. This conceptwhich is commonly called inert pair effect has been used to explainmany physical and chemical properties of the elementof these groups.ltbr. Which among the following is the strongest oxidising agent ? |
|
Answer» `SnO_(2)` |
|
| 14. |
The heavier elements of groups 13 and 14 besides the group oxidation state exhibit another oxidationstate which is two units lower than the group oxidationstate and the stability of this lower oxidation state increases down the group. This conceptwhich is commonly called inert pair effect has been used to explainmany physical and chemical properties of the elementof these groups. The strongest reductant among the following is |
|
Answer» `GeCl_(2)` |
|
| 15. |
The heats of neutralisation of HCl with NH_4OH and that of NaOH with CH_3COOH are repectively - 51.4 and - 50.6 KJ eq^(-1). The heat of neutralisation of acetic acid with NH_4OH will be |
|
Answer» `-44.6 KJ EQ ^(-1)` |
|
| 16. |
The heats of formation of CO_2, H_2O and C_2H_4 are -94.05, -68.2 and 12.6 K.Cals/ mole, respectively. The heat of combustion of ethylene is |
|
Answer» 337 K.Cal |
|
| 17. |
The heats of formation of CO_(2(g)) , H_2O_((l)) and CH_(4(g)) are respectively –394 kJ mol^(-1), -286 kJ mol^(-1) and -76 kJ mol^(-1). Calculate the heat of combustion of methane. |
|
Answer» Solution :`C _("graphite") + O _(2 (G)) to CO _(2 (g)) , DELTA H _(1) =- 394 kJ` `H _(2 (s)) + (1)/(2) O _(2 (s)) to H _(2) O _((l)) , Delta H _(2) =- 286 kJ` `C _("graphite") + 2H _(2 (g)) to CH _(4(g)) , Delta H _(4 (g)) , Delta H _(3) =- 76 kJ` The requird equation is, `CH _(4 (g)) + 2O _(2 (*g)) to CO _(2 (g)) + 2H _(2) O _((l)) : Delta H =?` `Delta H = Delta H _(1) + 2 Delta H _(2) - Delta H _(3) =- 394+ 2 (-286) - (-76)` The heat of combustion of methane `= - 890 kJ mol^(-1).` |
|
| 18. |
The heats of combustion of hydrogen and cyclohexene are -241 kJ mol^(-1) and -3800 kJ mol^(-1).The heat of hydrogenation of cyclohexene is -121kJ mol^(-1). Calculate the heat of combustion of cyclohexane. |
|
Answer» |
|
| 19. |
The heats of combustion of ethane, ethene and acetylene are -341.1 kcal, -330 kcal and -310.9 kCal mol^(-1) respectively. The best fuel among them is |
|
Answer» ETHANE |
|
| 20. |
The heats of combustion of carbon hydrogen and acetylene are -394kJ, -286kJ and -1301 kJ respectively. Calculate heat of formation of C_(2)H_(2) |
|
Answer» 621 kJ `Delta H` of reaction `= SIGMA` Heat of COMBUSTION of reactants `- Sigma` Heat of formation of products `Delta H= 2 (-394) + (-286) - (-1301) = 227KJ` |
|
| 21. |
The heats of combustion of carbon and carbon monoxide are -393.5 and - 283.5 kJ mol^(-1) respectively. The heat of formation ( in kJ ) of carbon monoxideper mole is : |
|
Answer» `676.5` `CO(g) + (1)/(2) O_(2)(g)rarr CO_(2)(g), DeltaH= - 283.5 kJ mol^(-1)`…(II) Aim `: C(s) + (1)/(2)O_(2)(g)rarr CO(g)` Eqn. (i) -Eqn. (ii) GIVES required eqn. with `DeltaH =- 393.5 - ( - 283.5) kJ = -110 kJ` |
|
| 22. |
The heats of combustion of carbon and carbon monoxide are -393.5 and -283.5 kJ "mol"^(-1), respectively. The heat of formation (in kJ) of carbon monoxide per mole is …..... |
|
Answer» `-110.5` `CO_((g)) + 1/2O_(2(g)) to CO_(2(g)) DeltaH`= - 283.5 …(2) HEAT of FORMATION of `CO_((g))` `rArr` (1)-(2) `C_((s)) + 1/2O_(2(g)) to CO_(2(g)) DeltaH`=-110.0 KJ |
|
| 23. |
The heats of combustion of carbon and carbon monoxide are -393.5 and -283.5 kJ"mol"^(-1) respectively. The heat of formation (in kJ) of carbon monoxide per mole is |
|
Answer» `110.5` (II) `CO(g) +1/2 O_(2)(g) to CO_(2)(g)DeltaH = -283.5 kJ"mol"^(-1)` SUBTRACTING equation (ii) from equation (i) `C(s) + 1/2O_(2)(g) to CO(g)= - 110.0 kJ"mol"^(-1)`. |
|
| 24. |
The heats of absorption in physisorption (or physical absorption ) lies in the range ( in kJ/mole) |
|
Answer» 40-400 |
|
| 25. |
The heat required to raise the temperature of a body by 1 K is called ____ |
| Answer» SOLUTION :THERMAL CAPACITY | |
| 26. |
The heat produced from the complete combustion of 1 g of fuel or food is called its "…............." |
| Answer» SOLUTION :CALORIFIC VALUE | |
| 27. |
The heat of transition of S_(alpha) into S_(beta) is +2.5 kJ. The heat of combustion of monoclinic sulphur is - 300 kJ mol^(-1). Calculate heat of combustion of rhombic sulphur(S_(alpha)). |
|
Answer» Solution :`S _(ALPHA ) to S _(beta) , Delta H = + 2.5 kJ and S _(beta) + O _(2 (G)) to SO _(2 (g)) , Delta H =- 300 kJ` Adding these TWO, the heat of combustion of `Salpha`is OBTAINED. `S _(alpha ) + O _(2 (g)) to SO_(2 (g)) , Delta H =- 297.5KJ.` |
|
| 28. |
The heat of reaction for an endothermic reaction at constant volume in equilibrium is 1200 cal more than al constant pressure at 300K, then |
|
Answer» <P>`Deltan_((g))= -2` `DeltaU-DeltaH=1200=Delta n_((g))RT` `Delta n_((g))=(-1200)/(2 xx 300)= -2 implies (Kp)/(Kc)=(RT)^(-2)=(0.0821 xx 300)^(-2)=1.648 xx 10^(-3)` |
|
| 29. |
The heat of reaction at constant volume and temperature is represented by |
|
Answer» <P>`Delta E` |
|
| 30. |
The heat of neutralization of a strong acid and strong base is around _____ |
| Answer» SOLUTION :`-57.32` KJ | |
| 31. |
The heat of neutralisation of strong base and strong acid is - 57.0 KJ. The heat released when 0.5 mole of HNO_3 solution is added to 0.20 moles of NaOH solution is |
|
Answer» 57.0 KJ |
|
| 32. |
The heat of neutralisation of strong acid and strong base is ………….. |
|
Answer» `+57.32KJ` |
|
| 33. |
The heat of neutralisation of a strong base and a strong acid is 13.7 kcal. The heat released when 0.6 mole HCI solution is added to 0.25 mole of NaOH is |
|
Answer» 3.425 KCAL `= 0.25 XX 13.7 = 3.425` kcal. |
|
| 34. |
The heat of neutralisation is maximum when |
|
Answer» SODIUM hydroxide is neutralised by acetic ACID |
|
| 35. |
The heat of ionisation of weak acid and a weak base are .x. and .y. k.cals. Then the heat of neutralisation of that weak acid and weak base is ....K.cal. |
|
Answer» `13.7` |
|
| 36. |
The heat of formation of the oxides of third period are given in "kJ mol"^(-1) {:(,Na_(2)O,MgO,Al_(2)O_(3),SiO_(2),P_(4)O_(10),SO_(3),Cl_(2)O_(3)),(,-416,-602,-911,-2984,-395,+250):} Arrange these oxides in increasing order of stability. |
|
Answer» Solution :More negative the value of heat of formation, lesser the amount of ENERGY left with the OXIDE. In this way the potential energy of the oxide is low and is more STABLE. `P_(4)O_(10) gt Al_(2)O_(3) gt SiO_(2) gt MgO gt Na_(2)O gt SO_(3) gt Cl_(2)O_(7)` |
|
| 37. |
The heat of formation of CO and CO_(2), are -26.4 kcal and -94 kcal, respectively. Heat of combustion of carbon monoxide will be ........... |
|
Answer» `+26.4Kcal` `DeltaH_(C)^(0)(CO)=-94KCal-[-26KCal+0]` `Delta_(C)^(CO)`=-94KCal+26.4KCal `Delta_(C)^(0)(CO)`=-67.4KCal |
|
| 38. |
The heat of formation of CO_(2) is -407KJ//mol . The energy required for the process 3CO_(2)(g)rarr3C(g)+2O_(3)(g) is |
|
Answer» LESS than `1221KJ` `DeltaH=3DeltaH_("ATOMIZATION" )C(s)rarrC(g))+2Delta_(f)^(@)(O_(3)(g))-3DeltaH_(f)^(@)` `(CO_(2))(g))` `DeltaH=3DeltaH_("atomiztion")C(s)rarrC(g))2DeltaH_(f)^(@)(O_(3)(g))+3xx407` As ENERGY will be REQUIRED for the processes `C(s)rarrC(g)` and `3O_(2)(g)rarr2O_(3)(g)` `:. DeltaH` will be more than `1221KJ` . |
|
| 39. |
The heat of formation of C_(2)H_(5)OH(l) is -66 " kcal"//"mole". The heat of combustion of CH_(3)OCH_(3)(g) is -348 " kcal"//"mole",DeltaH_(f) for H_(2)O(l) and CO_(2)(g) are -68 kcal//"mole" and -94 kcal//"mole" respectively,. Then the DeltaH for the isomerisation reaction, C_(2)H_(5)OH(l)toCH_(3)OCH(g), and DeltaE for the same are : (Take: T_("surr")=298 K) |
|
Answer» `DeltaH=18 KCAL//"MOLE",DeltaE=17.301 kcal//"mole"` |
|
| 40. |
The heat of formation of CO and CO_(2) are -26.4kCaland -94 kCal, respectively. Heat of combustion of carbon monoxide will be |
|
Answer» Solution :`C+1//2O_2 to CO "" DeltaH`=-26.4 K.cal …(1) `C+O_2 to CO_2 "" DeltaH`=-94 K.cal …(2) Heat of COMBUSTION of CO is `C+1//2O_2 to CO_2 "" DeltaH`=? EQUATION (1) is reversed . `{:(CO to cancelC+1/cancel2O_2 "" DeltaH="+26.4 K.cal"),(cancelC+cancelO_2to CO_2 "" DeltaH="-94 K.cal" """[Add equation (2)]"):}/(C_((g))+1//2O_(2(g)) to CO_(2(g))""DeltaH="-94 +26.4")` `DeltaH`=-67.6 K.cal |
|
| 41. |
The heat of dissociation of H_(20 is 435kJ mol^(-1). If C_((s)) rarr C_((g)), DeltaH=720kJ mol^(-1), calculate the bond energy per mole of the C-H bond in CH_(4) molecule . (DeltaH_(f) fo r CH_(4)=-75kJ mol^(-1)) |
|
Answer» |
|
| 42. |
The heat of combustion of solid naphthalene. (C_(10)H_(10)) at constant volume was -4984kJmol^(-1) " at " 298K. Calculate the value of enthalpy change. Given: C_(10)H_(8_((s)))+120_(2_((g)))rarr10CO_(2_((g)))+4H_(2)O_((l)),DeltaU=-4984 kJ.mol^(-1) DeltaU=-4984 kJmol^(-1),R=8.314JK^(-1)mol^(-) T=298K |
|
Answer» SOLUTION :`DELTAN=10-12=-2mol` `DeltaH=DeltaU+Rt(Deltan)` `=-4984xx10^(3)J+8.314Jk^(-1)MOL^(-1)xx298kxx(-2)mol.` `=-4984000J-4955.144J =-4988955.144J` `DeltaH=-4988.955kJ` |
|
| 43. |
The heat of combustion of solid benzoic acid at constant volume is -321.30 KJ at 27^(@) C . The heat of combustion at constant pressure is |
|
Answer» `-321.30 - 300 `R |
|
| 44. |
The heat of combustion of naphthalene (C_(10)H_(8)(s)) at constant volume was found to be - 5133 k J mol^(-1) . Calculatethe value of enthalpy change. |
|
Answer» `DELTAH = DeltaU + Deltan_(g) RT = - 5132 kJ mol^(-1) + ( - 2 mol) ( 8.314 XX 10^(-3)kJK^(-1) mol^(-1)) ( 298K)` `= -5133kJ mol^(-1)-5 kJ mol^(-1) = - 5138 kJ mol^(-1)` |
|
| 45. |
The heat of combustion of hydrocarbon C_(x)H_(y) is ''a'' calories and heat of formation of CO_(2) and H_(2)O are ''b'' and 'c' calories respectively then the heat of formation of hydrocarbon C_(8)H is (in calories) |
|
Answer» `XA + YC -B` `Delta H_("comb") = x Delta H_(f_(CO_(2))) + (y)/(2) Delta H_(f_(H_(2)O)) - Delta H_(f_(C_(x)H_(y)))` `a= xb + (y)/(2) C- Delta H_(f_(C_(x)H_(y)))` `Delta H_(f_(C_(x)H_(y))) = xb + (yc)/(2) - a` |
|
| 46. |
The heat of combustion of carbon to CO_(2) is -393.5 kJ// mol . The heat released uponformation of 35.2 g ofCO_(2) from carbon and oxygen gas is |
|
Answer» `+ 315kJ` For formation of44g , `DeltaH= -393.5 kJ` `:. `For formation of`35.2 g `of`CO_(2)` `DeltaH= - ( 393.5)/( 544) xx35.2 =- 314.8 =-315 kJ` |
|
| 47. |
The heat of combustion of CH_(4)(g) at constant volume is measured in a bomb calorimeter at 298 . 2 Kand found to be -885389 J // mol . Find the value of enthalpy change. |
|
Answer» |
|
| 48. |
The heat of combustion of carbon to CO_(2) is -393.5 "kJ/mol". The heat released upon formation of 35.2 g of CO_2 from carbon and oxygen gas is : |
|
Answer» `-630 kJ` `C_((s)) + O_(2(g)) toCO_(2(g))`, `Delta_(F) H= - 393.5 "kJ MOL"^(-1)` (1 MOLE = 44 g) Heat RELEASED on formation of `44 gCO_2` `=393.5 "kJ mol"^(-1)` `= (393.5 "kJ mol"^(-1) )/( 44g ) xx 35.2 g` `+315 kJ` |
|
| 49. |
The heat of combustion of carbon to CO_(2) is -393.5 kJ/mol. The heat released for the formation of 35.2 g of CO_(2) from carbon and oxygen gas is: |
|
Answer» `-630` kJ Heat released for forming 1 mol (44G) of `CO_(2) = -393.5 kJ ` Heat released for formation of 35.2 g of `CO_(2) = (-393.5 xx 35.2)/(44)` ` = -314.8` or = `- 315 kJ` |
|
| 50. |
Enthalpy of combustion of carbon to CO_(2) is -393.5 kJ mol^(-1). Calculate the heat released upon formation of 35.2 g of CO_(2) from carbon and dioxygen gas. |
|
Answer» `+315kJ` |
|