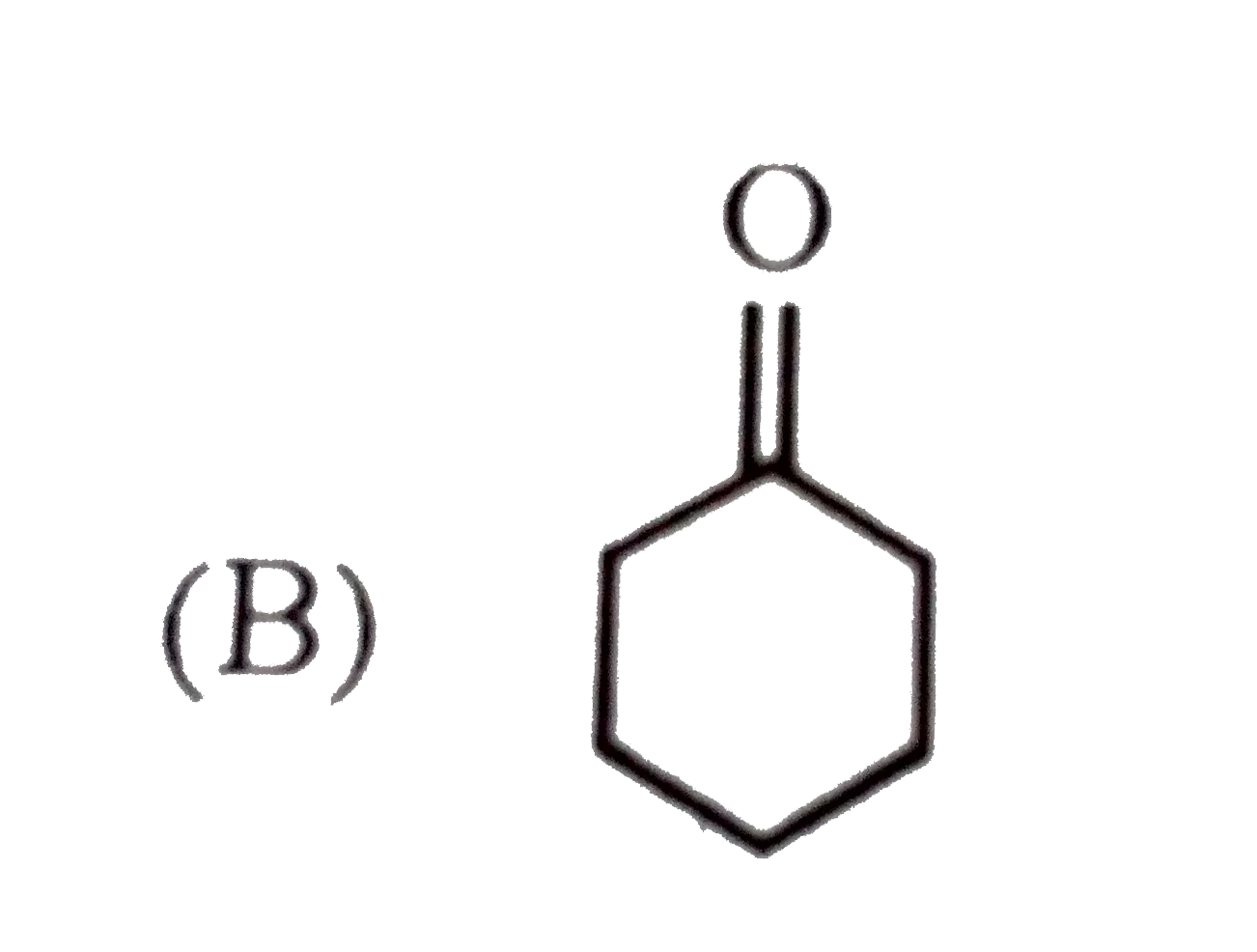Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What is Boyle.s temperature ? Give its relation with value of Z. |
|
Answer» Solution :Boyle point : The temperature at which a real GAS obeys ideal gas law over an appreciable range of pressure is called Boyle temperature or Boyle point. Boyle point of a gas depends upon its nature. Their Boyle point, real gases show positive deviations from IDEALITY and Z values are greater than one `(Z gt 1)`. ATTRACTION forces are weak. Below Boyle temperature real gases first show decrease in Z value with increasing pressure, which REACHES as minimum value. |
|
| 2. |
Which of the following has correct increasing basic strenght ? |
|
Answer» `MgO LT BEO lt CaO lt BAO` |
|
| 3. |
Which of the following ions is the most stable? |
|
Answer» `Sn^(2+)` |
|
| 4. |
The wavelength of a radiation emitted by a sodium lamp is 600 nm. Find the frequency ? |
|
Answer» Solution :GIVEN `lambda = 600nm` Frequency `V = ?` `"We know that V"= (C)/(lambda)=(3.0xx10^(8)ms^(-1))/(600xx10^(-9)m)=5XX10^(14)S^(-1)` |
|
| 5. |
Which of the following species can function both as oxidizing as well as reducing agent? |
|
Answer» `CL^(-)` |
|
| 6. |
Write the IUPAC names of the following compounds. (i) (C Cl_(3))_(3)C Cl (ii) (CH_(3))_(3)C CH=C(Cl)C_(6)H_(4)I-p. |
|
Answer»
 (ii) `(CH_(3))_(3)C CH=C(Cl)C_(6)H_(4)I-p`,  . .
|
|
| 7. |
Which of the following compounds contain (s) no covalent bond (s) ? KCl, PH_(3),O_(2), B_(2), H_(6), H_(2)SO_(4) |
|
Answer» `KCl,B_(2)H_(6), PH_(3)` compounds contain covalent bond . |
|
| 8. |
What are metallic hydrides? Explain about it. |
|
Answer» Solution :(i) METALLIC hydrides are obtained by hydrogenation of metals and alloys in which hydrogen occupies the interstitial sites (voids). Hence, they are called interstitial hydrides. (ii) The hydrides show properties similar to parent metals and hence they are also KNOWN as metallic hydrides. (iii) They are mostly non-stoichiometric with variable composition (`TiH_(1.5-1.8)`and `PdH_(0.6-0.8)`). (iv) Some are relatively light, inexpensive and thermally unstable which makes them useful for hydrogen STORAGE applications. Example, `TiH_(2)`, `ZrH_(2)`, `ZnH_(2)`. |
|
| 9. |
Which of the following shows geometrical isomerism ? (i)CHCl=CHCl , (ii)CH_2=C Cl_2 , (iii) C Cl_2=CHCl Give structures of its cis- and trans-forms. |
Answer» Solution :Only (i), i.e., CICH=CHCl has two DIFFERENT substituents on each carbon atom of the double BOND and HENCE show GEOMETRICAL isomerism.  The other two compounds, i.e., (ii) and (iii) do not show geometrical isomerism because one of the carbon atoms of the double bond in each case has two IDENTICAL atoms, i.e., Cl atoms. |
|
| 10. |
Which of the following oxide is unstable |
|
Answer» `Li_(2)O` |
|
| 12. |
The term that corrects for the attractive forces present in a real gas in the van der Waals equation is |
|
Answer» NB |
|
| 13. |
What , is the numberof photonsof ligthwith awavelengthof 400pm thatprovide1 j ofenergy |
|
Answer» Solution :Wavelength`(LAMBDA)= 400 pm:. , 1 pm = 10^(12) m` Energy(E ) = 1J If numberof photon =N energyof Nphoton= `(Nhc )/(lambda ) = E` `N = (E xx lambda )/( hc )` `=(11xx 4000 xx 10^(12)m)/( 6.626 xx 10^(34) j s xx3.0xx 10^(8) MS^(-1))` 2.012 `xx10^(16)`number ofphoton |
|
| 14. |
What is the correct representation of reaction occurring when HCl is heated with MnO_(2) ? |
|
Answer» `MnO_(4)^(-)+5Cl^(-)+8H^(+)RARR Mn^(2+)+5Cl^(-)+5H_(2)O` or in IONIC form `MnO_(2)+2Cl^(-)+4H^(+)rarr Mn^(2+) + Cl_(2) + 2H_(2)O` |
|
| 15. |
Which of the following is/are correct for a solution of a particular concentrations ? |
|
Answer» MOLARITY is always LESS than molality |
|
| 16. |
Whichof the followingis a temporaryeffect? |
|
Answer» electromericeffect |
|
| 17. |
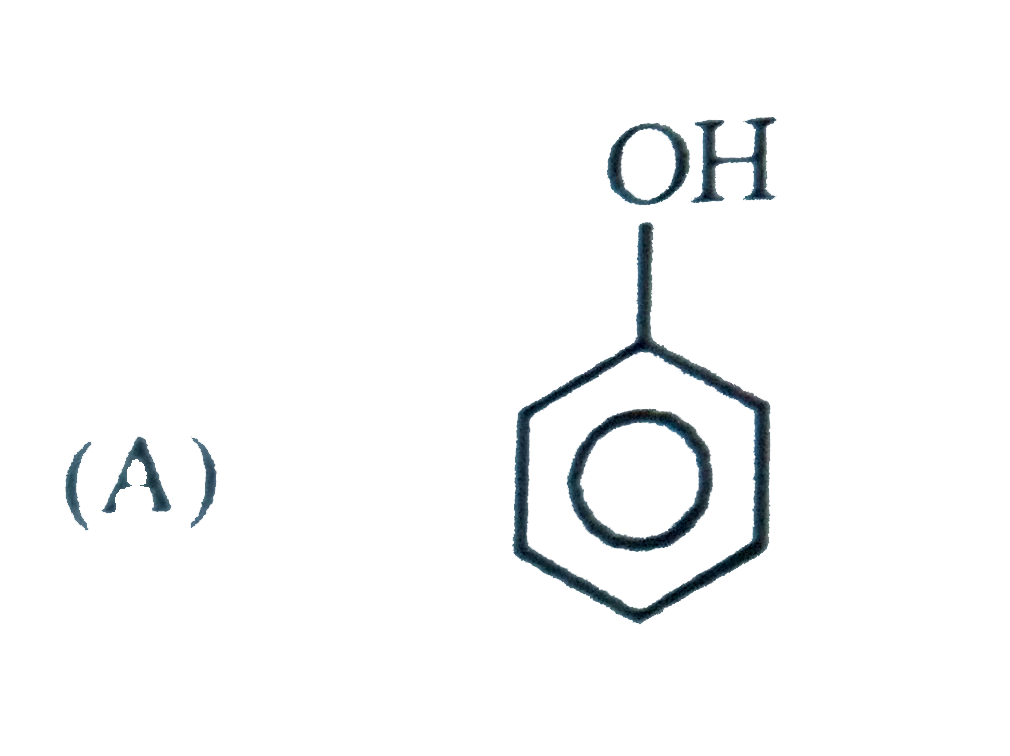
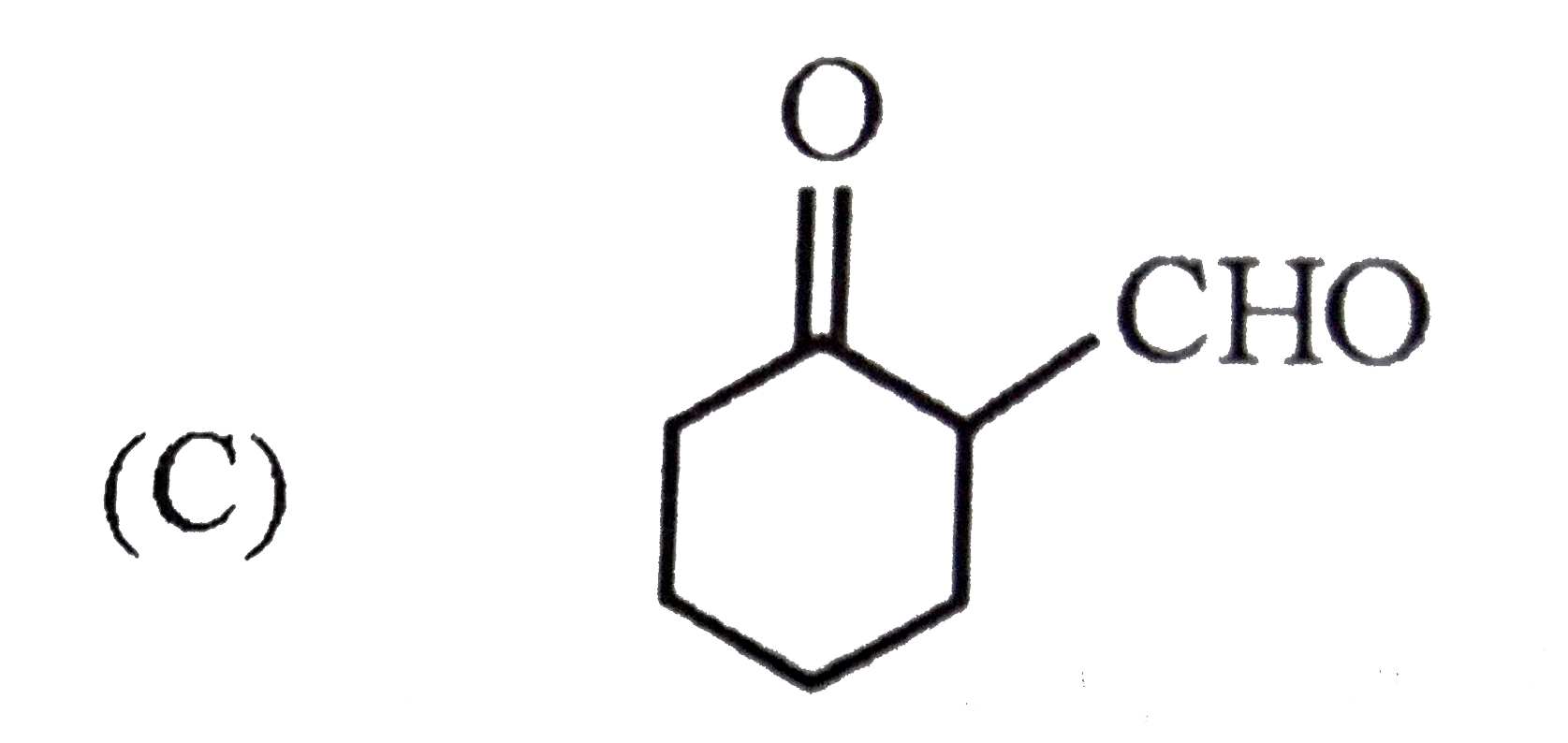
Which of the following will give characteristic colour with FeCl_(3) ? |
|
Answer»
|
|
| 18. |
What do you understand by laminar flow of a liquid ? |
| Answer» Solution :The type of flow in which there is a REGULAR gradient of VELOCITY in going from ONE layer to the next is called LAMINAR flow. | |
| 19. |
Which of the following on reductive ozonolysis will give only glyoxal ? |
|
Answer» ethene |
|
| 20. |
Which organic structure among the following is not an isomer of the compound CH_(3)-CO-CH_(2)CH_(2)CH_(2)CH_(3) ? |
|
Answer» `CH_(3)CH_(2) OCH = CHCH_(2)CH_(3)` |
|
| 21. |
Which of the following kinds of catalysis can be explained by the adsorption theory? |
|
Answer» HOMOGENEOUS catalysis |
|
| 22. |
Which of the following cannot decompose on heating to give CO_(2) in a dry test tube ? |
|
Answer» `Li_(2)CO_(3)` |
|
| 24. |
Which is not a system of units for physical quantities ? |
|
Answer» PPM |
|
| 25. |
Which of the following solutions will have maximum amount of NaOH? |
|
Answer» 4 L of `0.1` M NaOH solution |
|
| 26. |
Which is not a property of H_(2)O_(2) |
|
Answer» Conc `H_2 O_2` solutionisacidicin NATURE |
|
| 27. |
Write the reasonance structure of CO_(3)^(2-) and HCO_(3)^(-). |
Answer» Solution :RESONANCE STRUCTURE of `CO_(3)^(2-)` ions :  Resonance structure of `HCO_(3)^(-)` ion : 
|
|
| 28. |
What is the use of burette in laboratory ? |
|
Answer» To MEASURE VOLUME |
|
| 29. |
Which of the following species does not exist? |
|
Answer» `[BF_6]^(3-)` |
|
| 30. |
Which bond of ethyl alcohol undergoes cleavage during its reaction with sodamide? |
|
Answer» `C-C` |
|
| 31. |
Which one of the following will have largest number of atoms ? (i) 1 g Au_((s)) (ii) 1g Na_((s)) (iii) 1 g Li_((s)) (iv) 1 g Cl_(2(g)) |
|
Answer» `=(1)/(197)xx6.022xx10^(23)` Au atom (ii) 1 gm `Na=(1)/(23)` mol Na molecule `=(1)/(23) xx 6.022 xx 10^(23)` Na atom (iii) 1 gm `Li=(1)/(7)` mol Li molecule `= 1/7 xx 6.022 xx 10^(23)` Li atom (IV) 1 gm `Cl_(2) = (1)/(71) mol Cl_(2)` molecule `=(1)/(71) xx 6.022 xx 10^(23)` `=(2)/(71) xx 6.022 xx 10^(23) Cl_(2)` atom |
|
| 32. |
Which one of the following is not a property of hydrophilic sols |
|
Answer» High concentrations of DISPERSED phase can be easily attained |
|
| 33. |
What happens when a. Borax is heated strongly. b. Boric acid is added to water. c. Aluminium is treated with dil NaOH. d. BF_(3) is reacted with ammonia. |
|
Answer» Solution :a.When borax is strongly heated, a transparent glassy bead which CONSISTS of sodium metaborate and boric anhydride is formed. `underset("Borax")(Na_(2)B_(4)O_(7).10H_(2)O)OVERSET(Delta)toNa_(2)B_(4)O_(7)+10H_(2)O` `Na_(2)B_(4)O_(7)underset("RED Heat")overset(Delta)tounderset(ubrace({:(" Sodium,Boric"),("metaborate,anhydride"):})_("Transport glassy bead"))(2NaBO_(2)" + "B_(2)O_(3))` b. Boric acid behaves as a Lewis acid and hence accepts `overset(ө)OH` ion form `H_(2)O` and realeses `H^o+` in the solution. c. `2Al_((s))+underset(("dil"))(NaOH_((aq)))+6H_(2)Oto2Na^(o+)[Al(OH)_(4)]_((aq))^()+3H_(2(g)) or underset("Sodium meta-aluminate(III)")(Na_(2)AlO_(2).2H_(2)O)` d.`F_(3)B+:NH_(3)to[F_(3)Blarr:NH_(3)]` `BF_(3)` is a Lewis acid and accepts a pair of electron from `N` of `NH_(3)` molecule to form an adduct, `[F_(3)Blarr:NH_(3)]` 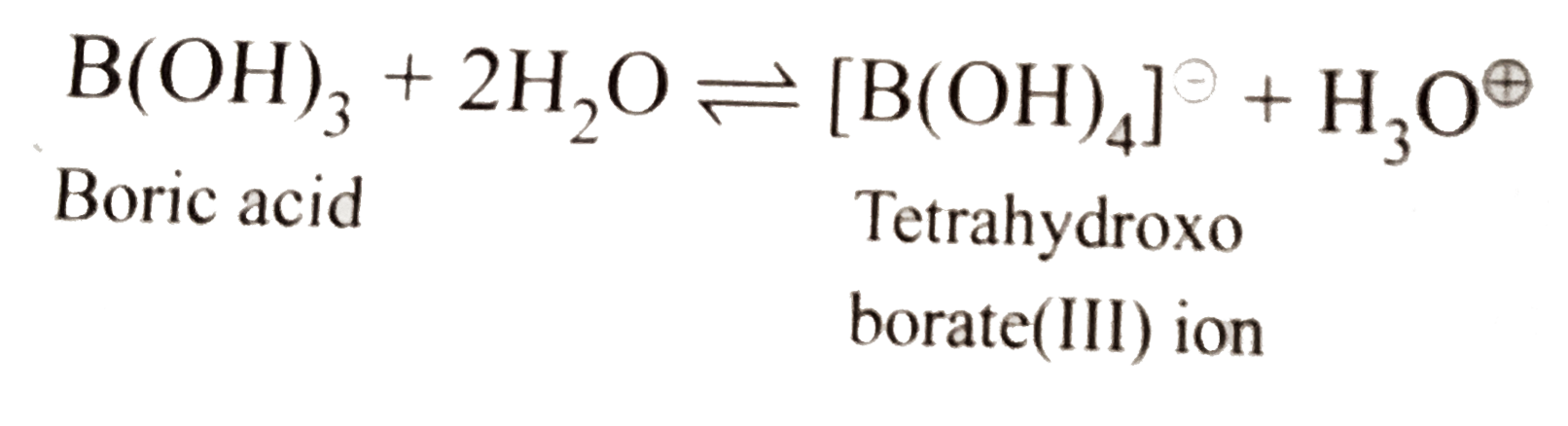
|
|
| 34. |
To what volume must 50 mL of 3.50 M H_2SO_4 be diluted in order to make 2 M H_(2)SO_(4) ? [Hint: Use Rule 5] |
|
Answer» |
|
| 35. |
Write the formal charges on atoms in CO_(3)^(2-) and HNO_(3). |
|
Answer» Solution :`CO_(3)^(2-)` (carbonate ion ) : TOTAL valence electron= C (4) + three O (18) + (-2) charge (2) = 24 Formal charge = `("valence" e^(-)) - ("lone pair" e^(-)) -` (no. of bond)  F = x- nb-b F = (x - nb - b ) `therefore ` Formal charge of C = 4 - 4 = 0 (`because` Total relation with C) Formal charge of O (1) = 6 - 6 -1 = - 1 Formal charge of O (2) = 6 - 4 - 4 = 0 Formal charge of O (3)=6- 6 -1 = -1 Valence`e^(-) ` of O = 6 Two bonding andsix non boding `e^(-) ` with single bond contain O. Four non boding and two boding `e^(e)` on double bond containing O. So, structure : Formal charge on atoms of `HNO_(3)` : F =(x - nb - b) O, N and H the valence `e^(-)` 6,5 and 1 are respectively. Formal charge of O (2) = 6 - 6 - 1 = -1 Formal charge of O (1) = 6 - 4 - 2 = 0 Formal charge of O (3) = 6 - 4 - 2 = 0 Formal charge of N = 5 - 0 - 4 = + 1 Formal charge of H = 1 - O - 1 = O So, structure of `HNO_(3)` with formal charge : 
|
|
| 36. |
Write ionic equilibrium and equation of solubility product of concentrated solution of Barium sulphate (BaSO_4). |
|
Answer» SOLUTION :Ionic equilibrium : `BaSO_(4(s)) hArr Ba_((AQ))^(2+) + SO_(4(aq))^(2-)` `K_(sp)=([Ba^(2+)][SO_4^(2-)])/([BaSO_4])` For SOLID `BaSO_4 , [BaSO_4]` = constant `therefore K_(sp)=[Ba^(2+)][SO_4^(2-)]` |
|
| 37. |
Which is maximum stable ? |
|
Answer» But `1`- ene |
|
| 38. |
What do you understand by the term mole ? |
| Answer» Solution :The MOLE is defined as the amound of a substance which CONTAINS `6.02 xx 10^(23)` particles such as atoms, moleculs or ions. It is represendted by the SYMBOL ..n... | |
| 39. |
Which of the following halogen exchange reaction will occur in acetone? |
|
Answer» `R-I+NaCl` |
|
| 40. |
Write the state of hybridization of all the atoms in CH_(2)=C=CH_(2) and draw its orbital structure. Do the two atoms on C_(1) lie in the same plane in which hydrogens on C_(3) lie. Explain. |
|
Answer» Solution :The gross structure of allene `(C_(3)H_(4))` is `OVERSET(1)(C)H_(2)=overset(2)(C)=overset(3)(C)H_(2)` The carbon atoms 1 and 3 are `sp^(2)`-hybridized SINCE each one of them is joined by a double bond. In CONTRAST, carbon atom 2 is sp-hybridized since it has two double bonds. Thus, the two `pi`-bonds in allene like in acetylene are perpendicular to each other as shown below.  whereas `H_(c)` and `H_(d)` lie in the plane of the paper while `H_(a)` and `H_(B)` lie in a plane perpendicular to the plane of the paper. Thus, the molecule as a WHOLE is non-planar. |
|
| 41. |
Which of the following is more soluble in enther? |
|
Answer» `BeCl_(2)` |
|
| 42. |
What will be the conjugate bases for the following Bronsted acids : HF, H_2SO_4 and HCO_3? |
|
Answer» Solution :Conjugate base = ACID - `H^+` The conjugate bases should have one proton and +1 charge LESS than acid. Conjugate base ofHF=`(HF-H^+)=F^-` Conjugate base of `H_2SO_4 = (H_2SO_4-H^+) =HSO_4^-` Conjugate base of `HCO_3^(-) = (HCO_3^(-)- H)=CO_3^(2-)` |
|
| 44. |
Which of the following exist as polymer in solid state? |
|
Answer» `NaHCO_(3)` |
|
| 45. |
When phenol is made to react with PCl_(5) in presence of pyridine , the resulting product contains. |
|
Answer» `C_(6)H_(5)Cl` only |
|
| 46. |
Which is the correct lewis structure of BF_(3)? |
|
Answer» `:UNDERSET(* *)OVERSET(* *)F - underset(* *)underset( :F: )underset(|)B-underset(* *)overset(* *)F:` |
|
| 47. |
What are boranes ? Give chemical equation for the preparation of diborane. |
|
Answer» SOLUTION :Boranes are the binary compounds of boron and hydrogen like an Alkanes. These are covalent hydrides of formula `B_2H_6` called diborane. PREPARATION of diborane : It is prepared by treating boron trifluoride with `LiAlH_4` in diethyl ETHER. `4BF_3 + 3LiAlH_4 to 2B_2H_6 + 3LiF + 3AlF_3` `B_2H_6` is produced on an industrial scale by the REACTION of `BF_3` with NaH (Sodium HYDRIDE) `2BF_3 + 6NaH overset"450 K"to B_2H_6 + 6NaF` |
|
| 48. |
Which property is lower for deuterium than hydrogen? |
|
Answer» LATENT HEAT of vapourisation |
|
| 49. |
Which of the following hydrocabon has least C-C bond length? |
|
Answer» `C_2 H_6` |
|