Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
One part of an clement A combines with two parts of B (another element). Six parts of clement C combine with four parts of element B. If A and C combine together the ratio of their masses will be governed by: |
|
Answer» LAW of definite proportions |
|
| 2. |
One of the statements of Dalton's atomic theory is given below : " Compounds are formed when atoms of different elements combine in a fixed ratio " Which of the following laws is not related to this statement ? |
|
Answer» LAW of conservation of mass |
|
| 3. |
One of the reactions that take place in producing steel from ore is the reduction of iron (II) oxide by carbon monoxide to give iron metal and CO_(2) Fe O(s) + CO(g)hArr Fe (s) + CO_(2) (g) , K_(p) = 0*265 " at " 1050 K What are the equilibrium partial pressures of CO and CO_(2) at 1050 K if the intial pressures are : p_(co) = 1*4 " atm " and p _(CO_(2))= 0*80" atm" ? |
|
Answer» SOLUTION :` {:(,FeO(s),+,CO(g),hArr,Fe(s),+,CO_(2)) ,("Intial pressures",,,1*4 "atm",,,,0*80"atm"),(,,,,,,,):}` ` Q_(p) = p_(CO_(2))/ (p_(CO))= = (0*80)/1*4 = 0*571 ` As ` Q_(p) gt K_(p) ,` reaction will MOVE in the BACKWARD direction , i.e., pressure of `CO_(2)`will DECREASE and that of CO will increase to attain equilibrim . Hence , if p is the decrease in pressure of ` CO_(2)`increase in pressure of CO= p ` :. "At equilibrium " , p_(CO_(2)) = (0*80- p) "atm" , p_(CO) = (1*4 + p)"atm "` ` K_(p) = p_(CO_(2))/(p_(CO)) :.0*265 = (0*80 - p)/(1*4 +p) or 0*265 (1*4 +p)= 0*80 - p ` or ` 0* 371 + 0*265 p = 0*80 - por 1 * 265 p = 0*49 or p = 0*339 "atm"` ` :. (p_(CO))_(eq) = 1*4 + 0*339 " atm" = 1*739 " atm" , (p_(CO_(2)))_(eq)= 0*80 - 0*339 "atm" = 0*46` |
|
| 4. |
One of the reaction that takes place in producing steel from iron ore is the reduction of iron (II) oxide by carbon monoxide to give iron metal and CO_2. FeO_((s))+CO_((g)) hArr Fe_((s)) + CO_(2(g)) K_p=0.265 atm at 1050 K. What are the equilibrium partial pressures of CO and CO_2 at 1050 K if the initial partial pressures are: p_(CO) = 1.4 atm and 2p_(CO_2)=0.80atm ? |
|
Answer» Solution :`{:("Equillbrium REACTION :", FeO_((s))+ CO_((g)) hArr , Fe_((s))+CO_(2(g))),("Initial partial PRESSURE :", "1.4 ATM","0.80 atm"),("Change in pressure :", "-x atm", "+x atm"),("Partial pressure at EQUILIBRIUM:",(1.4-x),(0.80-x)):}` where , x=1 pressure change in forward reaction. `K_p=(p_(CO_2))/(p_(CO))` `therefore 0.265=(0.80+x)/(1.4-x)` `therefore` 0.265(1.4)-0.265x=0.80 +x `therefore` 0.371 -0.80 =x + 0.265x `therefore` 1.265x=-0.429 `therefore x=(-0.429)/1.265`=-0.3391 atm So, `p_(CO)`=1.4 - (-0.3391)=1.7391 `p_(CO_2)`= 0.80+x =0.80-0.3391 =0.4609 = 0.461 atm |
|
| 5. |
One of the products of burning of coal is a toxic gas 'A'. The gas A combines with the haemoglobin to form a compound 'B'. Concentration of A greater than 750 ppm leads to disease called 'C' in human beings. However, the patient suffering from C' can be treated in a high pressure chamber containing gas 'D'. The main product formed on keeping in gas 'D' is the product E which helps the person to recovery. What are A, B, C, D and E? Write the reactions involved. |
|
Answer» SOLUTION :A= CARBON monoxide (CO) B=Carboxyhaemoglobin (HbCO) C = Anoxia or Asphyxiation D=Oxygen `(O_2)` E = Oxyhaemoglobin `(HbO_2)` Reaction. `C+1/2O_2 to CO` `Hb+CO to UNDERSET"Carboxyhaemoglobin""HbCO"` `HbCO+O_2 to underset"Oxyhaemoglobin"(HbO_2)+CO` |
|
| 6. |
One of the methods for the manufacture of H_2O_2 involve the electrolysis of |
|
Answer» CON. `H_(2)SO_(4)` |
|
| 7. |
One of the major requirement in atomic structure is determination of location of electron inside an atom. The wave mechanical model establishes this in accordance with Heisenberg's uncertainity principle through the concept of orbitals. The orbitals are defined as that '3D' space in which probability of finding electron is maximum and are represented by wave functions Psi_(n,l,m) where n,l and m are quantum number. The variation of Psi is analysed in terms of polar coordinates and hence Psi = f( r, 0 , phi) where 'r' represents radius vector and 0 and phi represents angle (/_) Which the radius vector with x-axis respectively. The expressions of Psi_(r,0, phi) are often given in terms of sigma instead of r where sigma=(2Zr)/(nalpha_(0)) and Z = atomic number and n= shell number. Which of the following statement is incorrect with respect to Psi_((r)) us. r graph for H-atom? |
|
Answer» For a 3d orbital, the graphwill not intersect the x-axis at any finite, non-zero value. |
|
| 8. |
One of the important uses of quicklime is? |
|
Answer» As a purgative |
|
| 9. |
One of the important uses of quicklime is ? |
|
Answer» As a purgative |
|
| 10. |
One of the following reactions involves in decrease of entropy |
|
Answer» Sublimation of dry ice |
|
| 11. |
One of the following combinations illustrate law of reciprocal proportions: |
|
Answer» <P>`N_(2)O_(3), N_(2)O_(4), N_(2)O_(5)` |
|
| 12. |
One of the electrolyte used for the manufacture of H_(2)O_(2) by electrolysis method |
|
Answer» CON. `H_(2)SO_(4)` |
|
| 13. |
One of the assumptions of kinetic theory of gases states that ..there is no force of attraction between the molecules of a gas... How far is this statement correct ? Is it possible to liquefy an ideal gas ? Explain. |
| Answer» Solution :This STATEMENT is correct only for ideal GAS. It is impossible to LIQUEFY an ideal gas as there is no intermolecular forces of attractions between the MOLECULES of an ideal gas. | |
| 14. |
One of the assumptions of kinetic theory of gases is that there is no force of attraction between the molecules of a gas. State and explain the evidence that shows that the assumption is not applicable for real gases. |
| Answer» Solution :Real gases can be liquefied by COOLING and COMPRESSING the gas. This proves that forces FO attraction exist among the MOLECULES. | |
| 15. |
One of the assumptions of kinetic theory of gases is that there is no force of attraction between the molecules of a gas. State and explain the evidence that shows that the assumption is not applicable for real gases. |
|
Answer» Solution :REAL gases casn be liquefied by COOLING and compressing the GAS. This defines that force of attraction is present among the molecules. `….OVERSET(st)(H)-F….underset("H-bond"to)overset(st)(H)-overset(S.)(F)….overset(ST)(H)-F….overset(st)(H)-overset(s)(F)....` |
|
| 16. |
One of the aromatic compound X when heated with powder of Zn gives benzoic acid but on heated with sodalime gives phenol. So give the name of X compound. |
Answer» SOLUTION :The following reaction is possible.  So, it COMPOUND X-COOH and -OH phenolic group may PRESENT. So the compound X may possess the following structures. 
|
|
| 17. |
One of the assumptions of kinetic energy theory of gases states that ''there is no force of attraction between the molecules of a gas''. How far is this statement correct ? Is it possible to liquefy an ideal gas ? Explain. |
| Answer» Solution :The GIVEN statement is correct only for ideal GASES. As there are no intermolecular forces of ATTRACTION AMONG the molecules of an ideal gas, it cannot be LIQUEFIED. | |
| 18. |
One of the alloys of aluminium is looking like gold and was used for preparing coins in France. Write the compositions of the alloy. |
|
Answer» Solution :The ALLOY looking LIKE gold is aluminium bronze. It has a bright yellow COLOUR. The alloy is a corrosion resistant MATERIAL and it can be fused EASILY. The composition of bronze is mainly copper (88-90%) and aluminium (10-12%). |
|
| 19. |
One molecule of haemoglobin combines with 1.53mL of oxygen at body temperature (37^(@)C) and a pressure of 743 torr what is the molar mass of haemoglobin ? . |
|
Answer» |
|
| 20. |
One molecule of alkene 'X' an ozonlysis gave one mole of acctone. The IUPAC of 'X'is |
|
Answer» 2-methyl-1-butene |
|
| 21. |
One mole of un known compound was treated with excess of water and resulted in the evolution of two moles of a readily combustle gas . The resulting solution was treated with CO_(2) and resulted in the formation of white turbidity . The unknown compound is |
|
Answer» `CA` `underset("1 mole")(CaH_(2)) + 2 H_(2) O to Ca(OH)_(2)+ underset("2 moles")(2 H_(2))` `Ca(OH)_(2) + CO_(2)underset(" White turbidity")(CaCO_(3)) + H_(2)O` Since both Ca and `CaH_(2)` react with `H_(2)O` to produce combustible `H_(2)` gas but 1 mole of `CaH_(2)` produces two moles of `H_(2)` , therefore , option (b) is correct . |
|
| 22. |
One mole of the compound (A), C_8H_12 incapable of showing steroisomerism, reacts with only one mole of H_2 hydrogenation over Pd. (A) undergoes ozonolysis to give a symmetrical diketone (B), C_8H_12O_2. What are the structures of (A) and (B) ? |
Answer» SOLUTION :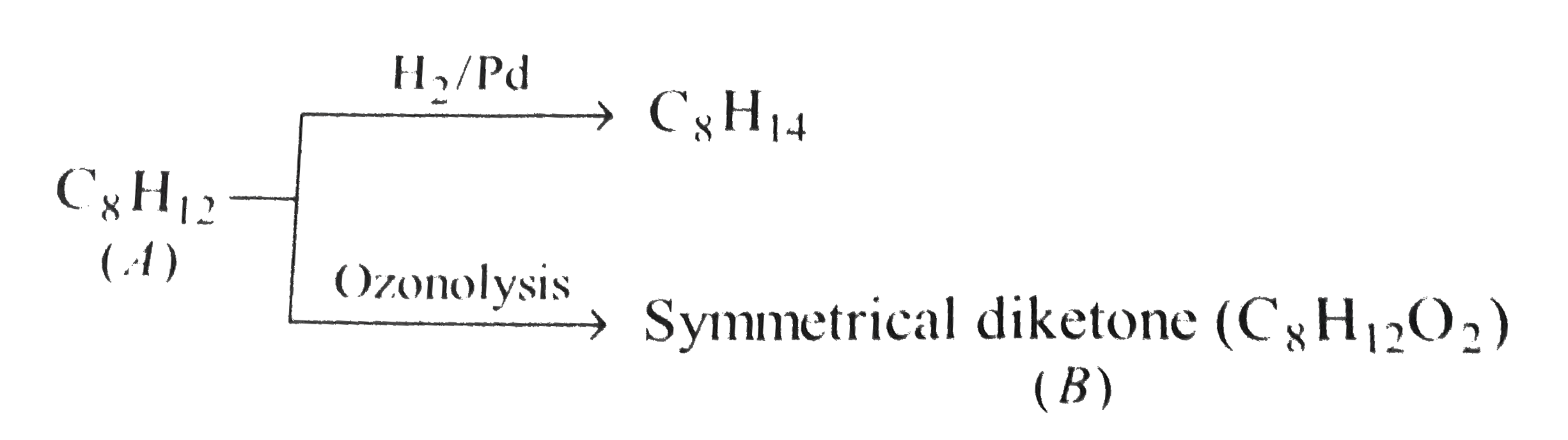 The STRUCTURE of A and B are as FOLLOWS : 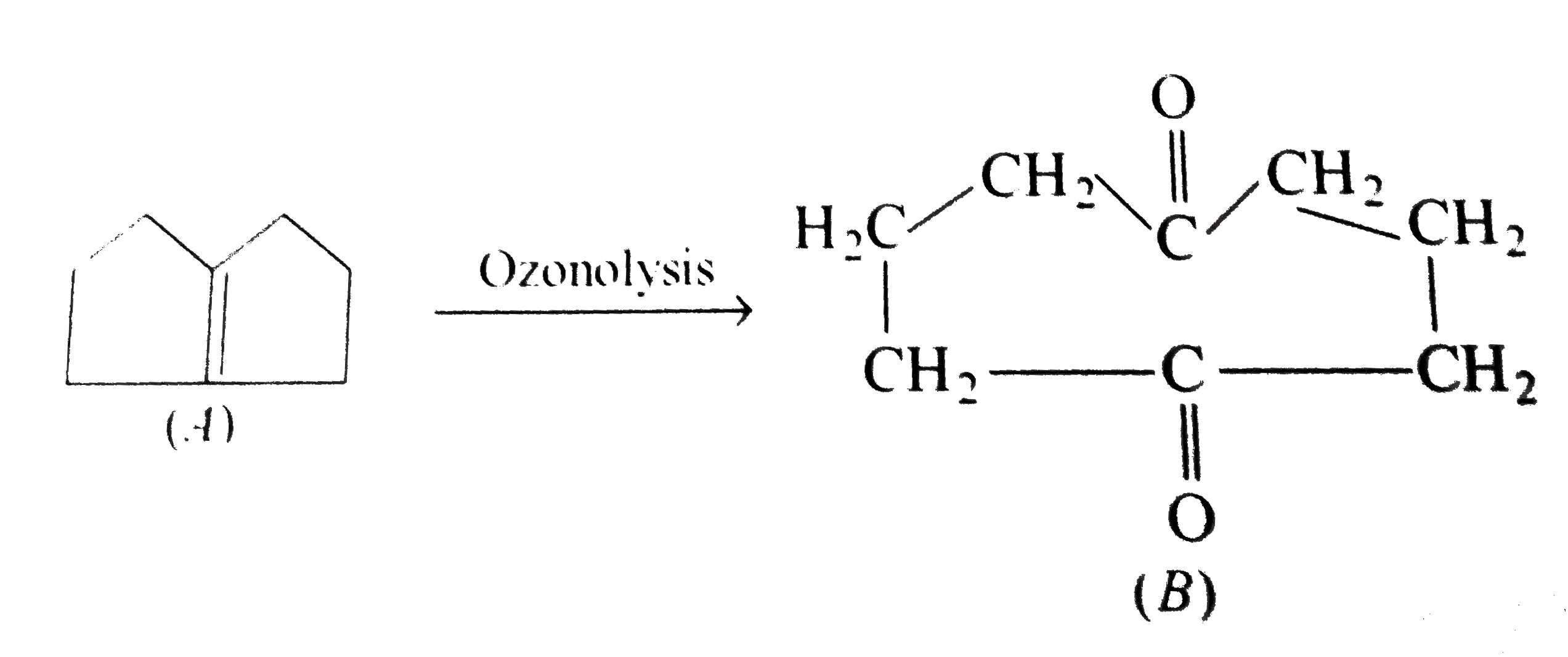
|
|
| 23. |
One mole of solite AB dissolved in 20 moles of water absorbed 15.9 KJ of heat. When one male of same se is dissolved in 250 moles of water 18.58KJof heat is absorbed. Calculate the enthalpy of dilution |
|
Answer» SOLUTION :During DILUTION, HEAT is ABSORBED from `15.9KJ`to `18.58KJ` ENTHALPY of dilution =`18.58KJ-15.9KJ=2.68KJ`. |
|
| 24. |
One mole of sodium represents |
|
Answer» `6.02 XX 10^(23)` ATOMS of sodium |
|
| 25. |
One mole of PCl_(5) is heated in one litre closed container. If 0.6 mole of chlorine is found at equilibrium, calculate the value of equilibrium constant. |
|
Answer» Solution :GIVEN that `[PCl_(5)]_("INITIAL")=("1 mole")/("1 DM"^(3))` `""[Cl_(2)]_(eq)=0.6" mol dm"^(-3)` `PCl_(5)hArrPCl_(3)+Cl_(2)` `[PCl_(3)]_(eq)=0.6" mole dm"^(-3)` `[PCl_(5)]_(eq)=0.4" mole dm"^(-3)` `:.K_(C)=([PCl_(3)][Cl_(2)])/([PCl_(5)])=(0.6xx0.6)/(0.4)` `K_(c)=0.9`. |
|
| 26. |
One mole of PCl_5 is heated in one litre closed container. If 0.6 mole of chlorine is found at equilibium, calculate the value of equilibrium constant. |
|
Answer» Solution :GIVEN that `[PCl_5]_("initial") = (1 mol)/(1 dm^3)` `[Cl_2]_(EQ) = 0.6 mol dm^(-3)` `PCl_5 HARR PCl_3 + Cl_2` `[PCl_3]_(aq) = 0.6 " mole" dm^(-3)` `[PCl_5]_(eq) = 0.4 " mole " dm^(-3)` `:. K_C =([PCl_3][Cl_2])/([PCl_5]) = (0.6 xx 0.6)/0.4` `K_C = 0.9` |
|
| 27. |
One mole of P_(2)O_(5) undergoes hydrolysis as P_(2)O_(5) +H_(2)O to H_(3)PO_(4) The normality of the phosphoric acid formed is (Volume of the solution is 1L) |
|
Answer» 2 `therefore` 1 L solution contains 4 moles of `H_(3)PO_(4)`. `therefore` molarity of `H_(3)PO_(4)` = 4M Normality = Molarity x Basicity = `4 xx 3N = 12N =10/10 xx 1000/100 = 2.5 m |
|
| 28. |
One mole of PCI_(5) is heated in one litre closed container. If 0.6 mole of chlorine is found at equilibrium, calculate the value of equilibrium constant. |
|
Answer» Solution :`[PCl_(5)]_("initial")=(1 mol)/(1 DM^(3))` `[Cl_(2)]_(EQ)=0.6 mol dm^(-3)` `PCl_(5)harrPCl_(3)+Cl_(2)` `[PCl_(3)]_(eq)=0.6 "mole" dm^(-3)` `[PCl_(5)]_(eq)=0.4 "mole" dm^(-3)` `therefore K_(C)=([PCl_(3)][Cl_(2)])/([PCl_(5)])=(0.6xx0.6)/(0.4)` `K_(C)=0.9` |
|
| 29. |
One mole of oxygen (O_(2)) is present in the following mass of sulphuric acid |
|
Answer» 98g |
|
| 30. |
One mole of oxygen gas at STP is equal to ...... |
|
Answer» `6.022xx10^(23)` molecules of oxygen |
|
| 31. |
One "mole" of NH_(4)HS(s) was allowed to decompose in a 1-L container at 200^(@)C. It decomposes reversibly to NH_(3)(g) and H_(2)S(g). NH_(3)(g) further undergoes decomposition to form N_(2)(g) and H_(2)(g). Finally, when equilibrium was set up, the ratio between the number of moles of NH_(3)(g) and H_(2)(g) was found to be 3. NH_(4)HS(s) hArr NH_(3)(g)+H_(2)S(g), K_(c)=8.91xx10^(-2) M^(2) 2NH_(3)(g) hArr N_(2)(g)+3H_(2)(g), K_(c)=3xx10^(-4) M^(2) Answer the following: What is the "mole" fraction of hydrogen gas in the equilibrium mixture in the gas phase? |
|
Answer» `1//4` `K_(C)=27xx10^(-2)X=8.91xx10^(-2)` `X=(8.91xx10^(-2))/(2.7xx10^(-1))=3.3xx10^(-1)` Number of mol of `NH_(3)=0.27` `H_(2)S=0.33` `N_(2)=0.03` `H_(2)=0.09` `X_(H_(2))=0.09/0.72=1/8` `{:(2NH_(3)(g),hArr,N_(2)(g),+,3H_(2)(g)),(3A,,1/3a,,a):}` `K_(c)=((a//3)a^(3))/((3a)^(2))=a^(4)/3xx1/(9a^(2))` `a^(2)/27=3xx10^(-4)` `:. a^(2)=81xx10^(-4)` `a=9xx10^(-2)` |
|
| 32. |
One mole of nitrogen gas at 0.8 atm takes 38 sec to diffuse, while one mole of a xenon fluoride at 1.6 atm takes 57 sec to diffuse. What is the formula of xenon fluoride ? |
| Answer» SOLUTION :`XeF_(6)` | |
| 33. |
One mole of nitrogen gas at 0.8 atm takes 38 s to diffuse through a pin hole whereas one mole of an unknown compound of Xenon with fluorine at 1.6 atm takes 57 s to diffuse through the same hole. Calculate the molecular formula of the compound. |
|
Answer» Solution :Rate of diffusion `prop(P)/(sqrt(M)):. (r_(N_(2)))/(r_(X))=(P_(N_(2)))/(P_(X))xxsqrt((M_(X))/(M_(N_(2))))` But `r_(N_(2))=1//38" mol" s^(-1)` and `r_(X)=1//57" mol" s^(-1)` `:. (r_(N_(2)))/(r_(X))==(1//38)/(1//57)=(57)/(38)=(3)/(2)=(0.8)/(1.6)sqrt((M_(X))/(28))"or" sqrt(M_(X))=3xxsqrt(28)"or" M_(X)=9xx28=252` Atomic mass of F=19 u `"" :. ` No. of F-atmos`=(121)/(19)~-6` HENCE, the compound is `XeF_(6)`. |
|
| 34. |
One mole of N_2O_(4(g)) at 300 K is kept in a closed container under one atmosphere. It is heated to 600 K to decompose to NO_(2(g)). If the resultant pressure at equilibrium is 2.4 atm, then which is correct? |
|
Answer» % DISSOCIATION = 20% Total no. of moles at equlibrium = `1+alpha` from Ideal gas equation `(P_(1))/(P_(2))=(n_(1)T_(1))/(n _(2)T_(2)), underset("equlibrium")underset("before")((P_(1))/(n_(1)T_(1)))=underset("equlibrium")underset("after")((P_(2))/(n_(2)T_(2)))` `(1)/(1 xx 300)=(2.4)/((1+alpha)600) implies alpha=0.2 implies %alpha = 20%` `P_(NO_(2))=(2alpha)/(1+alpha)`, Total pressure = 0.8 `K_(P)=(P_(NO2)^(2))/(P_(N_(2)O_(4)))=0.4` atm |
|
| 35. |
One mole of N_(2)H_4loses 10 mole electrons to from a new compound X Assuming that at the N_2 appears in new compound There is no change in oxidation state of H. What is tha oxidation state on N in X ? |
|
Answer» `:.`X CONTAINS all N-atoms `:. (N^(-2))_(2) rarr (2N)^(a) +10E^(-)` Therefore `2a-(-4) =10 IMPLIES a =+3` (or) `NO_3^(-) +7e^(-) rarr 1//2 N_2 H_4` Therefore , number of ELECTRONS involved in the reduction of `NO_3^(-)`ions is 7 . |
|
| 36. |
One mole of N_(2) H_(4) loses 10 moles of electrons to form a new compound X . Assuming that all the nitrogen appears in the new compound , the oxidation state of nitrogen in X is (there is no change in the oxidation number of hydrogen) |
| Answer» ANSWER :C | |
| 37. |
One mole of N_(2) H_(4) loses 10 mole electrons to form a new compound Y. Assuming that all the N_(2) appearsin new compound, what is oxidation state of N in Y ? There is no charge in oxidant state of H. |
|
Answer» |
|
| 38. |
One mole of N_(2) gas undrgoesfollowingprocess underset(1 atm) underset(0^(@)C)"State" Aunderset ("irreversible process")overset("One step")to underset( 0^(@))underset(2 atm)"stateB"Choose the correct statement (s): |
|
Answer» `DeltaH=0` |
|
| 39. |
One mole of N_(2) and 3 mole of PCI_(5) are placed in a 100 litre vessel heated to 227^(@)c. The equilibrium pressure is 2.05 atm. Assuming ideal behaviour, If K_(p)of the reaction PCl_(5) hArr PCl_(3)+Cl_(2) is y xx 10^(-1) then what is 'y'? |
Answer»  TOTAL no. of moles at equilibrium = 4+X Given, total pressure at equilibrium = 2.05 Now PV=nRT (at equilibrium) `2.05 xx 100=(4+x)0.0821 xx 500`, x = 0.9939 Now degree of dissociation of `PCl_(5)=("Mole dissociation")/("Total mole")=(0.9939)/(3)=0.3313` `:. K_(P)=[(""^(n)PCl_(3) xx ""^(n)Cl_(2))/(""^(n)PCl_(5))] xx [(P)/(sum n)]^(+1)` `=(x^(2))/((3-x)) xx ((2.05)/(4+x))` `=0.2 = 2 xx 10^(-1)=y xx 10^(-1) implies y=2` |
|
| 40. |
One mole of molecules of dioxygen represents |
|
Answer» `6.02xx10^(23)` molecules of oxygen |
|
| 41. |
Disproportionation products of one mole of MnO_(4)^(-2) in aqueous acidic medium are |
|
Answer» 2/3 mole `MnO_(4)^(-) and` 1/3 mole of `MnO_(2)` |
|
| 42. |
0.8 mole of a mixture of CO and CO_(2) requires exactly 40 g of NaOH in solution for complete conversion of all the CO_(2) into Na_(2)CO_(3). How many more moles of NaOH would it require for conversion into Na_(2)CO_(3). If the mixture is completely oxidised to CO_(2)? |
|
Answer» 80g |
|
| 43. |
One mole of methanol, when burnt in oxygen, gives out 723 KJ mol^(-1) heat. If one mole of oxygen is used, what will be the amount of heat evolved ? |
| Answer» ANSWER :C | |
| 44. |
One mole of magnesium nitride on reaction with excess of water gives.... |
|
Answer» One mole of ammonia |
|
| 45. |
One mole of methane (CH_4) contains |
|
Answer» `6.02xx10^(23)` atoms of Hydrogen |
|
| 46. |
One mole of magnesium nitride on reaction with an excess of water gives |
|
Answer» one mole of ammonia |
|
| 47. |
One moleof magnesiumin thevapourstateabsorbed1200 kJ mol^(-1)of energy. It thefirstand secondionizationenergiesof Mg are750and1450 kJ mol^(-1) respectively, the finalcompositionof themixture is |
|
Answer» `31%MG^(+)+ 69 % Mg^(2+)` Energyleft UNUSED= 1200- 750kJ `% ` of`Mg^(+) (g)`convertedinto `Mg^(2+)(g)` `= (450 )/(1450 )xx 100= 31%` Thusthe% of`Mg^(+)(g) =100 - 31 = 69 %` |
|
| 48. |
One mole of lithium nitride is decomposed by H_(2)O and resultant products are neutralised by HCl required is : |
|
Answer» |
|
| 49. |
One mole of KMnO_(4) is used for complete oxidation of FeSO_(4), FeC_(2)O_(4) and H_(2)C_(2)O_(4) respectively and separately. Pick up the correct statement. |
|
Answer» 5 MOLE of `FeSO_(4)` can be oxidized `FeC_(2)O_(4)=bimpliesH_(3)C_(2)O_(4)=c` a) `KMnO_(4)` vs `FeSO_(4)` c) `KMnO_(4)` vs `FeC_(2)O_(4)` `underset(a=5)(1xx5=axx1)""underset(b=5//3)(1xx5=bxx3)` d) `KMnO_(4)` vs `H_(2)CO_(2)O_(4)` `1xx5=Cxx2impliesC=5//2=2.5` |
|