Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
One mole of ice is converted into water at 273 K. The entropies of H_2O_((s)) and H_2O_((l)) are 38.20 and 60.01 J mol^(-1)K^(-1) respectively. The enthalpy change for the conversion is |
|
Answer» `595.4 J ML ^(-1) |
|
| 2. |
One mole of hydrozine loses 10 moles of electrons.If all the hydrogen content is present in the product, the oxidation number of hydrogen in product is +x. What is value of x. |
|
Answer» Solution :Chemical formula of hydrazine is `H_(2)-NH_(2)` Oxidation NUMBER of each N is -2 `2 XX 02 overset("loss of 10e"^(-1))to ?` `or 1 x -2 overset("loss of 5e"^(-1))to ?` Oxidation number of N in the product =-2-(-5)=+3 |
|
| 3. |
One mole of hydrazine loses 10 moles of electrons. If all the nitrogen content is present in the product, what is the oxidation number of nitrogen in the product? |
|
Answer» Solution :Chemical FORMULA of hydrazine is `H_(2)N-NH_(2)` Oxidation number of each Nis `-2` `2xx-2overset("LOSS of "10e^(-))RARR?` or `1xx-2overset("loss of "5E^(-))rarr?` Oxidation number of N in the PRODUCT `= -2-(-5)=+3`. |
|
| 4. |
One mole of H_(2)O and one mole of CO are taken in a 10 litre vessel and heated at 725 K. At equilibrium 40% of water (by mass) reacts with CO according to the equation : H_(2) O (g) + CO(g) hArrH_(2)(g) + CO_(2) (g) calculate the equilibrium constant for the reaction. |
|
Answer» Solution :` [H_(2) O ] = (1 - 0*40 )/10 "MOL"L^(-1) = 0*06 "mol"L^(-1) , [CO] = 0 *06 "mol" L^(-1)` ` [H_(2)] = (0*4)/10 "mol" L^(-1) = 0*04 "mol" L^(-1) , [CO_(2) ] = 0*04 "mol"L^(-1) ` ` K = ([H_(2) ] [ CO_(2)] )/([H_(2)O][CO])=(0*04 xx0*04 )/(0*06 xx 0*06)= 0*444 .` |
|
| 5. |
One mole of H_2O and one mole of CO are taken in 10 L vessel and heated to 725 K. At equilibrium 40% of water (by mass) reacts with CO according to the equation: H_2O_((g)) + CO_((g)) hArr H_(2(g)) + CO_(2(g)) Calculate the equilibrium constant for the reaction. |
|
Answer» Solution :In starting 1 MOL `H_2O` present. Then at equilibrium reaction is occurred by 40% `H_2O` by mass. `therefore` REACTED `H_2O` = 40% of 1 =0.4 mol `{:("Equilibrium reaction:", H_2O_((g)) + ,CO_((g)) hArr , H_(2(g)) + , CO_(2(g))),("Inital mol:", 1,1,0,0),("Change in mol :", -0.4, -0.4 , +0.4 , +0.4),("Mol of equilibrium :", (1-0.4) , (1-0.4) , 0.4 , 0.4),("10 L vessel",0.6,0.6,0.4,0.4),(therefore "mol L"^(-1) "(at equilibrium):",0.6/10,0.6/10,0.4/10,0.4/10),(,0.06,0.06,0.04,0.04):}` The expression of equilibrium CONSTANT `K_c` is , `K_c=([H_2][CO_2])/([H_2O][CO])=((0.04)(0.04))/((0.06)(0.06))=4/9`=0.4444 |
|
| 6. |
One mole of H_(2) and mole of I_(2) are allowed to attain equilibrium in 1 lit container . If the equilibrium mixture contains 0.4 mole of HI . Calculate the equilibrium constant . |
|
Answer» Solution :GIVEN data : `[H_(2)] = 1` mole , `[I_(2)] = 1` mole At equilibrium , [HI] = 0.4 mole `K_(C)` = ? `H_(2) (G) + I_(2) (g) hArr 2HI (g)`  `therefore "" K_(C) = ([HI]^(2))/([H_(2)] [I_(2)]) = (0.4 xx 0.4)/(0.8 xx 0.8) = 0.25` |
|
| 7. |
One mole of fluorine reacted with two moles of hot concentrated KOH. The products formed are KF, H_(2)O and O_(2). Themolar ratio of KF, H_(2)O and O_(2) respectively is |
|
Answer» `1:1:2` |
|
| 8. |
One mole of ethanol is produced reacting graphite, H_(2) and O_(2) together.The standard enthalpy of formation is -277.7 kJ mol^(-1).Calculate the standard enthalpy of the reaction when 4 moles of graphite is involved. |
|
Answer» `-277.7` `Delta_(f)H = -277.7 KJ"mol"^(-1)` Doubling the equation, `4C(s) +O_(2)(g) + 6H_(2)(g to 2C_(2)H_(5)OH(l)` `DeltaH = 2 xx (-277.7) = -555.4 kJ"mol"^(-1)`. |
|
| 9. |
One mole of diatomic ideal gas undergoes change such that pressure drops to 4 times and volume increases to 2 times. Then, calculate the entropy change in system during the process: |
|
Answer» `-0.693 /K` |
|
| 10. |
One mole of compound AB reacts with one mole of a compound CD according to the equation AB_((g)) + CD_((g)) hArr AD_((g)) + CB_((g)) . When equilibrium had been established it was found that 3/4 mole of reactants AB and CD had been converted to AD and CB . There is no change in volume . The equilibrium constant for the reaction is |
|
Answer» `9//16` |
|
| 11. |
One mole of CO_2 corresponds to |
|
Answer» 22.4 L at 1 ATM and `25^@C` |
|
| 12. |
One mole of CO_(2) contains |
|
Answer» |
|
| 13. |
One mole of chlorine combines with certain weight of metal giving 111 gm of its chloride. The same amount of metal can displace. 2gm hydrogen from an acid. The equivalent weight of metal is |
|
Answer» 40 `M +71 = 111` `M = 111 - 71 = 40` 2GM of `H_(2)` displaced by 40gm of meyal 1gm of `H_(2)` displaced by? |
|
| 14. |
One mole of chlorine combines with certain weight of a metal giving 111 g of its chloride. The same amount of metal can displace 2 g of hydrogen from an acid. The atomic weight of the metal is: |
|
Answer» 40 |
|
| 15. |
One mole of CO_(2) at 300 K and1 atm pressure is heated in a closed vessel so that temperature is 500 K is pressureis 5 atm. Then it is cooled so that temperature is 300 K and pressure is 1 atm. What is the change in internal energy of the gas ? |
| Answer» SOLUTION :No CHANGE because internal ENERGYIS a state function and the SYSTEM has returned to ORIGINAL state. | |
| 16. |
7.5g of a gas occupies a volume of 5.6 litres at 0^@ C and 1atm pressure . The gas is |
|
Answer» <P> SOLUTION :`[a = 3.59` atm `dm^(6) mol^(-2)], [b = 4.27 xx 10^(-2) dm^(3) mol^(-1)` and R = `0.082 dm^(3)` atm `deg^(-1) mol^(-1)`]V = 1.32 lit T = `48 + 273 = 321 K` R = `0.082 dm^(3)` atm `K^(-1).mol^(-1)` (n = 1 MOLE) For ideal behaviour , PV = RT `P = (RT)/(V) = (0.082 xx 321)/(1.32)` `P_("ideal") = 19.94` atm For real behaviour , `P = (RT)/(V - b) - (a)/(V^(2))( n = 1 ` mole) For ideal behaviour , For ideal behaviour , `PV = RT` `P = (RT)/(V) = (0.082 xx 321)/(1.32)` ` P_("ideal") = 19.94` atm For real behaviour , `P = (RT)/(V -b) = (a)/(V^(2)) (n = 1`mole) `P = ((0.082 xx 321)/(1.32 - 0.0427)) - ((3.59)/((1.32)^(2)))` = `20.6 - 2.06` `P_("real") = 18.54` atm |
|
| 17. |
One mole of CH_(4) contains |
|
Answer» `6.02 xx 10^(23)` atoms of hydrogen |
|
| 18. |
One mole of calcium phosphorus on reaction with excess water gives |
|
Answer» `1 mol` of phophine `Ca_(3)P_(2)+6H_(2)Orarr3Ca(OH)_(2)+2PH_(3)` |
|
| 19. |
One mole of carbon-dioxide was found to occupy a volume of 1.32 litre at 48^(@) C and at a pressure of 16.4 atm. Calculate the pressure of the gas that would have been expected to behave ideally and non-ideally. |
|
Answer» Solution :`[a = 3.59` atm `dm^(6) mol^(-2)], [b = 4.27 xx 10^(-2) dm^(3) mol^(-1)` and R = `0.082 dm^(3)` atm `DEG^(-1) mol^(-1)`] V = 1.32 lit T = `48 + 273 = 321 K` R = `0.082 dm^(3)` atm `K^(-1).mol^(-1)` (N = 1 mole) For ideal behaviour , PV = RT `P = (RT)/(V) = (0.082 xx 321)/(1.32)` `P_("ideal") = 19.94` atm For real behaviour , `P = (RT)/(V - b) - (a)/(V^(2))( n = 1 ` mole) For ideal behaviour , For ideal behaviour , `PV = RT` `P = (RT)/(V) = (0.082 xx 321)/(1.32)` ` P_("ideal") = 19.94` atm For real behaviour , `P = (RT)/(V -b) = (a)/(V^(2)) (n = 1`mole) `P = ((0.082 xx 321)/(1.32 - 0.0427)) - ((3.59)/((1.32)^(2)))` = `20.6 - 2.06` `P_("real") = 18.54` atm |
|
| 20. |
One mole of calcium phosphide on reaction with excess of water gives. |
|
Answer» One mole of PHOSPHINE |
|
| 21. |
One mole of calcium phosphide on reactioin with excess of water gives |
|
Answer» one MOLE of phosphine |
|
| 22. |
One mole of calcium phosphide on reaction with excess of water gives |
|
Answer» <P>ONE mole of `PH_(3)` |
|
| 23. |
One mole of atoms of oxygen represents |
|
Answer» `6.02xx10^(23)` ATOMS of oxygen |
|
| 24. |
One mole of argon will have least density at |
| Answer» Answer :D | |
| 25. |
One mole of AO_(2)^(-) is oxidised to A^(n+) in acidic solutions by 0.4 mole of permanganate. Calculate the value of n in A^(n+). |
|
Answer» SOLUTION :In a redox REACTION, the product of NUMBER of MOLES and change in oxidation number is same for oxidant and reductant. The change in oxidation number of Mn is 5. The change in oxidation number of A say x. (0.4 mol) (5)=(1 mol), x=2 Oxidation number of A in `AO_(2)^(-)" is "+3`. It is oxidised to `A^(n+)`, where the INCREASES in the oxidation number is 2. It is oxidised to `A^(n+)` where the increases in the oxidation number is 2. The value of n is u=+3 +2= +5. |
|
| 26. |
One mole of any substance contains 6.022xx10^(23)atoms/molecules. Number of molecules of H_(2)SO_(4)present in 100 mL of 0.02M H_(2)SO_(4) solution is ........ . |
|
Answer» `12.044xx10^(20)` molecules Hence, Number of MILLIMOLES of `H_(2)SO_(4)` `=` molarity `xx` volume in mL `= 0.02xx100 =2` millimoles `= 2 xx 10^(-3)` mol Number of molecules `= "number of moles" xx N_(A)` `= 2 xx 10^(-3) xx 6.022 xx 10^(23)` `= 12.044xx10^(20)` molecules |
|
| 27. |
One mole Of an idealtriatomic gas undergoescompression process adiabatically . Then: |
|
Answer» Temperature change of GAS will be more if processis carried out reversibly |
|
| 28. |
One mole of an ideal monoatomic gasundergo process from thestateA to stateC as :underset(300 k)underset(1 atm)"State"Aunderset(process)overset("isochoric")tounderset(600K)"state"Bunderset("process")overset("Adiabaticreversible")tounderset(64 atm)"State" CChoose the correct option (s). |
|
Answer» Pressure of gas at STATE B in 2 atm |
|
| 29. |
One mole of an ideal monoatomic gas is mixed with 1 mole of an ideal diatomic gas. The molar specific heat of the mixture at constant volume is |
|
Answer» Solution :`C_(v)= (3)/(2) R` formonoatomic and `C_(v)= ( 5)/(2) R` for diatomic gases. HENCE, for the mixture, `C_(v) = ((3)/(2)R+(5)/(2)R)/(2)= 2R= 4 cal` |
|
| 30. |
One mole of an ideal monoatomic gas expands isothermally against constant external pressure of 1 atm from initial volume of 1l to a state where its final pressure becomes equal to external pressure. If initial temperature of gas is 300K then total entropy change of system in the above process is: [R =0.082L atm mol^(-1)K^(-1)= 8.3J mol^(-1) K^(-1)] |
|
Answer» 0 `P_(1) = (1 XX R xx 300)/(1) = 24.6` |
|
| 31. |
One mole of an ideal monoatomic gas at temperature T and volume 1 L expands to 2 L against a constant external pressure of one atm under adibatic conditions, then final temperature of gas will be : |
|
Answer» `T+(2)/(3xx0.0821)` |
|
| 32. |
One mole of an ideal gas undergoes reversible isothermal expansion from an initial volume ofV_(1) to a final volume of 10V_(1) and does 10kJof work. The initial pressure was 1xx 10^(7) Pa. (a) Calculate V_(1). (b) If there were 2 moles of gas, what must its temperature have been ? |
|
Answer» Solution :(a) `w = 2.303nRT log. (V_(2))/(V_(1))` `10 xx 10^(3) J= 2.303 xx 1 xx 8.314 xx T xx log . ( 10V_(1))/( V_(1))` or`T = 522.3 K` For initial conditions, ` P_(1)V_(1) = n_(1)RT` i.e., `( 10^(7)Pa) V_91) = 1 xx 8.314 xx 522.3` or ` V_(1) = 4.342 xx 10^(-4) m^(3) = 4.342 xx 10^(2) CM^(3) =434 .2 cm^(3)` Note. We cannot apply the formula `-x = P Delta V` because expansion is not against constant PRESSURE. (B) If there were 2 moles of the gas, applying `P_(1)V_91) = n_(1)RT ` , we GET `( 10^(7) Pa) ( 4.342 xx 10^(-4)m^(3)) =2 xx 8.314 xx T` or`T = 261.1 K`, i.e. half of the first value |
|
| 33. |
One mole of an ideal monoatomic gas at 27^(@)C is subjected to a reversible isoentropic compression until final temperature reaches 327^(@)C. If the initial pressure was 1.0 atm then find the value of ln P_(2): |
|
Answer» SOLUTION :ISOENTROPIC `rArr q= 0 rArr T_(1)^(GAMMA), P_(1)^(1- gamma) = T_(2)^(gamma) P_(1)^(1- gamma)` `(300)^(5//3) xx 1^((1-5//3)) = (600)^(5//3) xx P_(2)^((1- 5//3))` `((1)/(2))^(5//3) = P_(2)^(-(2//3)) rArr (5)/(3)" ln" (1)/(2) = (-2)/(3) ln P_(2)` `rArr ln P_(2) = (5)/(2) ln 2= 1.73` |
|
| 34. |
One mole of an ideal gas is put through a series of changes as shown below in a cyclic process. Name the process A rarrB, BrarrC and C rarrA. |
|
Answer» SOLUTION :`ArarrB` : ISOBARIC process `B RARRC` : ISOTHERMAL process `CrarrA` : Isochoric process |
|
| 35. |
One mole of an ideal gas is put through a series fo changes as shown below in a cyclic process Name the processArarrB,BrarrC and CrarrA. |
|
Answer» Solution :`ARARRB` : Isobaric PROCESS `BRARRC` : ISOTHERMAL process `CrarrA` : Isochoric process |
|
| 36. |
One mole of an ideal gas is subjected to adiabatic expansion form initail state of 16atm, 200K to final pressure of 1 atm against constant external pressure. Select the incorrect optioin if gamma=(4)/(3) for the gas |
|
Answer» `deltaU=0` |
|
| 37. |
One mole of an ideal gas expands at a constant temperature of 300 K from an initial volume of 10 litre to a final volume of 20 litre.The work done in expanding the gas is |
|
Answer» 750 Joule ` = -2.303 xx 1 xx 8.314 xx 300"log "(20)/(10)` = -1728 J `THEREFORE` Work done = 1728 J |
|
| 38. |
One mole of an ideal gas (C_(v,m)=(5)/(2)R) at 300 K and 5 atm is expanded adiabatically to a final pressure of 2 atm against a constant pressure of 2 atm. Final temperature of the gas : |
| Answer» ANSWER :C | |
| 39. |
One mole of an ideal gas expands isothermally and reversibles from 5 atmospheres to 1 atmosphere at 50^(@)C. Calculate the work done. |
|
Answer» <P> SOLUTION :`T = 50^(@)C = 50 + 273 = 323 K, P_(1) = 5` atm, `P_(2) = 1` atm`R = 8.314 J//K//mol , n = 1` MOLE `W = -2.303 nRT log ((P_(1))/(P_(2))) = -2.303 xx 1 xx 8.314 xx 323 xx log((5)/(1))` `= -2.303 xx 1 xx 8.314 xx 323 xx 0.6990` `= -4,322.98 "joule" = -4.323 kJ` |
|
| 40. |
One mole of an idealgas at 300 Kis expanded isothermally from an initial volume of 1litre to 10 litres. The DeltaE for this process is ( R = 2 cal mol^(-1) K^(-1)) |
|
Answer» 16.3cal |
|
| 41. |
One mole of an ideal gas at 300K is expanded isothermally reversibly from an initial volume of 1 litre to 10 litres. The Delta E for this process is (R = 2 cal mol^(-1) K^(-1)) |
|
Answer» 163.7 cal |
|
| 42. |
One mole of an ideal gas at 300 Kin thermal contact with surroundings expandsisothermally from 1.0 L to 2.0 L against a constant pressure of 3.0 atm . In this process, the change in entropy of surroundings ( DeltaS_("surr") ) in JK^(-1)is( 1L atm = 101.3J ) |
|
Answer» 5.763 `DeltaU=0` As `q= DeltaU-W :. Q= - W` As gas EXPANDS against constant pressure. `W_("system") = - P DeltaV =- 3atm ( 2-1) L` `= - 3 L atm =- 3 xx101.3 J` `:. q_("system") = +3 xx 101.3 J` `q_("surr") = - q_("system") = - 3 xx 101.3J` `DeltaS_("surr.") = (q_("surr"))/(T) = ( - 3 xx 101.3J)/(300K) = -1.013J` |
|
| 43. |
One mole of an ideal diatomic gas(C_(v)= 5 cal) was transformedis 25^(@)and 1 L to thestatewhentemperature is 100^(@) and volume10 L.Thenfor the process :(R=2 Calories/mole/K) (takecalopries as unit of energyand kelvinfor tempertaure ) |
|
Answer» `DeltaH=525` |
|
| 44. |
One mole of an ideal gas at 27^(@)C undergoes isothermal expansion reversible from a volume of 10 dm^(3) to a volume of 20 dm^(3). Calculate the work done on the gas. |
|
Answer» Solution :`T = 27^(@)C = 27 + 273 = 300K, V_(1) = 10 dm^(3), V_(2) = 20 dm^(3)` `R = 8.314 J//K//mol` `W = -2.303 nRT log((V_(2))/(V_(1)))` `w = -2.303 xx 1 xx 8.314 xx 300 xx log((20)/(10))` `= -2.303 xx 8.314 xx 300 xx log 2` `= -2.303 xx 8.314 xx 300 xx 0.3010 = -1729 "joule" = -1.723 KJ`. |
|
| 45. |
One mole of an ideal gas at 25^(@)C expands in volume from 1.0 L to 4.0 L at constant temperature. What work (in J) is done if the gas expands against vacuum (P_("external") = 0)? |
| Answer» Solution :`w=-intPdV=0` | |
| 46. |
One mole of aluminium can completely oxidise |
|
Answer» 1.0 MOLE of HCl |
|
| 47. |
One mole of alkene on ozonolysis gives of butanone. The alkene is |
|
Answer» 3,4-dimethylhex-2-ene 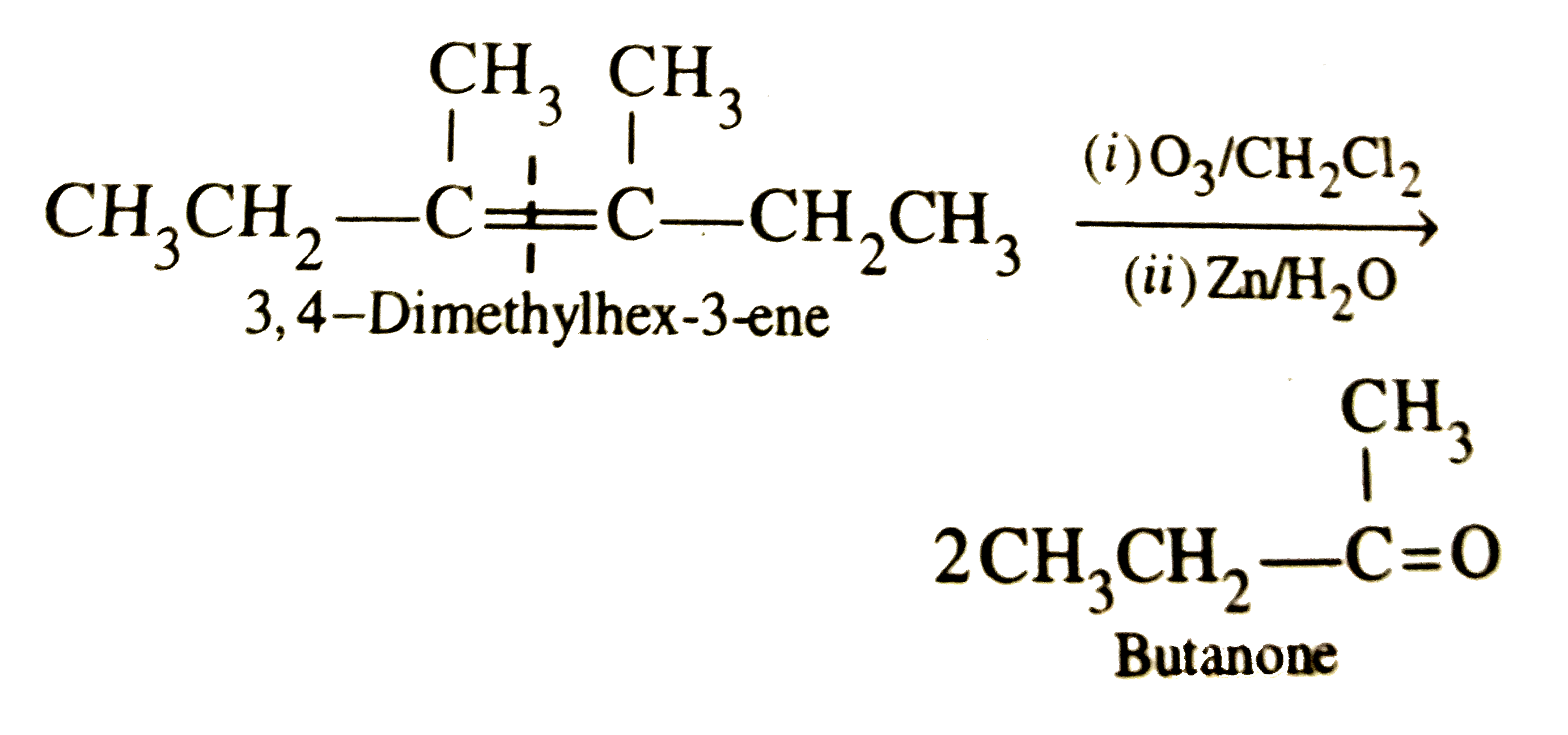
|
|
| 48. |
One mole of A_((g)) is heated to 300^(@) Cin a closed one litre vessel till the following equilibrium is reached. A_((g)) hArr B_((g) .The equilibrium constant of the reaction at 300^(@) C is 4. What is the conc. of B ("in. mole. lit"^(-1))at equilibrium ? |
|
Answer» 0.2  `K_(C)=(x)/(1-x)=4 implies 4-4x=x` 5x-4, x=0.8 M=[B] |
|
| 49. |
One mole of acid is dissolved in one L, solution then from the following which acid will give 1N solution ? |
|
Answer» `H_(2)SO_(4)` |
|